Molecules of 2-iodobenzaldehyde 4-nitrophenylhydrazone, C
13H
10IN
3O
2, are effectively planar and are linked by an N-H

O hydrogen bond [H

O = 2.04 Å, N

O = 2.905 (6) Å and N-H

O = 166°] and a two-centre iodo-nitro interaction [I

O = 3.361 (4) Å] into a chain of edge-fused
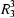
(18) rings. There are no direction-specific interactions between adjacent chains. Molecules of 2-iodobenzaldehyde 2,4-dinitrophenylhydrazone, C
13H
9IN
4O
4, are disordered over two orientations with occupancies of 0.681 (5) and 0.319 (5). In the major orientation, there are no direction-specific intermolecular interactions, while for the minor form, a single C-H

O hydrogen bond generates centrosymmetric dimers.
Supporting information
CCDC references: 208004; 208005
For each of (II) and (III), a finely powdered mixture of the aldehyde and the appropriate phenylhydrazine (1:1 molar ratio) was gently heated on a hot-plate until effervescence ceased. The mixtures were cooled and crystallized from ethanol.
Compound (II) is orthorhombic and the systematic absences permitted Pca21 and Pcam (= Pbcm) as possible space groups. Pca21 was chosen and confirmed by the analysis. Compound (III) is triclinic: space group P-1 was selected and confirmed by the structure analysis. In (III), the sites N1 and I22 are common to both orientations. In the minor orientation of (III), the nitrated ring C911–C916 was treated as a rigid hexagon and all non-H atoms in the minor orientation were refined isotropically. DFIX constraints were applied to the nitro group centred on N914. All H atoms were treated as riding atoms with distances C—H = 0.95 Å and N—H = 0.88 Å.
For both compounds, data collection: KappaCCD Software (Nonius, 1997); cell refinement: DENZO-SMN (Otwinowski & Minor, 1997); data reduction: DENZO-SMN (Otwinowski & Minor, 1997); program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: PLATON (Spek, 2003); software used to prepare material for publication: SHELXL97 (Sheldrick, 1997) and PRPKAPPA (Ferguson, 1999).
(II) 2-Iodobenzaldehyse-4-nitrophenylhydrazone
top
Crystal data top
| C13H10IN3O2 | F(000) = 712 |
| Mr = 367.14 | Dx = 1.900 Mg m−3 |
| Orthorhombic, Pca21 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2c -2ac | Cell parameters from 2800 reflections |
| a = 24.0858 (11) Å | θ = 3.4–27.4° |
| b = 5.0699 (2) Å | µ = 2.50 mm−1 |
| c = 10.5080 (4) Å | T = 120 K |
| V = 1283.16 (9) Å3 | Plate, red |
| Z = 4 | 0.32 × 0.08 × 0.06 mm |
Data collection top
KappaCCD
diffractometer | 2800 independent reflections |
| Radiation source: Rotating Anode | 2082 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.063 |
| ϕ scans, and ω scans with κ offsets | θmax = 27.4°, θmin = 3.4° |
Absorption correction: multi-scan
DENZO-SMN (Otwinowski & Minor, 1997) | h = −31→27 |
| Tmin = 0.502, Tmax = 0.865 | k = −6→6 |
| 8490 measured reflections | l = −13→13 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.036 | H-atom parameters constrained |
| wR(F2) = 0.080 | w = 1/[σ2(Fo2) + (0.0298P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.02 | (Δ/σ)max = 0.002 |
| 2800 reflections | Δρmax = 1.20 e Å−3 |
| 160 parameters | Δρmin = −0.47 e Å−3 |
| 1 restraint | Absolute structure: Flack (1983), 1259 Friedel pairs |
| Primary atom site location: structure-invariant direct methods | Absolute structure parameter: −0.08 (4) |
Crystal data top
| C13H10IN3O2 | V = 1283.16 (9) Å3 |
| Mr = 367.14 | Z = 4 |
| Orthorhombic, Pca21 | Mo Kα radiation |
| a = 24.0858 (11) Å | µ = 2.50 mm−1 |
| b = 5.0699 (2) Å | T = 120 K |
| c = 10.5080 (4) Å | 0.32 × 0.08 × 0.06 mm |
Data collection top
KappaCCD
diffractometer | 2800 independent reflections |
Absorption correction: multi-scan
DENZO-SMN (Otwinowski & Minor, 1997) | 2082 reflections with I > 2σ(I) |
| Tmin = 0.502, Tmax = 0.865 | Rint = 0.063 |
| 8490 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.036 | H-atom parameters constrained |
| wR(F2) = 0.080 | Δρmax = 1.20 e Å−3 |
| S = 1.02 | Δρmin = −0.47 e Å−3 |
| 2800 reflections | Absolute structure: Flack (1983), 1259 Friedel pairs |
| 160 parameters | Absolute structure parameter: −0.08 (4) |
| 1 restraint | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| I22 | 0.876111 (14) | 0.80511 (6) | 0.99980 (7) | 0.02451 (12) | |
| O41 | 0.75761 (18) | −0.9173 (10) | 0.3440 (4) | 0.0298 (10) | |
| O42 | 0.8286 (2) | −0.7960 (8) | 0.2309 (4) | 0.0342 (11) | |
| N1 | 0.8383 (2) | 0.0228 (9) | 0.6790 (5) | 0.0238 (12) | |
| N2 | 0.88431 (19) | 0.1797 (8) | 0.6758 (5) | 0.0181 (11) | |
| N4 | 0.7976 (2) | −0.7716 (10) | 0.3259 (5) | 0.0252 (12) | |
| C11 | 0.8295 (2) | −0.1731 (11) | 0.5902 (5) | 0.0169 (13) | |
| C12 | 0.7819 (3) | −0.3277 (11) | 0.6028 (6) | 0.0203 (14) | |
| C13 | 0.7719 (2) | −0.5242 (10) | 0.5156 (7) | 0.0217 (14) | |
| C14 | 0.8089 (2) | −0.5666 (12) | 0.4170 (5) | 0.0190 (12) | |
| C15 | 0.8567 (2) | −0.4147 (13) | 0.4038 (5) | 0.0211 (13) | |
| C16 | 0.8674 (2) | −0.2158 (9) | 0.4942 (19) | 0.0216 (14) | |
| C21 | 0.9338 (2) | 0.5366 (11) | 0.7712 (5) | 0.0209 (13) | |
| C22 | 0.9396 (2) | 0.7336 (11) | 0.8643 (6) | 0.0206 (13) | |
| C23 | 0.9849 (2) | 0.8953 (13) | 0.8680 (6) | 0.0217 (9) | |
| C24 | 1.0273 (2) | 0.8620 (11) | 0.7729 (6) | 0.0217 (9) | |
| C25 | 1.0228 (3) | 0.6848 (11) | 0.6870 (6) | 0.0259 (10) | |
| C26 | 0.9777 (2) | 0.5148 (11) | 0.6803 (6) | 0.0259 (10) | |
| C27 | 0.8866 (2) | 0.3589 (12) | 0.7629 (6) | 0.0224 (15) | |
| H1 | 0.8136 | 0.0473 | 0.7395 | 0.029* | |
| H2 | 0.8570 | 0.3748 | 0.8224 | 0.027* | |
| H12 | 0.7567 | −0.2975 | 0.6708 | 0.024* | |
| H13 | 0.7396 | −0.6307 | 0.5229 | 0.026* | |
| H15 | 0.8816 | −0.4445 | 0.3352 | 0.025* | |
| H16 | 0.9003 | −0.1126 | 0.4890 | 0.026* | |
| H23 | 0.9883 | 1.0269 | 0.9318 | 0.026* | |
| H24 | 1.0590 | 0.9732 | 0.7740 | 0.026* | |
| H25 | 1.0514 | 0.6689 | 0.6253 | 0.031* | |
| H26 | 0.9761 | 0.3843 | 0.6155 | 0.031* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| I22 | 0.02463 (18) | 0.02820 (19) | 0.02070 (17) | 0.00047 (14) | 0.0017 (3) | −0.0016 (4) |
| O41 | 0.030 (3) | 0.027 (2) | 0.032 (3) | −0.010 (2) | −0.006 (2) | −0.003 (2) |
| O42 | 0.036 (3) | 0.045 (3) | 0.021 (2) | −0.004 (2) | 0.002 (2) | −0.012 (2) |
| N1 | 0.026 (3) | 0.029 (3) | 0.016 (2) | 0.000 (2) | 0.005 (2) | −0.003 (2) |
| N2 | 0.021 (3) | 0.014 (3) | 0.020 (3) | 0.000 (2) | −0.001 (2) | 0.003 (2) |
| N4 | 0.023 (3) | 0.027 (3) | 0.025 (3) | 0.007 (2) | −0.010 (2) | −0.001 (2) |
| C11 | 0.016 (3) | 0.017 (3) | 0.017 (3) | 0.000 (2) | −0.006 (2) | 0.006 (3) |
| C12 | 0.019 (3) | 0.022 (4) | 0.019 (3) | 0.002 (3) | 0.005 (3) | −0.003 (3) |
| C13 | 0.017 (3) | 0.023 (3) | 0.026 (4) | 0.0011 (19) | −0.006 (3) | 0.004 (4) |
| C14 | 0.022 (3) | 0.016 (3) | 0.018 (3) | 0.001 (3) | −0.005 (3) | 0.003 (3) |
| C15 | 0.022 (3) | 0.028 (3) | 0.013 (3) | 0.005 (3) | 0.001 (3) | 0.003 (3) |
| C16 | 0.016 (3) | 0.025 (2) | 0.023 (4) | −0.002 (2) | 0.008 (5) | 0.006 (6) |
| C21 | 0.022 (3) | 0.018 (3) | 0.023 (3) | 0.003 (2) | −0.005 (3) | 0.003 (3) |
| C22 | 0.019 (3) | 0.018 (3) | 0.024 (3) | 0.007 (2) | 0.000 (3) | −0.002 (3) |
| C23 | 0.013 (2) | 0.019 (2) | 0.034 (2) | −0.0023 (17) | −0.0059 (18) | 0.008 (2) |
| C24 | 0.013 (2) | 0.019 (2) | 0.034 (2) | −0.0023 (17) | −0.0059 (18) | 0.008 (2) |
| C25 | 0.021 (2) | 0.030 (3) | 0.027 (2) | 0.0056 (17) | 0.002 (2) | 0.012 (2) |
| C26 | 0.021 (2) | 0.030 (3) | 0.027 (2) | 0.0056 (17) | 0.002 (2) | 0.012 (2) |
| C27 | 0.021 (3) | 0.023 (3) | 0.023 (3) | 0.003 (2) | −0.003 (3) | 0.002 (3) |
Geometric parameters (Å, º) top
| C11—C16 | 1.377 (16) | N1—H1 | 0.88 |
| C11—N1 | 1.380 (7) | N2—C27 | 1.291 (8) |
| C11—C12 | 1.396 (8) | C27—C21 | 1.454 (8) |
| C12—C13 | 1.375 (8) | C27—H2 | 0.95 |
| C12—H12 | 0.95 | C21—C22 | 1.405 (8) |
| C13—C14 | 1.384 (8) | C21—C26 | 1.430 (8) |
| C13—H13 | 0.95 | C22—C23 | 1.364 (8) |
| C14—C15 | 1.391 (8) | C22—I22 | 2.121 (6) |
| C14—N4 | 1.440 (8) | C23—C24 | 1.439 (8) |
| N4—O41 | 1.229 (7) | C23—H23 | 0.95 |
| N4—O42 | 1.252 (7) | C24—C25 | 1.278 (8) |
| C15—C16 | 1.409 (16) | C24—H24 | 0.95 |
| C15—H15 | 0.95 | C25—C26 | 1.387 (8) |
| C16—H16 | 0.95 | C25—H25 | 0.95 |
| N1—N2 | 1.364 (6) | C26—H26 | 0.95 |
| | | |
| C16—C11—N1 | 120.4 (6) | C11—N1—H1 | 119.0 |
| C16—C11—C12 | 121.7 (6) | C27—N2—N1 | 115.3 (5) |
| N1—C11—C12 | 117.8 (5) | N2—C27—C21 | 120.8 (5) |
| C13—C12—C11 | 119.2 (5) | N2—C27—H2 | 119.6 |
| C13—C12—H12 | 120.4 | C21—C27—H2 | 119.6 |
| C11—C12—H12 | 120.4 | C22—C21—C26 | 116.5 (5) |
| C12—C13—C14 | 119.9 (5) | C22—C21—C27 | 124.0 (5) |
| C12—C13—H13 | 120.0 | C26—C21—C27 | 119.4 (5) |
| C14—C13—H13 | 120.0 | C23—C22—C21 | 121.7 (6) |
| C13—C14—C15 | 121.5 (6) | C23—C22—I22 | 117.0 (4) |
| C13—C14—N4 | 119.2 (5) | C21—C22—I22 | 121.2 (4) |
| C15—C14—N4 | 119.3 (5) | C22—C23—C24 | 118.5 (5) |
| O41—N4—O42 | 122.1 (5) | C22—C23—H23 | 120.7 |
| O41—N4—C14 | 118.6 (5) | C24—C23—H23 | 120.7 |
| O42—N4—C14 | 119.3 (5) | C25—C24—C23 | 120.8 (6) |
| C14—C15—C16 | 118.7 (7) | C25—C24—H24 | 119.6 |
| C14—C15—H15 | 120.7 | C23—C24—H24 | 119.6 |
| C16—C15—H15 | 120.7 | C24—C25—C26 | 122.6 (6) |
| C11—C16—C15 | 119.0 (5) | C24—C25—H25 | 118.7 |
| C11—C16—H16 | 120.5 | C26—C25—H25 | 118.7 |
| C15—C16—H16 | 120.5 | C25—C26—C21 | 119.8 (6) |
| N2—N1—C11 | 121.9 (5) | C25—C26—H26 | 120.1 |
| N2—N1—H1 | 119.0 | C21—C26—H26 | 120.1 |
| | | |
| C16—C11—C12—C13 | −1.4 (10) | C15—C14—N4—O41 | 174.6 (5) |
| N1—C11—C12—C13 | 179.9 (5) | C13—C14—N4—O42 | 174.1 (5) |
| C11—C12—C13—C14 | 0.1 (8) | C15—C14—N4—O42 | −5.6 (8) |
| C12—C13—C14—C15 | 0.2 (9) | N2—C27—C21—C26 | 1.0 (8) |
| C12—C13—C14—N4 | −179.5 (5) | C26—C21—C22—C23 | −0.1 (8) |
| C13—C14—C15—C16 | 0.7 (10) | C27—C21—C22—C23 | 179.9 (6) |
| N4—C14—C15—C16 | −179.6 (7) | C26—C21—C22—I22 | 176.7 (4) |
| N1—C11—C16—C15 | −179.0 (7) | C27—C21—C22—I22 | −3.3 (8) |
| C12—C11—C16—C15 | 2.3 (13) | C21—C22—C23—C24 | 0.4 (8) |
| C14—C15—C16—C11 | −1.9 (13) | I22—C22—C23—C24 | −176.5 (4) |
| C16—C11—N1—N2 | −0.2 (9) | C22—C23—C24—C25 | −0.1 (8) |
| C11—N1—N2—C27 | 178.4 (5) | C23—C24—C25—C26 | −0.4 (9) |
| N1—N2—C27—C21 | 179.1 (5) | C24—C25—C26—C21 | 0.7 (9) |
| C12—C11—N1—N2 | 178.6 (5) | C22—C21—C26—C25 | −0.4 (8) |
| C22—C21—C27—N2 | −179.0 (5) | C27—C21—C26—C25 | 179.6 (5) |
| C13—C14—N4—O41 | −5.8 (8) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O41i | 0.88 | 2.04 | 2.905 (6) | 166 |
| Symmetry code: (i) −x+3/2, y+1, z+1/2. |
(III) 2-Iodobenzaldehyde 2,4-dinitrophenylhydrazone
top
Crystal data top
| C13H9IN4O4 | Z = 2 |
| Mr = 412.14 | F(000) = 400 |
| Triclinic, P1 | Dx = 1.978 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 7.8890 (5) Å | Cell parameters from 2924 reflections |
| b = 8.1405 (6) Å | θ = 1.7–27.4° |
| c = 12.176 (1) Å | µ = 2.34 mm−1 |
| α = 83.876 (4)° | T = 120 K |
| β = 83.679 (4)° | Plate, orange |
| γ = 63.151 (3)° | 0.08 × 0.06 × 0.01 mm |
| V = 691.98 (9) Å3 | |
Data collection top
KappaCCD
diffractometer | 2924 independent reflections |
| Radiation source: Rotating Anode | 2187 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.054 |
| ϕ scans, and ω scans with κ offsets | θmax = 27.4°, θmin = 1.7° |
Absorption correction: multi-scan
DENZO-SMN (Otwinowski & Minor, 1997) | h = −10→9 |
| Tmin = 0.835, Tmax = 0.977 | k = −10→10 |
| 7346 measured reflections | l = −15→15 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.047 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.132 | H-atom parameters constrained |
| S = 1.07 | w = 1/[σ2(Fo2) + (0.0694P)2 + 0.0755P]
where P = (Fo2 + 2Fc2)/3 |
| 2924 reflections | (Δ/σ)max < 0.001 |
| 243 parameters | Δρmax = 0.84 e Å−3 |
| 3 restraints | Δρmin = −1.88 e Å−3 |
Crystal data top
| C13H9IN4O4 | γ = 63.151 (3)° |
| Mr = 412.14 | V = 691.98 (9) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 7.8890 (5) Å | Mo Kα radiation |
| b = 8.1405 (6) Å | µ = 2.34 mm−1 |
| c = 12.176 (1) Å | T = 120 K |
| α = 83.876 (4)° | 0.08 × 0.06 × 0.01 mm |
| β = 83.679 (4)° | |
Data collection top
KappaCCD
diffractometer | 2924 independent reflections |
Absorption correction: multi-scan
DENZO-SMN (Otwinowski & Minor, 1997) | 2187 reflections with I > 2σ(I) |
| Tmin = 0.835, Tmax = 0.977 | Rint = 0.054 |
| 7346 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.047 | 3 restraints |
| wR(F2) = 0.132 | H-atom parameters constrained |
| S = 1.07 | Δρmax = 0.84 e Å−3 |
| 2924 reflections | Δρmin = −1.88 e Å−3 |
| 243 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| I22 | 0.53164 (6) | 0.06529 (5) | 0.14789 (3) | 0.0433 (2) | |
| C11 | 0.292 (2) | 0.375 (2) | 0.6377 (12) | 0.035 (4) | 0.681 (5) |
| C12 | 0.1102 (18) | 0.4286 (18) | 0.6936 (10) | 0.023 (2) | 0.681 (5) |
| C13 | 0.0548 (19) | 0.511 (2) | 0.7893 (12) | 0.033 (3) | 0.681 (5) |
| C14 | 0.178 (2) | 0.547 (2) | 0.8405 (10) | 0.028 (3) | 0.681 (5) |
| N14 | 0.121 (3) | 0.636 (2) | 0.9453 (11) | 0.038 (3) | 0.681 (5) |
| O41 | −0.0497 (15) | 0.700 (2) | 0.9755 (14) | 0.048 (4) | 0.681 (5) |
| O42 | 0.2389 (15) | 0.6504 (14) | 0.9943 (8) | 0.064 (3) | 0.681 (5) |
| C15 | 0.3615 (18) | 0.5001 (16) | 0.7941 (10) | 0.033 (3) | 0.681 (5) |
| C16 | 0.4195 (17) | 0.4165 (16) | 0.6933 (9) | 0.023 (2) | 0.681 (5) |
| N12 | −0.0359 (11) | 0.4134 (10) | 0.6527 (6) | 0.0397 (18) | 0.681 (5) |
| O121 | −0.0011 (10) | 0.3349 (8) | 0.5662 (5) | 0.0441 (16) | 0.681 (5) |
| O122 | −0.1960 (11) | 0.4781 (11) | 0.7002 (7) | 0.064 (2) | 0.681 (5) |
| C21 | 0.7318 (13) | 0.1108 (11) | 0.3385 (7) | 0.035 (2) | 0.681 (5) |
| C22 | 0.7557 (13) | 0.0478 (10) | 0.2313 (7) | 0.036 (2) | 0.681 (5) |
| C23 | 0.9306 (14) | −0.0210 (12) | 0.1744 (8) | 0.040 (2) | 0.681 (5) |
| C24 | 1.086 (3) | −0.033 (2) | 0.2216 (14) | 0.041 (2) | 0.681 (5) |
| C25 | 1.0691 (18) | 0.0367 (12) | 0.3246 (8) | 0.041 (2) | 0.681 (5) |
| C26 | 0.8912 (14) | 0.1087 (12) | 0.3821 (8) | 0.045 (2) | 0.681 (5) |
| C27 | 0.5497 (14) | 0.1763 (10) | 0.4046 (8) | 0.0410 (15) | 0.681 (5) |
| N2 | 0.5290 (11) | 0.2364 (9) | 0.4988 (6) | 0.0410 (15) | 0.681 (5) |
| N1 | 0.3590 (8) | 0.2881 (7) | 0.5470 (4) | 0.0373 (12) | |
| C911 | 0.322 (3) | 0.364 (4) | 0.6535 (17) | 0.017 (8)* | 0.319 (5) |
| C912 | 0.456 (2) | 0.386 (3) | 0.7081 (14) | 0.025 (7)* | 0.319 (5) |
| C913 | 0.403 (2) | 0.468 (3) | 0.8086 (14) | 0.014 (5)* | 0.319 (5) |
| C914 | 0.217 (3) | 0.529 (4) | 0.8545 (16) | 0.015 (8)* | 0.319 (5) |
| C915 | 0.084 (2) | 0.507 (4) | 0.7999 (19) | 0.025 (7)* | 0.319 (5) |
| C916 | 0.136 (3) | 0.424 (4) | 0.699 (2) | 0.034 (9)* | 0.319 (5) |
| N912 | 0.6091 (18) | 0.4068 (16) | 0.6531 (10) | 0.021 (3)* | 0.319 (5) |
| O921 | 0.6751 (16) | 0.3353 (15) | 0.5614 (9) | 0.028 (3)* | 0.319 (5) |
| O922 | 0.6951 (18) | 0.4628 (14) | 0.7071 (9) | 0.024 (3)* | 0.319 (5) |
| N92 | 0.260 (2) | 0.2378 (18) | 0.4910 (12) | 0.028 (3)* | 0.319 (5) |
| C927 | 0.338 (2) | 0.167 (2) | 0.3986 (13) | 0.027 (4)* | 0.319 (5) |
| C921 | 0.235 (2) | 0.102 (2) | 0.3353 (13) | 0.023 (3)* | 0.319 (5) |
| C922 | 0.307 (3) | 0.039 (2) | 0.2290 (14) | 0.031 (4)* | 0.319 (5) |
| C923 | 0.214 (2) | −0.036 (2) | 0.1712 (13) | 0.023 (3)* | 0.319 (5) |
| C924 | 0.049 (5) | −0.038 (5) | 0.215 (3) | 0.026 (6)* | 0.319 (5) |
| C925 | −0.022 (3) | 0.028 (2) | 0.3234 (16) | 0.023 (4)* | 0.319 (5) |
| C926 | 0.064 (2) | 0.102 (2) | 0.3799 (15) | 0.024 (3)* | 0.319 (5) |
| N914 | 0.168 (3) | 0.617 (6) | 0.956 (2) | 0.033 (10)* | 0.319 (5) |
| O941 | −0.002 (5) | 0.689 (6) | 0.990 (4) | 0.036 (4)* | 0.319 (5) |
| O942 | 0.291 (3) | 0.616 (3) | 1.0139 (16) | 0.036 (4)* | 0.319 (5) |
| H13 | −0.0708 | 0.5449 | 0.8215 | 0.040* | 0.681 (5) |
| H15 | 0.4472 | 0.5247 | 0.8307 | 0.040* | 0.681 (5) |
| H16 | 0.5444 | 0.3863 | 0.6610 | 0.028* | 0.681 (5) |
| H23 | 0.9444 | −0.0603 | 0.1019 | 0.048* | 0.681 (5) |
| H24 | 1.2077 | −0.0905 | 0.1836 | 0.049* | 0.681 (5) |
| H25 | 1.1765 | 0.0349 | 0.3543 | 0.049* | 0.681 (5) |
| H26 | 0.8767 | 0.1574 | 0.4519 | 0.054* | 0.681 (5) |
| H27 | 0.4441 | 0.1734 | 0.3756 | 0.049* | 0.681 (5) |
| H1 | 0.2805 | 0.2614 | 0.5151 | 0.045* | 0.681 (5) |
| H91 | 0.4691 | 0.2727 | 0.5131 | 0.045* | 0.319 (5) |
| H913 | 0.4945 | 0.4829 | 0.8459 | 0.017* | 0.319 (5) |
| H915 | −0.0433 | 0.5482 | 0.8313 | 0.030* | 0.319 (5) |
| H916 | 0.0448 | 0.4094 | 0.6621 | 0.041* | 0.319 (5) |
| H927 | 0.4601 | 0.1566 | 0.3719 | 0.032* | 0.319 (5) |
| H923 | 0.2676 | −0.0849 | 0.1013 | 0.027* | 0.319 (5) |
| H924 | −0.0172 | −0.0829 | 0.1756 | 0.032* | 0.319 (5) |
| H925 | −0.1336 | 0.0213 | 0.3566 | 0.027* | 0.319 (5) |
| H926 | 0.0087 | 0.1520 | 0.4492 | 0.029* | 0.319 (5) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| I22 | 0.0573 (3) | 0.0536 (3) | 0.0348 (3) | −0.0392 (2) | 0.0088 (2) | −0.01212 (19) |
| C11 | 0.038 (7) | 0.030 (6) | 0.034 (6) | −0.014 (5) | −0.015 (6) | 0.010 (5) |
| C12 | 0.017 (4) | 0.020 (4) | 0.032 (4) | −0.008 (3) | 0.002 (3) | −0.004 (3) |
| C13 | 0.025 (4) | 0.031 (4) | 0.044 (5) | −0.016 (3) | −0.004 (4) | 0.009 (3) |
| C14 | 0.044 (7) | 0.022 (6) | 0.016 (5) | −0.015 (5) | 0.008 (5) | −0.007 (4) |
| N14 | 0.044 (7) | 0.026 (6) | 0.030 (5) | −0.007 (6) | 0.014 (6) | −0.005 (5) |
| O41 | 0.045 (8) | 0.030 (4) | 0.052 (8) | −0.010 (6) | 0.031 (7) | −0.003 (5) |
| O42 | 0.058 (7) | 0.072 (7) | 0.044 (5) | −0.004 (5) | −0.029 (5) | −0.021 (4) |
| C15 | 0.025 (4) | 0.031 (4) | 0.044 (5) | −0.016 (3) | −0.004 (4) | 0.009 (3) |
| C16 | 0.017 (4) | 0.020 (4) | 0.032 (4) | −0.008 (3) | 0.002 (3) | −0.004 (3) |
| N12 | 0.040 (5) | 0.036 (4) | 0.037 (4) | −0.009 (3) | −0.011 (4) | −0.006 (3) |
| O121 | 0.060 (4) | 0.043 (3) | 0.033 (3) | −0.025 (3) | −0.008 (3) | −0.003 (3) |
| O122 | 0.033 (4) | 0.071 (5) | 0.077 (6) | −0.005 (4) | −0.009 (4) | −0.042 (4) |
| C21 | 0.041 (5) | 0.027 (4) | 0.026 (4) | −0.008 (4) | 0.004 (4) | 0.000 (3) |
| C22 | 0.060 (6) | 0.020 (4) | 0.024 (4) | −0.015 (4) | 0.003 (4) | −0.002 (3) |
| C23 | 0.050 (6) | 0.039 (5) | 0.032 (5) | −0.024 (4) | 0.009 (4) | 0.001 (4) |
| C24 | 0.054 (6) | 0.031 (4) | 0.031 (4) | −0.012 (4) | 0.007 (4) | −0.008 (3) |
| C25 | 0.054 (6) | 0.031 (4) | 0.031 (4) | −0.012 (4) | 0.007 (4) | −0.008 (3) |
| C26 | 0.048 (6) | 0.036 (5) | 0.035 (5) | −0.006 (4) | 0.001 (5) | 0.002 (4) |
| C27 | 0.047 (4) | 0.018 (2) | 0.036 (3) | 0.002 (2) | 0.015 (3) | −0.005 (2) |
| N2 | 0.047 (4) | 0.018 (2) | 0.036 (3) | 0.002 (2) | 0.015 (3) | −0.005 (2) |
| N1 | 0.048 (3) | 0.035 (2) | 0.026 (2) | −0.015 (2) | 0.000 (2) | −0.007 (2) |
Geometric parameters (Å, º) top
| I22—C922 | 2.019 (17) | C27—H27 | 0.95 |
| I22—C22 | 2.077 (10) | N2—N1 | 1.300 (9) |
| C11—N1 | 1.306 (14) | N1—N92 | 1.309 (15) |
| C11—C12 | 1.414 (13) | N1—C911 | 1.440 (17) |
| C11—C16 | 1.441 (18) | N1—H1 | 0.88 |
| C12—C13 | 1.339 (13) | N1—H91 | 0.88 |
| C12—N12 | 1.364 (14) | C911—C912 | 1.39 |
| C12—H916 | 0.7519 | C911—C916 | 1.39 |
| C13—C14 | 1.358 (18) | C912—N912 | 1.39 (2) |
| C13—H13 | 0.95 | C912—C913 | 1.39 |
| C13—H915 | 0.8288 | C913—C914 | 1.39 |
| C14—C15 | 1.389 (15) | C913—H913 | 0.95 |
| C14—N14 | 1.460 (12) | C914—C915 | 1.39 |
| N14—O42 | 1.214 (17) | C914—N914 | 1.42 (5) |
| N14—O41 | 1.23 (2) | C915—C916 | 1.39 |
| C15—C16 | 1.398 (12) | C915—H915 | 0.95 |
| C15—H15 | 0.95 | C916—H916 | 0.95 |
| C15—H913 | 1.2302 | N912—O922 | 1.242 (17) |
| C16—H16 | 0.95 | N912—O921 | 1.261 (17) |
| N12—O122 | 1.228 (11) | N92—C927 | 1.28 (2) |
| N12—O121 | 1.231 (9) | C927—C921 | 1.46 (2) |
| N12—H916 | 0.6459 | C927—H927 | 0.95 |
| C21—C26 | 1.410 (14) | C921—C926 | 1.40 (2) |
| C21—C22 | 1.416 (12) | C921—C922 | 1.41 (2) |
| C21—C27 | 1.463 (12) | C922—C923 | 1.42 (2) |
| C22—C23 | 1.368 (13) | C923—C924 | 1.36 (3) |
| C23—C24 | 1.37 (2) | C923—H923 | 0.95 |
| C23—H23 | 0.95 | C924—C925 | 1.43 (4) |
| C24—C25 | 1.40 (2) | C924—H924 | 0.95 |
| C24—H24 | 0.95 | C925—C926 | 1.36 (2) |
| C25—C26 | 1.390 (15) | C925—H925 | 0.95 |
| C25—H25 | 0.95 | C926—H926 | 0.95 |
| C26—H26 | 0.95 | N914—O941 | 1.24 (4) |
| C27—N2 | 1.261 (12) | N914—O942 | 1.26 (4) |
| | | |
| C922—I22—C22 | 121.7 (5) | C11—N1—H1 | 116.6 |
| N1—C11—C12 | 128.0 (13) | C911—N1—H1 | 125.4 |
| N1—C11—C16 | 117.7 (11) | C11—N1—H91 | 122.0 |
| C12—C11—C16 | 114.2 (9) | N92—N1—H91 | 113.9 |
| C13—C12—N12 | 111.2 (10) | C911—N1—H91 | 113.9 |
| C13—C12—C11 | 124.6 (11) | H1—N1—H91 | 120.7 |
| N12—C12—C11 | 124.1 (10) | C912—C911—C916 | 120.0 |
| C12—C13—C14 | 120.1 (10) | C912—C911—N1 | 124.5 (13) |
| C12—C13—H13 | 119.9 | C916—C911—N1 | 115.5 (13) |
| C14—C13—H13 | 119.9 | N912—C912—C913 | 111.3 (12) |
| C13—C14—C15 | 120.7 (8) | N912—C912—C911 | 123.0 (11) |
| C13—C14—N14 | 121.0 (13) | C913—C912—C911 | 120.0 |
| C15—C14—N14 | 118.3 (15) | C912—C913—C914 | 120.0 |
| O42—N14—O41 | 124.1 (12) | C912—C913—H913 | 120.0 |
| O42—N14—C14 | 119.4 (15) | C914—C913—H913 | 120.0 |
| O41—N14—C14 | 116.4 (15) | C915—C914—C913 | 120.0 |
| C14—C15—C16 | 119.6 (10) | C915—C914—N914 | 121.7 (16) |
| C14—C15—H15 | 120.2 | C913—C914—N914 | 118.3 (17) |
| C16—C15—H15 | 120.2 | C916—C915—C914 | 120.0 |
| C15—C16—C11 | 120.8 (9) | C916—C915—H915 | 120.0 |
| C15—C16—H16 | 119.6 | C914—C915—H915 | 120.0 |
| C11—C16—H16 | 119.6 | C915—C916—C911 | 120.0 |
| O122—N12—O121 | 121.8 (8) | C915—C916—H916 | 120.0 |
| O122—N12—C12 | 120.9 (8) | C911—C916—H916 | 120.0 |
| O121—N12—C12 | 117.3 (8) | O922—N912—O921 | 123.2 (13) |
| C26—C21—C22 | 118.0 (8) | O922—N912—C912 | 116.8 (13) |
| C26—C21—C27 | 119.6 (8) | O921—N912—C912 | 118.4 (13) |
| C22—C21—C27 | 122.4 (9) | C927—N92—N1 | 116.5 (13) |
| C23—C22—C21 | 120.7 (9) | N92—C927—C921 | 118.3 (15) |
| C23—C22—I22 | 116.1 (6) | N92—C927—H927 | 120.9 |
| C21—C22—I22 | 123.2 (7) | C921—C927—H927 | 120.9 |
| C24—C23—C22 | 120.2 (10) | C926—C921—C922 | 119.0 (15) |
| C24—C23—H23 | 119.9 | C926—C921—C927 | 120.5 (15) |
| C22—C23—H23 | 119.9 | C922—C921—C927 | 120.5 (15) |
| C23—C24—C25 | 121.8 (14) | C921—C922—C923 | 120.4 (15) |
| C23—C24—H24 | 119.1 | C921—C922—I22 | 122.5 (12) |
| C25—C24—H24 | 119.1 | C923—C922—I22 | 117.0 (12) |
| C26—C25—C24 | 118.1 (12) | C924—C923—C922 | 120.3 (19) |
| C26—C25—H25 | 121.0 | C924—C923—H923 | 119.8 |
| C24—C25—H25 | 121.0 | C922—C923—H923 | 119.8 |
| C25—C26—C21 | 121.1 (10) | C923—C924—C925 | 118 (2) |
| C25—C26—H26 | 119.4 | C923—C924—H924 | 120.9 |
| C21—C26—H26 | 119.4 | C925—C924—H924 | 120.9 |
| N2—C27—C21 | 121.8 (10) | C926—C925—C924 | 122 (2) |
| N2—C27—H27 | 119.1 | C926—C925—H925 | 118.9 |
| C21—C27—H27 | 119.1 | C924—C925—H925 | 118.9 |
| C27—N2—N1 | 114.2 (9) | C925—C926—C921 | 119.6 (18) |
| N2—N1—C11 | 126.9 (9) | C925—C926—H926 | 120.2 |
| N2—N1—N92 | 109.7 (8) | C921—C926—H926 | 120.2 |
| C11—N1—N92 | 123.5 (10) | O941—N914—O942 | 120 (2) |
| N2—N1—C911 | 117.6 (10) | O941—N914—C914 | 118 (2) |
| N92—N1—C911 | 132.1 (11) | O942—N914—C914 | 122 (2) |
| N2—N1—H1 | 116.6 | | |
| | | |
| N1—C11—C12—C13 | 176.9 (14) | N2—N1—C911—C912 | 0 (2) |
| C16—C11—C12—C13 | 0.5 (14) | C11—N1—C911—C912 | −149 (11) |
| N1—C11—C12—N12 | −7.7 (18) | N92—N1—C911—C912 | 170.4 (12) |
| C16—C11—C12—N12 | 175.8 (12) | N2—N1—C911—C916 | 177.3 (10) |
| N12—C12—C13—C14 | −176.9 (12) | C11—N1—C911—C916 | 29 (9) |
| C11—C12—C13—C14 | −1.1 (14) | N92—N1—C911—C916 | −12 (2) |
| C12—C13—C14—C15 | 0.6 (15) | C916—C911—C912—N912 | −151.0 (19) |
| C12—C13—C14—N14 | −179.7 (15) | N1—C911—C912—N912 | 26 (2) |
| C13—C14—N14—O42 | 173.0 (14) | C916—C911—C912—C913 | 0.0 |
| C15—C14—N14—O42 | −7 (2) | N1—C911—C912—C913 | 177 (3) |
| C15—C14—N14—O41 | 169.5 (15) | N912—C912—C913—C914 | 154.1 (16) |
| C13—C14—C15—C16 | 0.5 (16) | C911—C912—C913—C914 | 0.0 |
| N14—C14—C15—C16 | −179.3 (14) | C912—C913—C914—C915 | 0.0 |
| C14—C15—C16—C11 | −1.0 (17) | C912—C913—C914—N914 | −178 (3) |
| N1—C11—C16—C15 | −176.3 (12) | C913—C914—C915—C916 | 0.0 |
| C12—C11—C16—C15 | 0.6 (16) | N914—C914—C915—C916 | 178 (3) |
| C13—C12—N12—O122 | 2.9 (13) | C914—C915—C916—C911 | 0.0 |
| C11—C12—N12—O122 | −172.9 (10) | C912—C911—C916—C915 | 0.0 |
| C13—C12—N12—O121 | −178.1 (8) | N1—C911—C916—C915 | −177 (2) |
| C26—C21—C22—C23 | 3.0 (11) | C913—C912—N912—O922 | 12 (2) |
| C27—C21—C22—C23 | −176.7 (7) | C911—C912—N912—O922 | 165.5 (12) |
| C26—C21—C22—I22 | −173.7 (6) | C913—C912—N912—O921 | 178.5 (13) |
| C27—C21—C22—I22 | 6.6 (11) | C911—C912—N912—O921 | −28.3 (18) |
| C922—I22—C22—C23 | 149.4 (8) | N2—N1—N92—C927 | −6.2 (15) |
| C922—I22—C22—C21 | −33.8 (9) | C11—N1—N92—C927 | 174.3 (13) |
| C21—C22—C23—C24 | 1.4 (14) | C911—N1—N92—C927 | −177.1 (19) |
| I22—C22—C23—C24 | 178.3 (9) | N1—N92—C927—C921 | 177.7 (12) |
| C22—C23—C24—C25 | −5.2 (18) | N92—C927—C921—C926 | −6 (2) |
| C23—C24—C25—C26 | 4.3 (18) | N92—C927—C921—C922 | 174.1 (15) |
| C24—C25—C26—C21 | 0.3 (14) | C926—C921—C922—C923 | −4 (2) |
| C22—C21—C26—C25 | −3.8 (12) | C927—C921—C922—C923 | 175.4 (14) |
| C27—C21—C26—C25 | 175.9 (8) | C926—C921—C922—I22 | 171.6 (11) |
| C26—C21—C27—N2 | 2.1 (12) | C927—C921—C922—I22 | −9 (2) |
| N1—N2—C27—C21 | −179.6 (7) | C22—I22—C922—C921 | 34.0 (16) |
| C11—N1—N2—C27 | −171.0 (10) | C22—I22—C922—C923 | −149.9 (11) |
| N2—N1—C11—C12 | −178.0 (9) | C921—C922—C923—C924 | 4 (3) |
| N2—C27—C21—C22 | −178.2 (7) | I22—C922—C923—C924 | −172.5 (19) |
| C11—C12—N12—O121 | 6.1 (14) | C922—C923—C924—C925 | −3 (3) |
| C13—C14—N14—O41 | −10 (2) | C923—C924—C925—C926 | 3 (3) |
| C27—N2—N1—N92 | 9.5 (10) | C924—C925—C926—C921 | −4 (3) |
| C27—N2—N1—C911 | −178.1 (15) | C922—C921—C926—C925 | 5 (2) |
| C16—C11—N1—N2 | −1.6 (17) | C927—C921—C926—C925 | −175.2 (15) |
| C12—C11—N1—N92 | 1.4 (19) | C915—C914—N914—O941 | −6 (6) |
| C16—C11—N1—N92 | 177.7 (10) | C913—C914—N914—O941 | 172 (4) |
| C12—C11—N1—C911 | −143 (10) | C915—C914—N914—O942 | 170 (3) |
| C16—C11—N1—C911 | 33 (9) | C913—C914—N914—O942 | −12 (5) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O121 | 0.88 | 2.06 | 2.677 (9) | 126 |
| N1—H91···O921 | 0.88 | 2.07 | 2.716 (13) | 130 |
| C16—H16···O122i | 0.95 | 2.58 | 3.301 (18) | 133 |
| C23—H23···O41ii | 0.95 | 2.59 | 3.438 (18) | 149 |
| C913—H913···O942iii | 0.95 | 2.35 | 3.21 (3) | 150 |
| C916—H916···O922iv | 0.95 | 2.60 | 3.34 (3) | 136 |
| Symmetry codes: (i) x+1, y, z; (ii) x+1, y−1, z−1; (iii) −x+1, −y+1, −z+2; (iv) x−1, y, z. |
Experimental details
| | (II) | (III) |
| Crystal data |
| Chemical formula | C13H10IN3O2 | C13H9IN4O4 |
| Mr | 367.14 | 412.14 |
| Crystal system, space group | Orthorhombic, Pca21 | Triclinic, P1 |
| Temperature (K) | 120 | 120 |
| a, b, c (Å) | 24.0858 (11), 5.0699 (2), 10.5080 (4) | 7.8890 (5), 8.1405 (6), 12.176 (1) |
| α, β, γ (°) | 90, 90, 90 | 83.876 (4), 83.679 (4), 63.151 (3) |
| V (Å3) | 1283.16 (9) | 691.98 (9) |
| Z | 4 | 2 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 2.50 | 2.34 |
| Crystal size (mm) | 0.32 × 0.08 × 0.06 | 0.08 × 0.06 × 0.01 |
| |
| Data collection |
| Diffractometer | KappaCCD
diffractometer | KappaCCD
diffractometer |
| Absorption correction | Multi-scan
DENZO-SMN (Otwinowski & Minor, 1997) | Multi-scan
DENZO-SMN (Otwinowski & Minor, 1997) |
| Tmin, Tmax | 0.502, 0.865 | 0.835, 0.977 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 8490, 2800, 2082 | 7346, 2924, 2187 |
| Rint | 0.063 | 0.054 |
| (sin θ/λ)max (Å−1) | 0.648 | 0.648 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.036, 0.080, 1.02 | 0.047, 0.132, 1.07 |
| No. of reflections | 2800 | 2924 |
| No. of parameters | 160 | 243 |
| No. of restraints | 1 | 3 |
| H-atom treatment | H-atom parameters constrained | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 1.20, −0.47 | 0.84, −1.88 |
| Absolute structure | Flack (1983), 1259 Friedel pairs | ? |
| Absolute structure parameter | −0.08 (4) | ? |
Selected torsion angles (º) for (II) top| C11—N1—N2—C27 | 178.4 (5) | C22—C21—C27—N2 | −179.0 (5) |
| N1—N2—C27—C21 | 179.1 (5) | C13—C14—N4—O41 | −5.8 (8) |
| C12—C11—N1—N2 | 178.6 (5) | C13—C14—N4—O42 | 174.1 (5) |
Hydrogen-bond geometry (Å, º) for (II) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O41i | 0.88 | 2.04 | 2.905 (6) | 166 |
| Symmetry code: (i) −x+3/2, y+1, z+1/2. |
Selected torsion angles (º) for (III) top| N1—N2—C27—C21 | −179.6 (7) | N2—C27—C21—C22 | −178.2 (7) |
| C11—N1—N2—C27 | −171.0 (10) | C11—C12—N12—O121 | 6.1 (14) |
| N2—N1—C11—C12 | −178.0 (9) | C13—C14—N14—O41 | −10 (2) |
Hydrogen-bond geometry (Å, º) for (III) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1···O121 | 0.88 | 2.06 | 2.677 (9) | 126 |
| N1—H91···O921 | 0.88 | 2.07 | 2.716 (13) | 130 |
| C16—H16···O122i | 0.95 | 2.58 | 3.301 (18) | 133 |
| C23—H23···O41ii | 0.95 | 2.59 | 3.438 (18) | 149 |
| C913—H913···O942iii | 0.95 | 2.35 | 3.21 (3) | 150 |
| C916—H916···O922iv | 0.95 | 2.60 | 3.34 (3) | 136 |
| Symmetry codes: (i) x+1, y, z; (ii) x+1, y−1, z−1; (iii) −x+1, −y+1, −z+2; (iv) x−1, y, z. |
 O hydrogen bond [H
O hydrogen bond [H O = 2.04 Å, N
O = 2.04 Å, N O = 2.905 (6) Å and N-H
O = 2.905 (6) Å and N-H O = 166°] and a two-centre iodo-nitro interaction [I
O = 166°] and a two-centre iodo-nitro interaction [I O = 3.361 (4) Å] into a chain of edge-fused
O = 3.361 (4) Å] into a chain of edge-fused  O hydrogen bond generates centrosymmetric dimers.
O hydrogen bond generates centrosymmetric dimers.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2003/03/00/sk1609/sk1609fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2003/03/00/sk1609/sk1609fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2003/03/00/sk1609/sk1609fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/c/issues/2003/03/00/sk1609/sk1609fig4thm.gif)
![[Figure 5]](http://journals.iucr.org/c/issues/2003/03/00/sk1609/sk1609fig5thm.gif)




As part of a general study of the interplay of hydrogen bonds, iodo···nitro interactions and aromatic π···π stacking interactions in aromatic systems containing both iodo and nitro substituents, we have recently reported the molecular and supramolecular structures of a range of diaryl species (I) (see scheme) containing a variety of spacer units X, namely arenesulfonamides (Ia) and (Ib) (Kelly et al., 2002), Schiff-base imines (Ic) (Wardell et al., 2002) and (Id) (Glidewell, Howie et al., 2002), and benzylanilines (Ie) (Glidewell, Low et al., 2002). Here we report the structure of two analogous compounds containing the —CH═N—NH— linker unit, 2-iodobenzaldehyde 4-nitrophenylhydrazone (II) and 2-iodobenzaldehyde 2,4-dinitrophenylhydrazone (III), which prove to exhibit radically different patterns of supramolecular aggregation.
Molecules of (II) are essentially planar (Fig. 1), as demonstrated by the torsional angles (Table 1) that define the orientations of the rings relative to the central linked unit, of the nitro group relative to the adjacent ring. The only significant deviation from complete planarity is the 5.9 (3)° twist of the nitro group out of the plane of the aryl ring C11–C16.
The molecules are linked into molecular ladders by the combination of a single N—H···O hydrogen bond (Table 2) and a single two-centre iodo···nitro interaction. Atom I22 in the molecule at (x, y, z) forms an iodo···nitro interaction with atom O42 in the molecule at (x, 2 + y, 1 + z) [I···Oii 3.361 (4) Å, C—I···Oii 146.5 (2)°, I···Oii—Nii 146.9 (3)°; symmetry code ii = (x, 2 + y, 1 + z)], so generating by translation a C(12) chain (Starbuck et al., 1999) running parallel to the [021] direction. At the same time, the amino N1 at (x, y, z) acts as hydrogen-bond donor to O41 at (1.5 − x, 1 + y, 0.5 + z), while N1 at (1.5 − x, 1 + y, 0.5 + z) in turn acts as donor to O41 at (x, 2 + y, 1 + z), so producing a C(8) chain, parallel to [021], generated by the c-glide plane at x = 0.75. The combination of these two motifs generates a molecular ladder (Fig. 2) in which two C(12) iodo···nitro chains act as the uprights while the N—H···O hydrogen bonds act as the rungs. Alternatively, this sub-structure may be described as a chain of edge-fused R33(18) rings.
The [021] ladder lies in the domain 0.47 < x < 1.03. Related to this ladder by the action of the 21 screw axes is a second chain, running parallel to [0–21] and lying in the domain −0.03 < x < 0.53. There are no direction-specific interactions between adjacent chains. In particular, there are no C—H···O or C—H···π(arene) hydrogen bonds and no aromatic π···π stacking interactions.
Molecules of (III) exhibit orientational disorder (Figs. 3 and 4), with two orientations, having occupancies 0.681 (5) and 0.319 (5), respectively, related to one another approximately via a rotation about the line N1···I22. In both orientations, the molecules are nearly planar, as typified by the torsional angles for the major form (Table 3). In contrast to the supramolecular aggregation in (II), there are neither intermolecular N—H···O hydrogen bonds nor iodo···nitro interactions in (III). In each orientation there is an intramolecular N—H···O hydrogen bond (Table 4), but for most of the intermolecular C—H···O contacts the H···O distances are not significantly shorter than the sum of the van der Waals radii, and so these bonds are not structurally significant. The sole exception occurs for the minor orientation, where centrosymmetrically related molecules are linked into a dimer, via an R22(10) motif (Fig. 5). For the major orientation, however, the structure must be regarded as comprising isolated molecules
The occurrence of an iodo···nitro interaction in (II) may be compared with the absence of such an interaction not only in (III) but also in those analogues of types (Ic) and (Id) where the iodo substituent is in a 2-position (Wardell et al., 2002; Glidewell, Howie et al., 2002). Similarly, the absence of aromatic π···π stacking interactions from the structures of (II) and (III) may be compared with the case of 4-nitrobenzylidene-2'-iodoaniline, which is of the type (Id) but has the same disposition of substituents as (II). In 4-nitrobenzylidene-2'-iodoaniline, such interactions link hydrogen-bonded chains into sheets. Clearly, the qualitative prediction of which of the possible weak supramolecular interactions will be significant in compounds of these general types is far from straightforward.