Radial electron distributions and net atomic charges of magnetite Fe3O4 have been studied using single-crystal X-ray intensity data up to sin 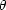 /
/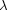 = 1.22 Å-1. The number of electrons, C(R), was calculated against the radius of an atomic sphere for each atom by the Fourier summation method from the observed crystal structure factors. The radial distribution function was then obtained by differentiating the C(R) curve to derive an electron distribution radius, rED, which can give a clear separation between neighboring atoms. The effective atomic charges of Fe ions thus obtained are +1.93 (4) e (A site) and +1.47 (4) e (B site), with rED = 1.13 and 1.19 Å, respectively. Fe ions occupying the A site are more ionic than those at the B site. The charge difference between sites A and B is +0.46 e, which is comparable to an ideal difference of +0.5 e between Fe3+ and [Fe2+Fe3+]. It suggests the existence of valence fluctuation between Fe2+ and Fe3+ in the B site. Crystal data: Fe3O4, Mr = 231.54, cubic, Fd3m, a = 8.375 (2) Å, V = 587.4 (4) Å3, Z = 8, Dx = 5.23 Mg m-3,
= 1.22 Å-1. The number of electrons, C(R), was calculated against the radius of an atomic sphere for each atom by the Fourier summation method from the observed crystal structure factors. The radial distribution function was then obtained by differentiating the C(R) curve to derive an electron distribution radius, rED, which can give a clear separation between neighboring atoms. The effective atomic charges of Fe ions thus obtained are +1.93 (4) e (A site) and +1.47 (4) e (B site), with rED = 1.13 and 1.19 Å, respectively. Fe ions occupying the A site are more ionic than those at the B site. The charge difference between sites A and B is +0.46 e, which is comparable to an ideal difference of +0.5 e between Fe3+ and [Fe2+Fe3+]. It suggests the existence of valence fluctuation between Fe2+ and Fe3+ in the B site. Crystal data: Fe3O4, Mr = 231.54, cubic, Fd3m, a = 8.375 (2) Å, V = 587.4 (4) Å3, Z = 8, Dx = 5.23 Mg m-3, 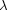 (Mo K
(Mo K ) = 0.7107 A,
) = 0.7107 A, 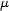 (Mo K
(Mo K ) = 14.88 mm-1, F(000) = 880, T = 296 K, final R = 0.016 wR = 0.010 for 78 reflections averaged out of 6049 within a hemisphere, with F > 3
) = 14.88 mm-1, F(000) = 880, T = 296 K, final R = 0.016 wR = 0.010 for 78 reflections averaged out of 6049 within a hemisphere, with F > 3 (F); u = 0.2555 (2).
(F); u = 0.2555 (2).

 journal menu
journal menu











