The crystal structure of the core region of the α′ subunit (α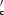 ) of soybean β-conglycinin has been determined at 2.3 Å resolution. α
) of soybean β-conglycinin has been determined at 2.3 Å resolution. α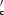 was superimposed on the known crystal structure of the β-conglycinin β subunit with a small root-mean square deviation of 0.77 Å, which is consistent with the high sequence identity of 75.5% between α
was superimposed on the known crystal structure of the β-conglycinin β subunit with a small root-mean square deviation of 0.77 Å, which is consistent with the high sequence identity of 75.5% between α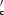 and the β subunit. It is known that the thermal stability of the β subunit is higher than that of the α′ subunit and that their thermal stabilities are conferred by highly homologous core regions. Comparisons of the three-dimensional structures and primary sequences between α
and the β subunit. It is known that the thermal stability of the β subunit is higher than that of the α′ subunit and that their thermal stabilities are conferred by highly homologous core regions. Comparisons of the three-dimensional structures and primary sequences between α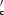 and the β subunit suggest that five factors account for this difference between subunits as regards the difference in thermal stability: (i) the total cavity volume is larger in α
and the β subunit suggest that five factors account for this difference between subunits as regards the difference in thermal stability: (i) the total cavity volume is larger in α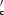 , (ii) the cluster of charged residues at the intermonomer interface is smaller in α
, (ii) the cluster of charged residues at the intermonomer interface is smaller in α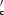 and α
and α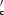 lacks the intermonomer salt bridge of the β subunit, (iii) the solvent-accessible surface is more hydrophobic in α
lacks the intermonomer salt bridge of the β subunit, (iii) the solvent-accessible surface is more hydrophobic in α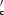 , (iv) there are fewer proline residues in α
, (iv) there are fewer proline residues in α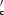 and (v) a loop region between helix 3 and strand J′ in α
and (v) a loop region between helix 3 and strand J′ in α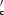 is more flexible owing to the insertion of five additional residues. Although more hydrogen bonds were found in α
is more flexible owing to the insertion of five additional residues. Although more hydrogen bonds were found in α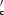 , this difference should be more than compensated for by the combined contributions of these other factors.
, this difference should be more than compensated for by the combined contributions of these other factors.

 journal menu
journal menu











