Three
L-hydroxylysine structures have been determined at 100 K by single-crystal X-ray diffraction. High-resolution data using either a laboratory or synchrotron source were collected and subjected to invariom- and independent atom-model (IAM) refinements. Anisotropic displacement parameters (ADPs) obtained from invariom refinement were compared (i) with results from a full multipole and (ii) with an IAM high-order refinement. Differences were visualized with the program
PEANUT and were complemented by quantitative results from a Hirshfeld test. Influences of scale factor differences, and of refinement against
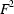 versus
versus 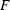
, have been investigated. Systematic errors were observed in the IAM, especially when only low-order data were available. Although these errors were reduced in high-order IAM refinements, they only disappeared in charge density - and likewise - invariom refinements.
Supporting information
CCDC references: 718181; 718182; 718183
Data collection: Bruker AXS APEX2, 2006 for (I); CrysAlis CCD, Oxford Diffraction Ltd., Version 1.171.31.7 (release 18-10-2006 CrysAlis171 .NET) (compiled Oct 18 2006,16:28:17) for (II), (III). Cell refinement: Bruker AXS APEX2 2006 for (I); CrysAlis RED, Oxford Diffraction Ltd., Version 1.171.31.7 (release 18-10-2006 CrysAlis171 .NET) (compiled Oct 18 2006,16:28:17) for (II), (III). Data reduction: Bruker AXS APEX2 2006 for (I); CrysAlis RED, Oxford Diffraction Ltd., Version 1.171.31.7 (release 18-10-2006 CrysAlis171 .NET) (compiled Oct 18 2006,16:28:17) for (II), (III). Program(s) used to solve structure: SHELXS (Sheldrick, 1997) for (I). For all compounds, program(s) used to refine structure: Koritsanszky et al., (2003); molecular graphics: Koritsanszky et al., (2003); software used to prepare material for publication: Koritsanszky et al., (2003).
Crystal data top
| C6H15N2O3·Cl | F(000) = 424.0 |
| Mr = 198.65 | Dx = 1.428 Mg m−3 |
| Orthorhombic, P212121 | Synchrotron radiation, λ = 0.4750 Å |
| Hall symbol: P 2ac 2ab | Cell parameters from 38562 reflections |
| a = 5.4315 (1) Å | θ = 2.4–39.9° |
| b = 9.8019 (1) Å | µ = 0.39 mm−1 |
| c = 17.3505 (2) Å | T = 100 K |
| V = 923.72 (2) Å3 | Rectangle, colorless |
| Z = 4 | 0.19 × 0.18 × 0.11 mm |
Data collection top
Bruker D8
diffractometer | 10455 independent reflections |
| Radiation source: synchrotron | 9078 reflections with F > 3u(F) |
| Si (111) monochromator | θmax = 40.0°, θmin = 1° |
| ω scans | h = −14→14 |
Absorption correction: empirical (using intensity measurements)
SADABS (Version 05/2007), G. S. Sheldrick, 2007 | k = −26→26 |
| Tmin = 0.655, Tmax = 0.748 | l = −46→46 |
| 232425 measured reflections | |
Refinement top
| Refinement on F | 0 restraints |
| Least-squares matrix: full | 0 constraints |
| R[F2 > 2σ(F2)] = 0.031 | H-atom parameters constrained |
| wR(F2) = 0.034 | w1 = 1/[s2(Fo)] |
| S = 1.90 | (Δ/σ)max < 0.001 |
| 9078 reflections | Δρmax = 1.67 e Å−3 |
| 169 parameters | Δρmin = −0.28 e Å−3 |
Special details top
Geometry. As part of the invariom modelling procedure bond distances to hydrogen atoms
were set to values from geometry optimizations of model compounds that mimick
the chemical environment of the hydrogen atom. These values are very similar
to bond distances obtained from neutron diffraction. |
Refinement. Refinement was performed on F with the program XDLSM. Hydrogen atom
positions were obtained by elongating the bond length along the vector of the
atom they are attached to and their refined positions from the independent
atom model. The following dummy atoms were used: DUM0 0 0 0 0 0 DUM1 0.07212 -
0.02361 - 0.02848 0 0 DUM2 - 0.05429 0.11225 0.01976 0 0 The Flack-parameter
was not refined since the anomalous signal was deemed to be absend for the
synchrotron radiation used. The X—H bond lengths using conventional
scattering factors are usually 10% too short when compared to results from
neutron diffraction. In invariom modelling this error is significantly reduced
by the non-spherical scattering factors. The highest residual peak is next the
the heaviest atom and disappears when the high resolution is reduced. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| CL(1) | 0.44577 (2) | 0.386220 (10) | 0.288400 (10) | 0.01 | |
| O(1) | 0.44924 (7) | −0.00531 (4) | −0.13983 (2) | 0.013 | |
| O(2) | 0.39641 (6) | −0.19193 (4) | −0.06884 (2) | 0.011 | |
| O(3) | −0.30706 (5) | 0.12897 (4) | 0.09962 (2) | 0.011 | |
| N(1) | −0.09186 (6) | −0.18791 (4) | −0.07872 (2) | 0.008 | |
| N(2) | −0.04153 (7) | 0.26456 (4) | 0.21651 (2) | 0.01 | |
| C(1) | 0.31669 (6) | −0.08753 (4) | −0.10341 (2) | 0.008 | |
| C(2) | 0.03884 (6) | −0.06002 (4) | −0.10053 (2) | 0.008 | |
| C(3) | −0.02906 (7) | 0.05632 (4) | −0.04541 (2) | 0.009 | |
| C(4) | 0.06789 (7) | 0.04238 (4) | 0.03728 (2) | 0.009 | |
| C(5) | −0.04951 (7) | 0.15102 (4) | 0.08924 (2) | 0.008 | |
| C(6) | 0.08080 (7) | 0.15998 (5) | 0.16687 (3) | 0.01 | |
| H(3) | −0.3161 | 0.04814 | 0.13087 | 0.024 (5) | |
| H(1A) | −0.05567 | −0.26419 | −0.11742 | 0.025 (4) | |
| H(1B) | −0.27528 | −0.16387 | −0.08028 | 0.026 (5) | |
| H(1C) | −0.05993 | −0.21455 | −0.02249 | 0.017 (4) | |
| H(2A) | −0.04951 | 0.35668 | 0.18888 | 0.033 (6) | |
| H(2B) | 0.05359 | 0.28586 | 0.26606 | 0.018 (4) | |
| H(2C) | −0.20902 | 0.23747 | 0.23889 | 0.027 (5) | |
| H(2) | −0.01089 | −0.03111 | −0.15997 | 0.025 (4) | |
| H(3A) | 0.04623 | 0.15071 | −0.06945 | 0.024 (4) | |
| H(3B) | −0.23008 | 0.05992 | −0.04118 | 0.018 (4) | |
| H(4A) | 0.26897 | 0.0482 | 0.03434 | 0.028 (5) | |
| H(4B) | 0.02994 | −0.05685 | 0.0636 | 0.014 (3) | |
| H(5) | −0.03284 | 0.25013 | 0.06015 | 0.020 (4) | |
| H(6A) | 0.0853 | 0.0606 | 0.19626 | 0.016 (3) | |
| H(6B) | 0.27658 | 0.18769 | 0.16241 | 0.021 (4) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| CL(1) | 0.00947 (2) | 0.01179 (3) | 0.00970 (3) | 0.00066 (3) | 0.00000 (2) | 0.00086 (2) |
| O(1) | 0.00746 (7) | 0.01443 (11) | 0.01577 (11) | −0.00140 (8) | 0.00103 (8) | 0.00588 (9) |
| O(2) | 0.00687 (6) | 0.01125 (10) | 0.01419 (11) | 0.00097 (7) | −0.00021 (7) | 0.00398 (8) |
| O(3) | 0.00684 (6) | 0.01132 (11) | 0.01345 (10) | 0.00011 (7) | 0.00032 (7) | 0.00038 (7) |
| N(1) | 0.00628 (6) | 0.00889 (9) | 0.01013 (9) | −0.00039 (7) | 0.00020 (6) | −0.00017 (7) |
| N(2) | 0.00982 (8) | 0.01146 (10) | 0.00999 (10) | 0.00138 (8) | 0.00028 (8) | −0.00064 (7) |
| C(1) | 0.00570 (7) | 0.00949 (11) | 0.00886 (10) | −0.00008 (7) | 0.00020 (7) | 0.00103 (8) |
| C(2) | 0.00593 (7) | 0.00885 (10) | 0.00845 (9) | 0.00016 (8) | −0.00023 (7) | 0.00056 (7) |
| C(3) | 0.00794 (9) | 0.00855 (10) | 0.01008 (11) | 0.00084 (8) | −0.00072 (8) | 0.00005 (8) |
| C(4) | 0.00833 (9) | 0.00933 (11) | 0.01026 (11) | 0.00138 (8) | −0.00090 (8) | −0.00091 (8) |
| C(5) | 0.00740 (8) | 0.00813 (9) | 0.00992 (10) | 0.00010 (8) | −0.00015 (8) | −0.00042 (7) |
| C(6) | 0.00853 (9) | 0.01060 (12) | 0.01115 (12) | 0.00148 (9) | −0.00099 (8) | −0.00110 (9) |
Geometric parameters (Å, º) top
| O(1)—C(1) | 1.2518 (5) | C(2)—H(2) | 1.1032 |
| O(2)—C(1) | 1.2627 (5) | C(3)—C(4) | 1.5345 (6) |
| O(3)—C(5) | 1.4269 (5) | C(3)—H(3A) | 1.0941 |
| O(3)—H(3) | 0.9614 | C(3)—H(3B) | 1.0948 |
| N(1)—H(1A) | 1.0239 | C(4)—C(5) | 1.5340 (6) |
| N(1)—H(1B) | 1.0241 | C(4)—H(4A) | 1.0948 |
| N(1)—H(1C) | 1.0248 | C(4)—H(4B) | 1.0941 |
| N(2)—H(2A) | 1.0233 | C(5)—C(6) | 1.5241 (6) |
| N(2)—H(2B) | 1.0245 | C(5)—H(5) | 1.0985 |
| N(2)—H(2C) | 1.0241 | C(6)—H(6A) | 1.0997 |
| C(1)—C(2) | 1.5339 (5) | C(6)—H(6B) | 1.1003 |
| C(2)—C(3) | 1.5333 (6) | | |
| | | |
| C(5)—O(3)—H(3) | 104.25 | C(4)—C(3)—H(3B) | 106.41 |
| H(1A)—N(1)—H(1B) | 109.72 | H(3A)—C(3)—H(3B) | 111.78 |
| H(1A)—N(1)—H(1C) | 113.94 | C(3)—C(4)—C(5) | 110.18 (3) |
| H(1B)—N(1)—H(1C) | 104.38 | C(3)—C(4)—H(4A) | 107.10 |
| H(2A)—N(2)—H(2B) | 103.57 | C(3)—C(4)—H(4B) | 113.87 |
| H(2A)—N(2)—H(2C) | 111.64 | C(5)—C(4)—H(4A) | 113.95 |
| H(2B)—N(2)—H(2C) | 100.52 | C(5)—C(4)—H(4B) | 107.08 |
| O(1)—C(1)—O(2) | 124.35 (4) | H(4A)—C(4)—H(4B) | 104.70 |
| O(1)—C(1)—C(2) | 117.98 (4) | O(3)—C(5)—C(4) | 112.12 (3) |
| O(2)—C(1)—C(2) | 117.67 (3) | O(3)—C(5)—C(6) | 110.63 (3) |
| C(1)—C(2)—C(3) | 112.81 (3) | O(3)—C(5)—H(5) | 105.83 |
| C(1)—C(2)—H(2) | 104.81 | C(4)—C(5)—C(6) | 111.49 (3) |
| C(3)—C(2)—H(2) | 109.46 | C(4)—C(5)—H(5) | 108.04 |
| C(2)—C(3)—C(4) | 115.75 (3) | C(6)—C(5)—H(5) | 108.47 |
| C(2)—C(3)—H(3A) | 107.53 | C(5)—C(6)—H(6A) | 111.66 |
| C(2)—C(3)—H(3B) | 107.80 | C(5)—C(6)—H(6B) | 113.64 |
| C(4)—C(3)—H(3A) | 107.67 | H(6A)—C(6)—H(6B) | 103.28 |
Crystal data top
| C6H16N2O3·2(Cl) | F(000) = 248.0 |
| Mr = 235.11 | Dx = 1.366 Mg m−3 |
| Monoclinic, P21 | Mo Kα radiation, λ = 0.7107 Å |
| Hall symbol: P 2yb | Cell parameters from 16801 reflections |
| a = 8.6224 (2) Å | θ = 2.8–57.4° |
| b = 7.0489 (2) Å | µ = 0.55 mm−1 |
| c = 9.8061 (2) Å | T = 100 K |
| β = 106.486 (2)° | Rectangle, colorless |
| V = 571.50 (3) Å3 | 0.37 × 0.25 × 0.22 mm |
| Z = 2 | |
Data collection top
Oxford Diffraction Xcalibur S
diffractometer | 9042 independent reflections |
| Radiation source: Enhance (Mo) X-ray Source | 6540 reflections with F > 3u(f) |
| Graphite monochromator | Rint = 0.036 |
| Detector resolution: 16.0009 pixels mm-1 | θmax = 45.3°, θmin = 2.8° |
| ω scans | h = −17→17 |
Absorption correction: analytical
CrysAlis RED, Oxford Diffraction Ltd.,
Version 1.171.31.7 (release 18-10-2006 CrysAlis171 .NET)
(compiled Oct 18 2006,16:28:17)
Analytical numeric absorption correction using a multifaceted crystal
model based on expressions derived by R.C. Clark & J.S. Reid.
(Clark, R. C. & Reid, J. S. (1995). Acta Cryst. A51, 887-897) | k = −14→14 |
| Tmin = 0.878, Tmax = 0.916 | l = −19→19 |
| 29885 measured reflections | |
Refinement top
| Refinement on F | H-atom parameters constrained |
| Least-squares matrix: full | w1 = 1/[s2(Fo)] |
| R[F2 > 2σ(F2)] = 0.023 | (Δ/σ)max < 0.001 |
| wR(F2) = 0.018 | Δρmax = 0.60 e Å−3 |
| S = 1.73 | Δρmin = −0.25 e Å−3 |
| 6540 reflections | Absolute structure: (Dittrich et al., 2006) |
| 181 parameters | Absolute structure parameter: −0.00 (2) |
| 0 restraints | |
Special details top
Geometry. As part of the invariom modelling procedure bond distances to hydrogen atoms
were set to values from geometry optimizations of model compounds that mimick
the chemical environment of the hydrogen atom. These values are very similar
to bond distances obtained from neutron diffraction. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| CL(1) | 0.68112 (2) | 0.74566 | 0.88733 (2) | 0.018 | |
| CL(2) | −0.65514 (2) | 0.26022 (4) | 0.58001 (2) | 0.018 | |
| O(1) | 0.16852 (8) | 0.93309 (10) | 0.91323 (7) | 0.029 | |
| O(2) | 0.36093 (5) | 0.75261 (12) | 1.05516 (5) | 0.02 | |
| O(3) | −0.24657 (6) | 0.33080 (8) | 0.48082 (5) | 0.018 | |
| N(1) | 0.35366 (7) | 0.49854 (10) | 0.84942 (7) | 0.017 | |
| N(2) | −0.31376 (8) | 0.06809 (10) | 0.66336 (7) | 0.018 | |
| C(1) | 0.26032 (8) | 0.77895 (11) | 0.94447 (7) | 0.016 | |
| C(2) | 0.22081 (8) | 0.64061 (11) | 0.82109 (8) | 0.015 | |
| C(3) | 0.05566 (8) | 0.54548 (12) | 0.80251 (8) | 0.016 | |
| C(4) | −0.01243 (9) | 0.46390 (13) | 0.65301 (8) | 0.017 | |
| C(5) | −0.17532 (8) | 0.36406 (11) | 0.62835 (8) | 0.014 | |
| C(6) | −0.15640 (8) | 0.17076 (12) | 0.70019 (9) | 0.018 | |
| H(1) | 0.2132 | 1.02318 | 0.98734 | 0.031 (3) | |
| H(3) | −0.29036 | 0.44961 | 0.43978 | 0.025 (3) | |
| H(1A) | 0.36036 | 0.42324 | 0.9401 | 0.028 (3) | |
| H(1B) | 0.46356 | 0.56412 | 0.86947 | 0.023 (3) | |
| H(1C) | 0.34122 | 0.4158 | 0.76128 | 0.034 (3) | |
| H(2A) | −0.41305 | 0.15303 | 0.65097 | 0.030 (3) | |
| H(2B) | −0.32068 | −0.03333 | 0.73602 | 0.038 (4) | |
| H(2C) | −0.33534 | −0.00106 | 0.56813 | 0.027 (3) | |
| H(2) | 0.22069 | 0.71412 | 0.72217 | 0.026 (3) | |
| H(3A) | 0.06752 | 0.43872 | 0.88572 | 0.037 (3) | |
| H(3B) | −0.02988 | 0.65101 | 0.8193 | 0.032 (3) | |
| H(4A) | 0.07545 | 0.37179 | 0.62472 | 0.026 (3) | |
| H(4B) | −0.02831 | 0.58302 | 0.57909 | 0.030 (3) | |
| H(5) | −0.2606 | 0.44888 | 0.66748 | 0.023 (3) | |
| H(6A) | −0.07236 | 0.08005 | 0.66417 | 0.036 (3) | |
| H(6B) | −0.11295 | 0.18535 | 0.81647 | 0.034 (3) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| CL(1) | 0.02086 (7) | 0.01358 (7) | 0.01903 (7) | −0.00007 (8) | 0.00780 (5) | 0.00216 (8) |
| CL(2) | 0.01903 (6) | 0.01614 (8) | 0.01750 (7) | 0.00180 (8) | 0.00391 (5) | −0.00263 (8) |
| O(1) | 0.0291 (3) | 0.0192 (3) | 0.0321 (3) | 0.0082 (2) | −0.0070 (2) | −0.0078 (3) |
| O(2) | 0.0240 (2) | 0.0161 (2) | 0.0156 (2) | 0.0016 (3) | 0.0003 (2) | −0.0031 (3) |
| O(3) | 0.0203 (2) | 0.0157 (2) | 0.0148 (2) | −0.0015 (2) | 0.0020 (2) | −0.0010 (2) |
| N(1) | 0.0160 (3) | 0.0162 (3) | 0.0160 (3) | 0.0001 (2) | 0.0044 (2) | −0.0024 (2) |
| N(2) | 0.0193 (3) | 0.0154 (3) | 0.0184 (3) | −0.0003 (2) | 0.0057 (2) | 0.0028 (2) |
| C(1) | 0.0164 (2) | 0.0118 (4) | 0.0190 (3) | 0.0007 (2) | 0.0038 (2) | −0.0013 (3) |
| C(2) | 0.0148 (3) | 0.0140 (3) | 0.0138 (3) | −0.0009 (2) | 0.0026 (2) | −0.0011 (2) |
| C(3) | 0.0144 (3) | 0.0178 (4) | 0.0152 (3) | −0.0017 (2) | 0.0031 (2) | −0.0021 (3) |
| C(4) | 0.0152 (3) | 0.0205 (4) | 0.0144 (3) | −0.0029 (3) | 0.0034 (2) | −0.0014 (3) |
| C(5) | 0.0131 (3) | 0.0143 (3) | 0.0137 (3) | 0.0000 (2) | 0.0030 (2) | −0.0004 (2) |
| C(6) | 0.0162 (3) | 0.0172 (3) | 0.0185 (3) | 0.0011 (2) | 0.0036 (2) | 0.0036 (3) |
Geometric parameters (Å, º) top
| O(1)—C(1) | 1.3279 (9) | C(2)—C(3) | 1.5373 (10) |
| O(1)—H(1) | 0.9596 | C(2)—H(2) | 1.0995 |
| O(2)—C(1) | 1.1962 (8) | C(3)—C(4) | 1.5284 (10) |
| O(3)—C(5) | 1.4216 (9) | C(3)—H(3A) | 1.0933 |
| O(3)—H(3) | 0.9590 | C(3)—H(3B) | 1.0925 |
| N(1)—H(1A) | 1.0230 | C(4)—C(5) | 1.5276 (10) |
| N(1)—H(1B) | 1.0221 | C(4)—H(4A) | 1.0918 |
| N(1)—H(1C) | 1.0229 | C(4)—H(4B) | 1.0922 |
| N(2)—H(2A) | 1.0233 | C(5)—C(6) | 1.5213 (10) |
| N(2)—H(2B) | 1.0229 | C(5)—H(5) | 1.0979 |
| N(2)—H(2C) | 1.0226 | C(6)—H(6A) | 1.0973 |
| C(1)—C(2) | 1.5155 (10) | C(6)—H(6B) | 1.0994 |
| | | |
| C(1)—O(1)—H(1) | 106.37 | C(4)—C(3)—H(3B) | 107.98 |
| C(5)—O(3)—H(3) | 106.74 | H(3A)—C(3)—H(3B) | 106.28 |
| H(1A)—N(1)—H(1B) | 104.29 | C(3)—C(4)—C(5) | 114.25 (6) |
| H(1A)—N(1)—H(1C) | 113.90 | C(3)—C(4)—H(4A) | 111.23 |
| H(1B)—N(1)—H(1C) | 106.83 | C(3)—C(4)—H(4B) | 106.80 |
| H(2A)—N(2)—H(2B) | 106.23 | C(5)—C(4)—H(4A) | 111.04 |
| H(2A)—N(2)—H(2C) | 103.95 | C(5)—C(4)—H(4B) | 108.22 |
| H(2B)—N(2)—H(2C) | 105.75 | H(4A)—C(4)—H(4B) | 104.70 |
| O(1)—C(1)—O(2) | 124.59 (7) | O(3)—C(5)—C(4) | 110.45 (6) |
| O(1)—C(1)—C(2) | 110.98 (6) | O(3)—C(5)—C(6) | 106.39 (6) |
| O(2)—C(1)—C(2) | 124.43 (7) | O(3)—C(5)—H(5) | 107.85 |
| C(1)—C(2)—C(3) | 111.47 (6) | C(4)—C(5)—C(6) | 111.49 (6) |
| C(1)—C(2)—H(2) | 110.17 | C(4)—C(5)—H(5) | 111.76 |
| C(3)—C(2)—H(2) | 109.40 | C(6)—C(5)—H(5) | 108.69 |
| C(2)—C(3)—C(4) | 111.63 (6) | C(5)—C(6)—H(6A) | 111.51 |
| C(2)—C(3)—H(3A) | 108.94 | C(5)—C(6)—H(6B) | 110.90 |
| C(2)—C(3)—H(3B) | 109.11 | H(6A)—C(6)—H(6B) | 109.06 |
| C(4)—C(3)—H(3A) | 112.71 | | |
Crystal data top
| C6H16N2O3·H2O·2(Cl) | Z = 1 |
| Mr = 253.12 | F(000) = 134.00 |
| Triclinic, P1 | Dx = 1.436 Mg m−3 |
| Hall symbol: P 1 | Mo Kα radiation, λ = 0.7107 Å |
| a = 5.4491 (3) Å | Cell parameters from 34253 reflections |
| b = 7.3962 (4) Å | θ = 2.6–51.4° |
| c = 8.1337 (4) Å | µ = 0.55 mm−1 |
| α = 79.382 (4)° | T = 100 K |
| β = 73.191 (5)° | Rectangle, colorless |
| γ = 69.544 (5)° | 0.44 × 0.40 × 0.18 mm |
| V = 292.75 (3) Å3 | |
Data collection top
Oxford Diffraction Xcalibur S
diffractometer | 12708 independent reflections |
| Radiation source: Enhance (Mo) X-ray Source | 10693 reflections with F > 3u(F) |
| Graphite monochromator | Rint = 0.022 |
| Detector resolution: 16.0009 pixels mm-1 | θmax = 51.4°, θmin = 2.6° |
| ω scans | h = −11→11 |
Absorption correction: analytical
CrysAlis RED, Oxford Diffraction Ltd.,
Version 1.171.31.7 (release 18-10-2006 CrysAlis171 .NET)
(compiled Oct 18 2006,16:28:17)
Analytical numeric absorption correction using a multifaceted crystal
model based on expressions derived by R.C. Clark & J.S. Reid
(Clark, R. C. & Reid, J. S. (1995). Acta Cryst. A51, 887-897) | k = −16→16 |
| Tmin = 0.845, Tmax = 0.924 | l = −17→17 |
| 45855 measured reflections | |
Refinement top
| Refinement on F | H-atom parameters constrained |
| Least-squares matrix: full | w1 = 1/[s2(Fo)] |
| R[F2 > 2σ(F2)] = 0.020 | (Δ/σ)max < 0.001 |
| wR(F2) = 0.019 | Δρmax = 0.70 e Å−3 |
| S = 1.73 | Δρmin = −0.23 e Å−3 |
| 10693 reflections | Absolute structure: (Dittrich et al., 2006c) |
| 190 parameters | Absolute structure parameter: −0.03 (1) |
| 0 restraints | |
Special details top
Geometry. As part of the invariom modelling procedure bond distances to hydrogen atoms
were set to values from geometry optimizations of model compounds that mimick
the chemical environment of the hydrogen atom. These values are very similar
to bond distances obtained from neutron diffraction. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| CL(1) | 0.80699 | 0.2266 | 0.15254 | 0.017 | |
| CL(2) | 0.73962 (3) | 0.55375 (2) | 0.62145 (2) | 0.016 | |
| O(1) | 1.30652 (8) | −0.53581 (7) | 1.04021 (5) | 0.02 | |
| O(2) | 1.24225 (8) | −0.35346 (7) | 1.25037 (5) | 0.021 | |
| O(3) | 0.78915 (8) | −0.03575 (6) | 0.60225 (5) | 0.016 | |
| O(1W) | 0.21638 (8) | 0.07649 (7) | 0.38356 (5) | 0.019 | |
| N(1) | 0.71901 (8) | −0.18568 (7) | 1.26703 (5) | 0.014 | |
| N(2) | 0.37616 (8) | 0.32178 (7) | 0.55969 (5) | 0.014 | |
| C(1) | 1.16180 (9) | −0.40042 (7) | 1.14575 (6) | 0.013 | |
| C(2) | 0.87696 (9) | −0.30458 (7) | 1.11993 (5) | 0.012 | |
| C(3) | 0.88502 (9) | −0.18022 (7) | 0.94660 (5) | 0.013 | |
| C(4) | 0.60511 (9) | −0.06778 (8) | 0.91499 (5) | 0.013 | |
| C(5) | 0.62716 (9) | 0.06783 (7) | 0.74930 (5) | 0.012 | |
| C(6) | 0.34985 (9) | 0.18560 (8) | 0.71956 (6) | 0.015 | |
| H(1) | 1.47328 | −0.60217 | 1.07327 | 0.034 (3) | |
| H(3) | 0.76776 | −0.16195 | 0.62169 | 0.024 (3) | |
| H(1A) | 0.7381 | −0.26547 | 1.38211 | 0.022 (2) | |
| H(1B) | 0.51589 | −0.13231 | 1.27628 | 0.035 (3) | |
| H(1C) | 0.77532 | −0.06493 | 1.25552 | 0.030 (3) | |
| H(2A) | 0.4727 | 0.41362 | 0.57052 | 0.037 (3) | |
| H(2B) | 0.19222 | 0.41384 | 0.54513 | 0.025 (3) | |
| H(2C) | 0.46362 | 0.24433 | 0.45431 | 0.019 (2) | |
| H(2) | 0.78332 | −0.41633 | 1.12631 | 0.025 (2) | |
| H(3A) | 0.99143 | −0.07779 | 0.94161 | 0.029 (3) | |
| H(3B) | 0.99922 | −0.28077 | 0.84779 | 0.025 (2) | |
| H(4A) | 0.50096 | −0.16796 | 0.90951 | 0.028 (2) | |
| H(4B) | 0.47796 | 0.02397 | 1.01852 | 0.018 (2) | |
| H(5) | 0.73194 | 0.1696 | 0.75142 | 0.029 (3) | |
| H(6A) | 0.25431 | 0.08704 | 0.69692 | 0.028 (3) | |
| H(6B) | 0.22726 | 0.26754 | 0.8311 | 0.036 (3) | |
| H(1W) | 0.09377 | 0.02703 | 0.47576 | 0.042 (3) | |
| H(2W) | 0.13095 | 0.15269 | 0.29419 | 0.038 (3) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| CL(1) | 0.01395 (4) | 0.01147 (5) | 0.02175 (5) | −0.00312 (4) | −0.00709 (4) | 0.00072 (4) |
| CL(2) | 0.01591 (5) | 0.01369 (5) | 0.01557 (4) | −0.00602 (4) | −0.00587 (3) | 0.00126 (3) |
| O(1) | 0.01300 (10) | 0.0200 (2) | 0.0227 (2) | 0.00080 (10) | −0.00680 (10) | −0.00890 (10) |
| O(2) | 0.01350 (10) | 0.0232 (2) | 0.02230 (10) | −0.00130 (10) | −0.00870 (10) | −0.00860 (10) |
| O(3) | 0.01720 (10) | 0.0146 (2) | 0.01240 (10) | −0.00380 (10) | −0.00130 (10) | −0.00020 (10) |
| O(1W) | 0.01180 (10) | 0.0224 (2) | 0.02040 (10) | −0.00460 (10) | −0.00400 (10) | −0.00400 (10) |
| N(1) | 0.01230 (10) | 0.0128 (2) | 0.01190 (10) | −0.00180 (10) | −0.00200 (10) | 0.00010 (10) |
| N(2) | 0.01410 (10) | 0.0123 (2) | 0.01390 (10) | −0.00380 (10) | −0.00590 (10) | 0.00090 (10) |
| C(1) | 0.00980 (10) | 0.0113 (2) | 0.01360 (10) | −0.00190 (10) | −0.00370 (10) | −0.00150 (10) |
| C(2) | 0.00980 (10) | 0.0109 (2) | 0.01250 (10) | −0.00270 (10) | −0.00370 (10) | 0.00010 (10) |
| C(3) | 0.01070 (10) | 0.0144 (2) | 0.01130 (10) | −0.00290 (10) | −0.00340 (10) | 0.00070 (10) |
| C(4) | 0.01100 (10) | 0.0146 (2) | 0.01130 (10) | −0.00340 (10) | −0.00370 (10) | 0.00090 (10) |
| C(5) | 0.01100 (10) | 0.0111 (2) | 0.01200 (10) | −0.00350 (10) | −0.00410 (10) | 0.00010 (10) |
| C(6) | 0.01150 (10) | 0.0163 (2) | 0.01320 (10) | −0.00420 (10) | −0.00460 (10) | 0.00190 (10) |
Geometric parameters (Å, º) top
| O(1)—C(1) | 1.3164 (6) | C(2)—C(3) | 1.5329 (6) |
| O(1)—H(1) | 0.9595 | C(2)—H(2) | 1.0999 |
| O(2)—C(1) | 1.2121 (5) | C(3)—C(4) | 1.5319 (6) |
| O(3)—H(3) | 0.9596 | C(3)—H(3A) | 1.0926 |
| O(1W)—H(1W) | 0.9597 | C(3)—H(3B) | 1.0919 |
| O(1W)—H(2W) | 0.9618 | C(4)—C(5) | 1.5259 (6) |
| N(1)—H(1A) | 1.0224 | C(4)—H(4A) | 1.0926 |
| N(1)—H(1B) | 1.0216 | C(4)—H(4B) | 1.0920 |
| N(1)—H(1C) | 1.0221 | C(5)—C(6) | 1.5217 (6) |
| N(2)—H(2A) | 1.0221 | C(5)—H(5) | 1.0972 |
| N(2)—H(2B) | 1.0220 | C(6)—H(6A) | 1.0985 |
| N(2)—H(2C) | 1.0211 | C(6)—H(6B) | 1.0972 |
| C(1)—C(2) | 1.5235 (6) | | |
| | | |
| C(1)—O(1)—H(1) | 107.84 | C(2)—C(3)—H(3B) | 105.83 |
| H(1W)—O(1W)—H(2W) | 112.00 | C(4)—C(3)—H(3A) | 109.06 |
| H(1A)—N(1)—H(1B) | 105.21 | C(4)—C(3)—H(3B) | 109.62 |
| H(1A)—N(1)—H(1C) | 110.66 | H(3A)—C(3)—H(3B) | 109.86 |
| H(1B)—N(1)—H(1C) | 104.13 | C(3)—C(4)—C(5) | 110.78 (4) |
| H(2A)—N(2)—H(2B) | 103.03 | C(3)—C(4)—H(4A) | 110.32 |
| H(2A)—N(2)—H(2C) | 115.03 | C(3)—C(4)—H(4B) | 111.64 |
| H(2B)—N(2)—H(2C) | 107.50 | C(5)—C(4)—H(4A) | 110.53 |
| O(1)—C(1)—O(2) | 125.06 (4) | C(5)—C(4)—H(4B) | 106.16 |
| O(1)—C(1)—C(2) | 111.94 (4) | H(4A)—C(4)—H(4B) | 107.20 |
| O(2)—C(1)—C(2) | 122.94 (5) | C(4)—C(5)—C(6) | 111.33 (4) |
| C(1)—C(2)—C(3) | 110.09 (4) | C(4)—C(5)—H(5) | 111.93 |
| C(1)—C(2)—H(2) | 109.65 | C(6)—C(5)—H(5) | 107.91 |
| C(3)—C(2)—H(2) | 110.82 | C(5)—C(6)—H(6A) | 108.96 |
| C(2)—C(3)—C(4) | 113.65 (4) | C(5)—C(6)—H(6B) | 109.65 |
| C(2)—C(3)—H(3A) | 108.65 | H(6A)—C(6)—H(6B) | 111.67 |

 journal menu
journal menu











