The [H
3PtW
6O
24]
5- anion has close to
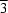 m
m symmetry, with Pt-O bond lengths in the range 1.97 (1)-2.01 (1) Å and W-O bond lengths in the ranges 1.72 (1)-1.78 (1) (O
t), 1.91 (1)-1.96 (1) (O
b) and 2.14 (1)-2.33 (1) Å (O
c), where O
t are terminal atoms, O
b bridge two W atoms, and O
c triply bridge one Pt and two W atoms. The protonated O atoms participate in strong interanion hydrogen bonds.
Supporting information
Key indicators
- Single-crystal X-ray study
- T = 298 K
- Mean
![[sigma]](//journals.iucr.org/logos/entities/sigma_rmgif.gif) (W-O) = 0.011 Å
(W-O) = 0.011 Å
- H-atom completeness 1%
- Disorder in solvent or counterion
- R factor = 0.053
- wR factor = 0.160
- Data-to-parameter ratio = 17.2
checkCIF results
No syntax errors found
ADDSYM reports no extra symmetry
 Alert Level C:
Alert Level C:
PLAT_302 Alert C Anion/Solvent Disorder ....................... 15.00 Perc.
General Notes
FORMU_01 There is a discrepancy between the atom counts in the
_chemical_formula_sum and the formula from the _atom_site* data.
Atom count from _chemical_formula_sum:H15 K5 O30 Pt1 W6
Atom count from the _atom_site data: K5 O30 Pt1 W6
CELLZ_01
From the CIF: _cell_formula_units_Z 4
From the CIF: _chemical_formula_sum H15 K5 O30 Pt W6
TEST: Compare cell contents of formula and atom_site data
atom Z*formula cif sites diff
H 60.00 0.00 60.00
K 20.00 20.00 0.00
O 120.00 120.00 0.00
Pt 4.00 4.00 0.00
W 24.00 24.00 0.00
Difference between formula and atom_site contents detected.
WARNING: H atoms missing from atom site list. Is this intentional?
0 Alert Level A = Potentially serious problem
0 Alert Level B = Potential problem
1 Alert Level C = Please check
The title crystals were isolated as described previously by reaction of K2WO4 and K2[Pt(OH)6] at pH 4.5 (Lee et al., 1983).
Data collection: STADI4 (Stoe & Cie, 1996); cell refinement: STADI4; data reduction: X-RED (Stoe & Cie, 1996); program(s) used to solve structure: SHELXS97 (Sheldrick, 1990); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: ORTEP-3 (Version 1.07; Farrugia, 1997) and DIAMOND (Brandenburg, 1998).
pentapotassium trihydrogenhexatungstoplatinate(IV) hexahydrate
top
Crystal data top
| K5[H3PtW6O24]·6H2O | F(000) = 3488 |
| Mr = 1988.81 | Dx = 4.337 Mg m−3 |
| Monoclinic, C2/m | Mo Kα radiation, λ = 0.71069 Å |
| Hall symbol: -C 2y | Cell parameters from 30 reflections |
| a = 11.449 (7) Å | θ = 13.5–16.3° |
| b = 19.829 (10) Å | µ = 27.92 mm−1 |
| c = 13.688 (14) Å | T = 298 K |
| β = 101.43 (7)° | Truncated octahedron, yellow |
| V = 3046 (4) Å3 | 0.28 × 0.16 × 0.16 mm |
| Z = 4 | |
Data collection top
Stoe Stadi-4
diffractometer | 3084 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.000 |
| Graphite monochromator | θmax = 27.5°, θmin = 1.5° |
| ω/2–θ scans | h = −14→14 |
Absorption correction: numerical
(X-SHAPE; Stoe & Cie, 1996) | k = 0→25 |
| Tmin = 0.013, Tmax = 0.128 | l = 0→17 |
| 3601 measured reflections | 3 standard reflections every 60 min |
| 3601 independent reflections | intensity decay: 3.8% |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | H-atom parameters not refined |
| R[F2 > 2σ(F2)] = 0.053 | w = 1/[σ2(Fo2) + (0.0929P)2 + 140.7849P]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.160 | (Δ/σ)max < 0.001 |
| S = 1.11 | Δρmax = 2.84 e Å−3 |
| 3601 reflections | Δρmin = −2.77 e Å−3 |
| 209 parameters | Extinction correction: SHELXL97, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| 0 restraints | Extinction coefficient: 0.00046 (4) |
| Primary atom site location: structure-invariant direct methods | |
Crystal data top
| K5[H3PtW6O24]·6H2O | V = 3046 (4) Å3 |
| Mr = 1988.81 | Z = 4 |
| Monoclinic, C2/m | Mo Kα radiation |
| a = 11.449 (7) Å | µ = 27.92 mm−1 |
| b = 19.829 (10) Å | T = 298 K |
| c = 13.688 (14) Å | 0.28 × 0.16 × 0.16 mm |
| β = 101.43 (7)° | |
Data collection top
Stoe Stadi-4
diffractometer | 3084 reflections with I > 2σ(I) |
Absorption correction: numerical
(X-SHAPE; Stoe & Cie, 1996) | Rint = 0.000 |
| Tmin = 0.013, Tmax = 0.128 | 3 standard reflections every 60 min |
| 3601 measured reflections | intensity decay: 3.8% |
| 3601 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.053 | 0 restraints |
| wR(F2) = 0.160 | H-atom parameters not refined |
| S = 1.11 | w = 1/[σ2(Fo2) + (0.0929P)2 + 140.7849P]
where P = (Fo2 + 2Fc2)/3 |
| 3601 reflections | Δρmax = 2.84 e Å−3 |
| 209 parameters | Δρmin = −2.77 e Å−3 |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Pt | 0.21219 (6) | 0.0000 | 0.15044 (6) | 0.0167 (2) | |
| W1 | 0.21388 (5) | 0.08566 (3) | −0.05557 (4) | 0.0194 (2) | |
| W2 | 0.23276 (5) | 0.16728 (3) | 0.15204 (4) | 0.0212 (2) | |
| W3 | 0.24516 (5) | 0.08082 (3) | 0.36674 (5) | 0.0223 (2) | |
| K1 | −0.0447 (8) | 0.2141 (5) | 0.2959 (6) | 0.112 (3) | |
| K2 | 0.5000 | 0.2319 (2) | 0.0000 | 0.0280 (10) | |
| K3 | 0.4361 (7) | 0.0000 | 0.6340 (6) | 0.0617 (19) | |
| K4 | 0.0000 | 0.0464 (10) | 0.5000 | 0.091 (7) | 0.50 |
| K5 | 0.4989 (14) | 0.0000 | 0.874 (3) | 0.118 (10) | 0.50 |
| Oc1 | 0.1175 (14) | 0.0000 | 0.0118 (11) | 0.024 (3) | |
| Oc2 | 0.3011 (8) | 0.0729 (5) | 0.0995 (8) | 0.020 (2) | |
| Oc3 | 0.1306 (8) | 0.0771 (5) | 0.2056 (8) | 0.019 (2) | |
| Oc4 | 0.3102 (12) | 0.0000 | 0.2894 (12) | 0.024 (3) | |
| Ob5 | 0.2905 (13) | 0.0000 | −0.0733 (12) | 0.026 (3) | |
| Ob6 | 0.1407 (9) | 0.1492 (5) | 0.0203 (8) | 0.025 (2) | |
| Ob7 | 0.3183 (8) | 0.1376 (5) | 0.2830 (8) | 0.020 (2) | |
| Ob8 | 0.1622 (13) | 0.0000 | 0.3950 (12) | 0.024 (3) | |
| Ot9 | 0.0989 (12) | 0.0857 (5) | −0.1637 (9) | 0.031 (3) | |
| Ot10 | 0.3279 (10) | 0.1327 (6) | −0.0857 (10) | 0.035 (3) | |
| Ot11 | 0.3445 (10) | 0.2184 (6) | 0.1264 (10) | 0.031 (3) | |
| Ot12 | 0.1292 (9) | 0.2212 (5) | 0.1884 (9) | 0.028 (2) | |
| Ot13 | 0.1376 (11) | 0.1365 (6) | 0.3958 (9) | 0.034 (3) | |
| Ot14 | 0.3626 (12) | 0.0809 (6) | 0.4677 (10) | 0.039 (3) | |
| Ow1 | 0.437 (2) | 0.132 (2) | 0.726 (3) | 0.21 (2) | |
| Ow2 | 0.0000 | 0.2871 (15) | 0.5000 | 0.120 (15) | |
| Ow3 | 0.185 (3) | 0.0000 | 0.667 (2) | 0.15 (2) | |
| Ow4 | 0.269 (4) | 0.308 (4) | 0.406 (4) | 0.40 (6) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Pt | 0.0184 (3) | 0.0159 (4) | 0.0157 (4) | 0.000 | 0.0032 (3) | 0.000 |
| W1 | 0.0190 (3) | 0.0203 (3) | 0.0193 (3) | −0.00073 (18) | 0.0045 (2) | 0.0024 (2) |
| W2 | 0.0234 (3) | 0.0171 (3) | 0.0241 (3) | −0.00171 (19) | 0.0076 (2) | −0.0004 (2) |
| W3 | 0.0256 (3) | 0.0228 (3) | 0.0182 (3) | −0.0007 (2) | 0.0038 (2) | −0.0027 (2) |
| K1 | 0.110 (5) | 0.150 (8) | 0.087 (5) | 0.076 (6) | 0.045 (4) | 0.056 (5) |
| K2 | 0.026 (2) | 0.024 (2) | 0.035 (3) | 0.000 | 0.0085 (18) | 0.000 |
| K3 | 0.059 (4) | 0.065 (5) | 0.053 (4) | 0.000 | −0.008 (3) | 0.000 |
| K4 | 0.114 (14) | 0.072 (11) | 0.120 (17) | 0.000 | 0.100 (14) | 0.000 |
| K5 | 0.034 (8) | 0.091 (15) | 0.24 (3) | 0.000 | 0.044 (12) | 0.000 |
| Oc1 | 0.033 (8) | 0.023 (7) | 0.011 (7) | 0.000 | −0.004 (6) | 0.000 |
| Oc2 | 0.017 (4) | 0.016 (4) | 0.027 (6) | 0.000 (4) | 0.006 (4) | 0.001 (4) |
| Oc3 | 0.016 (4) | 0.019 (5) | 0.022 (5) | 0.000 (3) | 0.005 (4) | −0.007 (4) |
| Oc4 | 0.022 (7) | 0.029 (8) | 0.024 (8) | 0.000 | 0.013 (6) | 0.000 |
| Ob5 | 0.026 (7) | 0.023 (7) | 0.031 (9) | 0.000 | 0.006 (6) | 0.000 |
| Ob6 | 0.029 (5) | 0.025 (5) | 0.020 (5) | 0.010 (4) | 0.001 (4) | 0.004 (4) |
| Ob7 | 0.016 (4) | 0.024 (5) | 0.020 (5) | −0.003 (4) | −0.002 (4) | −0.007 (4) |
| Ob8 | 0.026 (7) | 0.021 (7) | 0.028 (8) | 0.000 | 0.012 (6) | 0.000 |
| Ot9 | 0.048 (7) | 0.022 (5) | 0.020 (6) | −0.001 (5) | 0.001 (5) | 0.003 (4) |
| Ot10 | 0.035 (6) | 0.026 (6) | 0.046 (7) | −0.009 (5) | 0.015 (5) | −0.007 (5) |
| Ot11 | 0.031 (5) | 0.024 (5) | 0.040 (7) | −0.011 (4) | 0.011 (5) | −0.006 (5) |
| Ot12 | 0.032 (5) | 0.023 (5) | 0.033 (6) | 0.012 (4) | 0.016 (5) | 0.002 (5) |
| Ot13 | 0.052 (7) | 0.032 (6) | 0.025 (6) | 0.014 (5) | 0.024 (5) | 0.002 (5) |
| Ot14 | 0.045 (7) | 0.041 (7) | 0.023 (6) | −0.001 (5) | −0.011 (5) | −0.001 (5) |
| Ow1 | 0.057 (13) | 0.31 (5) | 0.25 (4) | −0.04 (2) | 0.014 (18) | −0.21 (4) |
| Ow2 | 0.12 (3) | 0.057 (18) | 0.16 (4) | 0.000 | −0.04 (3) | 0.000 |
| Ow3 | 0.08 (2) | 0.35 (7) | 0.040 (16) | 0.000 | 0.021 (15) | 0.000 |
| Ow4 | 0.19 (4) | 0.63 (12) | 0.29 (6) | 0.26 (6) | −0.13 (4) | −0.31 (8) |
Geometric parameters (Å, º) top
| W1—W1i | 3.397 (2) | K1—Ob7iii | 3.321 (13) |
| W1—W2 | 3.240 (3) | K1—Ow1iv | 3.33 (4) |
| W2—W3 | 3.381 (3) | K2—Ot11 | 2.731 (12) |
| W3—W3i | 3.205 (2) | K2—Ot11v | 2.731 (12) |
| Pt—Oc2 | 1.973 (10) | K2—Ob6vi | 2.837 (11) |
| Pt—Oc1 | 1.990 (14) | K2—Ob6vii | 2.837 (11) |
| Pt—Oc4 | 2.007 (16) | K2—Ot12vi | 2.862 (12) |
| Pt—Oc3 | 2.014 (10) | K2—Ot12vii | 2.862 (12) |
| W1—Ot10 | 1.720 (11) | K2—Ot10 | 2.866 (12) |
| W1—Ot9 | 1.775 (12) | K2—Ot10v | 2.866 (12) |
| W1—Ob6 | 1.927 (11) | K3—Ot14i | 2.775 (14) |
| W1—Ob5 | 1.949 (8) | K3—Ot14 | 2.775 (14) |
| W1—Oc2 | 2.174 (11) | K3—Oc4viii | 2.886 (16) |
| W1—Oc1 | 2.315 (11) | K3—Ow1i | 2.90 (3) |
| W2—Ot11 | 1.721 (11) | K3—Ow1 | 2.90 (3) |
| W2—Ot12 | 1.740 (10) | K3—Ow3 | 3.00 (3) |
| W2—Ob6 | 1.934 (11) | K3—Ot14ix | 3.330 (16) |
| W2—Ob7 | 1.956 (10) | K3—Ot14viii | 3.330 (16) |
| W2—Oc2 | 2.203 (10) | K4—Ob8x | 2.724 (15) |
| W2—Oc3 | 2.333 (10) | K4—Ob8 | 2.724 (15) |
| W3—Ot14 | 1.727 (12) | K4—Ot13xi | 2.933 (17) |
| W3—Ot13 | 1.758 (11) | K4—Ot13 | 2.933 (17) |
| W3—Ob7 | 1.913 (11) | K4—Ow3x | 2.93 (3) |
| W3—Ob8 | 1.940 (8) | K4—Ow3 | 2.93 (3) |
| W3—Oc4 | 2.135 (10) | K5—Ob5xii | 2.63 (2) |
| W3—Oc3 | 2.334 (11) | K5—Oc2ix | 2.671 (16) |
| Ob6—Ob6ii | 3.16 (2) | K5—Oc2viii | 2.671 (16) |
| K1—Ot12 | 2.705 (14) | K5—Ob5viii | 3.26 (3) |
| K1—Ot13 | 2.732 (14) | K5—Ow1i | 3.30 (6) |
| K1—Ot11iii | 2.760 (14) | K5—Ow1 | 3.30 (6) |
| K1—Ow4iii | 2.88 (5) | K5—Ot10xiii | 3.389 (17) |
| K1—Ow2 | 3.097 (17) | K5—Ot10xii | 3.389 (16) |
| K1—Ot9ii | 3.115 (16) | K5—Oc4viii | 3.42 (3) |
| | | |
| W1—W2—W3 | 119.54 (6) | Ot14—W3—Ob8 | 100.1 (6) |
| W3i—W3—W2 | 120.47 (3) | Ot13—W3—Ob8 | 94.7 (5) |
| W2—W1—W1i | 119.97 (3) | Ob7—W3—Ob8 | 153.5 (5) |
| Oc2i—Pt—Oc2 | 94.2 (6) | Pt—Oc1—W1 | 99.6 (5) |
| Oc2i—Pt—Oc1 | 83.4 (5) | Pt—Oc1—W1i | 99.6 (5) |
| Oc2i—Pt—Oc4 | 96.0 (4) | W1—Oc1—W1i | 94.4 (6) |
| Oc1—Pt—Oc4 | 179.1 (6) | Pt—Oc2—W1 | 105.1 (4) |
| Oc2i—Pt—Oc3i | 83.5 (4) | Pt—Oc2—W2 | 105.4 (5) |
| Ot10—W1—Ob6 | 101.9 (5) | W1—Oc2—W2 | 95.5 (4) |
| Ot9—W1—Ob6 | 96.8 (5) | Pt—Oc3—W2 | 99.5 (4) |
| Ob6—W1—Ob5 | 153.1 (6) | Pt—Oc3—W3 | 99.4 (4) |
| Ot10—W1—Oc2 | 94.2 (5) | W2—Oc3—W3 | 92.9 (3) |
| Ot9—W1—Oc2 | 159.3 (5) | Pt—Oc4—W3 | 106.6 (5) |
| Ob6—W1—Oc2 | 73.4 (4) | Pt—Oc4—W3i | 106.6 (5) |
| Ot11—W2—Ot12 | 105.8 (6) | W3—Oc4—W3i | 97.3 (6) |
| Ot11—W2—Ob6 | 102.1 (5) | W1i—Ob5—W1 | 121.3 (8) |
| Ot12—W2—Ob6 | 96.0 (5) | W1—Ob6—W2 | 114.1 (5) |
| Ot11—W2—Ob7 | 96.1 (5) | W1—Ob6—Ob6ii | 116.3 (5) |
| Ot12—W2—Ob7 | 99.7 (5) | W2—Ob6—Ob6ii | 120.8 (6) |
| Ob6—W2—Ob7 | 151.7 (4) | K2vii—Ob6—Ob6ii | 56.2 (2) |
| Ot14—W3—Ot13 | 106.8 (6) | W3—Ob7—W2 | 121.8 (5) |
| Ot14—W3—Ob7 | 96.6 (6) | W3—Ob8—W3i | 111.4 (7) |
| Ot13—W3—Ob7 | 100.1 (5) | | |
| Symmetry codes: (i) x, −y, z; (ii) −x, y, −z; (iii) x−1/2, −y+1/2, z; (iv) −x+1/2, −y+1/2, −z+1; (v) −x+1, y, −z; (vi) x+1/2, −y+1/2, z; (vii) −x+1/2, −y+1/2, −z; (viii) −x+1, −y, −z+1; (ix) −x+1, y, −z+1; (x) −x, −y, −z+1; (xi) −x, y, −z+1; (xii) x, y, z+1; (xiii) x, −y, z+1. |
Hydrogen-bond geometry (Å) top
| D—H···A | D···A |
| Oc1···Oc1ii | 2.64 (3) |
| Oc3···Ot9ii | 2.582 (16) |
| Ow1···Ob7ix | 2.83 (3) |
| Ow1···Ow4iv | 2.93 (6) |
| Ow1···Ot10xii | 3.08 (4) |
| Ow2···Ot14iv | 3.04 (3) |
| Ow3···Ot9xii | 3.18 (3) |
| Ow4···Ot13iv | 2.92 (4) |
| Symmetry codes: (ii) −x, y, −z; (iv) −x+1/2, −y+1/2, −z+1; (ix) −x+1, y, −z+1; (xii) x, y, z+1. |
Experimental details
| Crystal data |
| Chemical formula | K5[H3PtW6O24]·6H2O |
| Mr | 1988.81 |
| Crystal system, space group | Monoclinic, C2/m |
| Temperature (K) | 298 |
| a, b, c (Å) | 11.449 (7), 19.829 (10), 13.688 (14) |
| β (°) | 101.43 (7) |
| V (Å3) | 3046 (4) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 27.92 |
| Crystal size (mm) | 0.28 × 0.16 × 0.16 |
| |
| Data collection |
| Diffractometer | Stoe Stadi-4
diffractometer |
| Absorption correction | Numerical
(X-SHAPE; Stoe & Cie, 1996) |
| Tmin, Tmax | 0.013, 0.128 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 3601, 3601, 3084 |
| Rint | 0.000 |
| (sin θ/λ)max (Å−1) | 0.650 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.053, 0.160, 1.11 |
| No. of reflections | 3601 |
| No. of parameters | 209 |
| H-atom treatment | H-atom parameters not refined |
| w = 1/[σ2(Fo2) + (0.0929P)2 + 140.7849P]
where P = (Fo2 + 2Fc2)/3 |
| Δρmax, Δρmin (e Å−3) | 2.84, −2.77 |
Selected geometric parameters (Å, º) top| W1—W1i | 3.397 (2) | W2—Ot11 | 1.721 (11) |
| W1—W2 | 3.240 (3) | W2—Ot12 | 1.740 (10) |
| W2—W3 | 3.381 (3) | W2—Ob6 | 1.934 (11) |
| W3—W3i | 3.205 (2) | W2—Ob7 | 1.956 (10) |
| Pt—Oc2 | 1.973 (10) | W2—Oc2 | 2.203 (10) |
| Pt—Oc1 | 1.990 (14) | W2—Oc3 | 2.333 (10) |
| Pt—Oc4 | 2.007 (16) | W3—Ot14 | 1.727 (12) |
| Pt—Oc3 | 2.014 (10) | W3—Ot13 | 1.758 (11) |
| W1—Ot10 | 1.720 (11) | W3—Ob7 | 1.913 (11) |
| W1—Ot9 | 1.775 (12) | W3—Ob8 | 1.940 (8) |
| W1—Ob6 | 1.927 (11) | W3—Oc4 | 2.135 (10) |
| W1—Ob5 | 1.949 (8) | W3—Oc3 | 2.334 (11) |
| W1—Oc2 | 2.174 (11) | Ob6—Ob6ii | 3.16 (2) |
| W1—Oc1 | 2.315 (11) | | |
| | | |
| W1—W2—W3 | 119.54 (6) | W1—Ob6—W2 | 114.1 (5) |
| W3i—W3—W2 | 120.47 (3) | W3—Ob7—W2 | 121.8 (5) |
| W2—W1—W1i | 119.97 (3) | W3—Ob8—W3i | 111.4 (7) |
| W1i—Ob5—W1 | 121.3 (8) | | |
| Symmetry codes: (i) x, −y, z; (ii) −x, y, −z. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D···A |
| Oc1···Oc1ii | 2.64 (3) |
| Oc3···Ot9ii | 2.582 (16) |
| Ow1···Ob7iii | 2.83 (3) |
| Ow1···Ow4iv | 2.93 (6) |
| Ow1···Ot10v | 3.08 (4) |
| Ow2···Ot14iv | 3.04 (3) |
| Ow3···Ot9v | 3.18 (3) |
| Ow4···Ot13iv | 2.92 (4) |
| Symmetry codes: (ii) −x, y, −z; (iii) −x+1, y, −z+1; (iv) −x+1/2, −y+1/2, −z+1; (v) x, y, z+1. |

 journal menu
journal menu











![[Figure 1]](http://journals.iucr.org/e/issues/2002/12/00/mg6017/mg6017fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/e/issues/2002/12/00/mg6017/mg6017fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/e/issues/2002/12/00/mg6017/mg6017fig3thm.gif)




While investigating Anderson-type heteropolyoxometallates containing PtIV (Anderson, 1937; Tsigdinos, 1978), we found that gradual protonation and α,β-geometric isomerism occur in the hexamolybdoplatinate(IV) anion, [PtMo6O24]8− (Lee & Sasaki, 1984; Joo et al., 1994; Lee, 1994; Lee & Sasaki, 1994; Lee & Joo, 2000). Only three examples of the corresponding hexatungstoplatinate(IV) anions have been reported: K6Na2[PtW6O24].12H2O (Lee et al., 1984), Na5[H3PtW6O24].20H2O (Lee et al., 1983), and K2.5[H5.5PtW6O24]·2H2O (Lee & Sasaki, 1987). The location of the H atoms in Na5[H3PtW6O24].20H2O could not be determined.
The potassium salt of triprotonated hexatungstoplatinate (IV), [H3PtW6O24]5−, has now been obtained. The [H3PtW6O24]5− anions are packed in a different arrangement compared with the sodium salt. These compounds were prepared under conditions varying over a pH range as wide as 4.5–6.7. Fig. 1 shows the [H3PtW6O24]5− anion, with the O atoms classified in the same way as previously (Lee et al., 1984). It forms a dimer, [H6(PtW6O24)2]10−, by strong hydrogen bonding (Fig. 2). In contrast, hydrogen bonding between adjacent anions does not occur in the sodium salt. Therefore, the potassium salt (4.337 Mg m−3) is more densely packed than the sodium salt (3.362 Mg m−3). The protonated O atoms in the anion can be identified by examining the W—O distances and the W—Ob—W angles. Protonation increases the W—O distances and decreases the Ob—W—Ob angles by about 10°. The elongation of W—Oc bond distance is especially notable. From this result, it can be concluded that the Oc3 and Oc1 atoms are protonated. Because Oc1 is located in a special position (4i), the number of H+ is also 0.5. Thus, the number of 2.5 H+ is explained. The remaining protonated O atoms could not be identified with certainty. Examination of the W1—Ob6—W2 [114.1 (5)°] and W3—Ob8—W3' [111.4 (7)°] angles suggests that either the Ob6 or Ob8 atom may be protonated. Since the Ob6 atom is in a general position (8j), it can only be protonated 50% of the time. If interanion Oc3–Ot9 and Oc1–Oc1 hydrogen bonds are formed, the Ob6—Ob6 interanion distance that results is too long to be considered hydrogen bonding. Because Ob8 is in a special position (4i), the number of H+ is also 0.5 and it is likely that Ob8 is protonated.
The K+ ions are coordinated at distances of 2.7 to 3.4 Å by six or eight O atoms from anions or water molecules. All water molecules are coordinated to K+ ions and form hydrogen bonds with neighbouring water molecules or with the O atoms of anions. Fig. 3 shows the unit-cell packing.