The crystal structure and the results of theoretical calculations for the new organoarsenate salt
o-anisidinium dihydroarsenate (systematic name: 2-methoxyanilinium dihydrogen arsenate), C
7H
10NO
+·H
2AsO
4−, are reported. The salt, crystallizing in the triclinic space group
P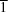
, was synthesized using a solution method and was characterized by single-crystal X-ray diffraction analysis. It possesses a layered supramolecular architecture in the crystal. The intermolecular interactions were studied using Hirshfeld surface analysis which confirmed that hydrogen bonds and H

H contacts play dominant roles in the crystal structure of the investigated system. An analysis of the electronic structure and molecular modelling using charge distribution confirms the good electrophilic reactivity of the title compound.
Supporting information
CCDC reference: 1886678
Data collection: CrysAlis PRO (Rigaku OD, 2015); cell refinement: CrysAlis PRO (Rigaku OD, 2015); data reduction: CrysAlis PRO (Rigaku OD, 2015); program(s) used to solve structure: SHELXT (Sheldrick, 2015a); program(s) used to refine structure: SHELXL2018 (Sheldrick, 2015b); molecular graphics: OLEX2 (Dolomanov et al., 2009),
DIAMOND (Brandenburg & Putz, 2005) and
Mercury (Macrae et al., 2008); software used to prepare material for publication: PLATON (Spek, 2009).
2-Methoxyanilinium dihydrogen arsenate
top
Crystal data top
| C7H10NO+·H2AsO4− | Z = 2 |
| Mr = 265.10 | F(000) = 268 |
| Triclinic, P1 | Dx = 1.767 Mg m−3 |
| a = 5.5235 (2) Å | Cu Kα radiation, λ = 1.54184 Å |
| b = 9.4385 (4) Å | Cell parameters from 5519 reflections |
| c = 10.4633 (5) Å | θ = 4.9–71.6° |
| α = 70.769 (4)° | µ = 4.67 mm−1 |
| β = 75.984 (4)° | T = 100 K |
| γ = 89.624 (3)° | Plate, light pink |
| V = 498.12 (4) Å3 | 0.54 × 0.21 × 0.12 mm |
Data collection top
Rigaku OD SuperNova Dual source
diffractometer with an Atlas detector | 1778 independent reflections |
| Radiation source: micro-focus sealed X-ray tube | 1769 reflections with I > 2σ(I) |
| Detector resolution: 5.2195 pixels mm-1 | Rint = 0.023 |
| ω scans | θmax = 67.0°, θmin = 5.0° |
Absorption correction: analytical
[CrysAlis PRO (Rigaku OD, 2015), based on expressions derived by Clark &
Reid
(1995)] | h = −5→6 |
| Tmin = 0.118, Tmax = 0.444 | k = −11→11 |
| 6075 measured reflections | l = −12→12 |
Refinement top
| Refinement on F2 | 23 restraints |
| Least-squares matrix: full | Hydrogen site location: mixed |
| R[F2 > 2σ(F2)] = 0.035 | H atoms treated by a mixture of independent and constrained refinement |
| wR(F2) = 0.083 | w = 1/[σ2(Fo2) + (0.0298P)2 + 1.5814P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.09 | (Δ/σ)max = 0.001 |
| 1778 reflections | Δρmax = 1.18 e Å−3 |
| 153 parameters | Δρmin = −0.62 e Å−3 |
Special details top
Geometry. All esds (except the esd in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell esds are taken
into account individually in the estimation of esds in distances, angles
and torsion angles; correlations between esds in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell esds is used for estimating esds involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| C1 | 0.3599 (7) | 0.5900 (4) | 0.2151 (4) | 0.0272 (7) | |
| C2 | 0.2284 (9) | 0.5662 (5) | 0.1281 (5) | 0.0469 (10) | |
| H2 | 0.104266 | 0.487460 | 0.161277 | 0.056* | |
| C3 | 0.2821 (9) | 0.6611 (6) | −0.0107 (5) | 0.0535 (11) | |
| H3 | 0.192238 | 0.646967 | −0.070721 | 0.064* | |
| C4 | 0.4672 (7) | 0.7749 (5) | −0.0584 (4) | 0.0380 (9) | |
| H4 | 0.502557 | 0.837980 | −0.151204 | 0.046* | |
| C5 | 0.6034 (6) | 0.7983 (4) | 0.0289 (4) | 0.0260 (7) | |
| H5 | 0.729597 | 0.876006 | −0.005196 | 0.031* | |
| C6 | 0.5503 (6) | 0.7050 (4) | 0.1676 (3) | 0.0213 (7) | |
| C7 | 0.8484 (8) | 0.8389 (6) | 0.2247 (5) | 0.0449 (10) | |
| H7A | 0.978574 | 0.836834 | 0.145870 | 0.067* | |
| H7B | 0.919249 | 0.832393 | 0.301500 | 0.067* | |
| H7C | 0.768607 | 0.931172 | 0.199874 | 0.067* | |
| N1 | 0.3018 (7) | 0.4979 (3) | 0.3626 (4) | 0.0403 (9) | |
| H1A | 0.419 (6) | 0.438 (4) | 0.379 (5) | 0.048* | |
| H1B | 0.164 (5) | 0.442 (5) | 0.384 (5) | 0.048* | |
| H1C | 0.282 (9) | 0.555 (4) | 0.414 (4) | 0.048* | |
| O1 | 0.6675 (5) | 0.7141 (3) | 0.2650 (2) | 0.0328 (6) | |
| As1 | 0.24779 (6) | 0.81556 (4) | 0.54369 (3) | 0.02194 (15) | |
| O2A | 0.211 (3) | 0.6330 (13) | 0.5512 (10) | 0.021 (3) | 0.53 (4) |
| H4A | −0.035 (7) | 0.913 (3) | 0.652 (4) | 0.032* | |
| H5A | 0.577 (8) | 0.873 (3) | 0.591 (4) | 0.032* | |
| O2B | 0.117 (3) | 0.6741 (13) | 0.5214 (11) | 0.016 (3) | 0.47 (4) |
| O3 | 0.2758 (4) | 0.9644 (3) | 0.4007 (2) | 0.0311 (6) | |
| O4 | 0.0495 (4) | 0.8451 (3) | 0.6847 (2) | 0.0267 (5) | |
| O5 | 0.5254 (5) | 0.7928 (3) | 0.5888 (3) | 0.0303 (6) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| C1 | 0.0287 (18) | 0.0207 (16) | 0.0264 (17) | −0.0031 (13) | 0.0074 (14) | −0.0106 (14) |
| C2 | 0.0470 (18) | 0.0529 (18) | 0.0444 (17) | −0.0151 (14) | −0.0005 (14) | −0.0288 (14) |
| C3 | 0.0517 (19) | 0.071 (2) | 0.0442 (17) | −0.0086 (15) | −0.0116 (14) | −0.0283 (15) |
| C4 | 0.0346 (16) | 0.0489 (17) | 0.0282 (15) | 0.0037 (13) | −0.0062 (12) | −0.0113 (13) |
| C5 | 0.0242 (17) | 0.0240 (16) | 0.0238 (17) | −0.0004 (13) | −0.0007 (13) | −0.0039 (14) |
| C6 | 0.0194 (15) | 0.0213 (16) | 0.0223 (16) | 0.0034 (12) | −0.0032 (13) | −0.0077 (13) |
| C7 | 0.028 (2) | 0.071 (3) | 0.041 (2) | −0.0123 (19) | −0.0043 (17) | −0.028 (2) |
| N1 | 0.049 (2) | 0.0179 (15) | 0.0332 (18) | 0.0066 (14) | 0.0189 (16) | −0.0030 (13) |
| O1 | 0.0234 (12) | 0.0477 (16) | 0.0243 (12) | −0.0029 (11) | −0.0071 (10) | −0.0074 (11) |
| As1 | 0.0240 (2) | 0.0226 (2) | 0.0183 (2) | −0.00798 (14) | 0.00011 (14) | −0.00942 (15) |
| O2A | 0.021 (5) | 0.022 (3) | 0.020 (3) | −0.002 (4) | −0.003 (3) | −0.007 (3) |
| O2B | 0.014 (5) | 0.015 (4) | 0.020 (3) | 0.000 (3) | −0.005 (3) | −0.007 (3) |
| O3 | 0.0211 (12) | 0.0489 (15) | 0.0163 (11) | 0.0016 (11) | −0.0047 (9) | −0.0020 (11) |
| O4 | 0.0239 (12) | 0.0377 (14) | 0.0166 (11) | 0.0090 (10) | −0.0050 (9) | −0.0070 (10) |
| O5 | 0.0267 (13) | 0.0264 (12) | 0.0317 (13) | 0.0129 (10) | −0.0067 (10) | −0.0027 (11) |
Geometric parameters (Å, º) top
| C1—C2 | 1.365 (6) | C7—H7A | 0.9600 |
| C1—C6 | 1.392 (5) | C7—H7B | 0.9600 |
| C1—N1 | 1.456 (5) | C7—H7C | 0.9600 |
| C2—C3 | 1.392 (7) | N1—H1A | 0.864 (10) |
| C2—H2 | 0.9300 | N1—H1B | 0.869 (10) |
| C3—C4 | 1.365 (6) | N1—H1C | 0.871 (10) |
| C3—H3 | 0.9300 | As1—O2B | 1.631 (6) |
| C4—C5 | 1.384 (5) | As1—O3 | 1.656 (2) |
| C4—H4 | 0.9300 | As1—O5 | 1.703 (2) |
| C5—C6 | 1.387 (5) | As1—O2A | 1.710 (8) |
| C5—H5 | 0.9300 | As1—O4 | 1.714 (2) |
| C6—O1 | 1.357 (4) | O4—H4A | 0.819 (10) |
| C7—O1 | 1.431 (5) | O5—H5A | 0.817 (10) |
| | | |
| C2—C1—C6 | 121.6 (3) | O1—C7—H7C | 109.5 |
| C2—C1—N1 | 121.1 (4) | H7A—C7—H7C | 109.5 |
| C6—C1—N1 | 117.3 (3) | H7B—C7—H7C | 109.5 |
| C1—C2—C3 | 119.3 (4) | C1—N1—H1A | 110 (3) |
| C1—C2—H2 | 120.3 | C1—N1—H1B | 109 (3) |
| C3—C2—H2 | 120.3 | H1A—N1—H1B | 107 (5) |
| C4—C3—C2 | 119.7 (4) | C1—N1—H1C | 110 (3) |
| C4—C3—H3 | 120.2 | H1A—N1—H1C | 111 (5) |
| C2—C3—H3 | 120.2 | H1B—N1—H1C | 109 (5) |
| C3—C4—C5 | 121.3 (4) | C6—O1—C7 | 117.2 (3) |
| C3—C4—H4 | 119.4 | O2B—As1—O3 | 108.2 (6) |
| C5—C4—H4 | 119.4 | O2B—As1—O5 | 118.0 (8) |
| C4—C5—C6 | 119.5 (3) | O3—As1—O5 | 110.12 (12) |
| C4—C5—H5 | 120.2 | O3—As1—O2A | 124.7 (4) |
| C6—C5—H5 | 120.2 | O5—As1—O2A | 93.7 (7) |
| O1—C6—C5 | 126.1 (3) | O2B—As1—O4 | 106.3 (3) |
| O1—C6—C1 | 115.2 (3) | O3—As1—O4 | 110.58 (12) |
| C5—C6—C1 | 118.7 (3) | O5—As1—O4 | 103.41 (12) |
| O1—C7—H7A | 109.5 | O2A—As1—O4 | 111.1 (2) |
| O1—C7—H7B | 109.5 | As1—O4—H4A | 106 (3) |
| H7A—C7—H7B | 109.5 | As1—O5—H5A | 109 (3) |
| | | |
| C6—C1—C2—C3 | −1.4 (6) | C2—C1—C6—O1 | −179.1 (3) |
| N1—C1—C2—C3 | 177.1 (4) | N1—C1—C6—O1 | 2.4 (4) |
| C1—C2—C3—C4 | 0.9 (7) | C2—C1—C6—C5 | 1.0 (5) |
| C2—C3—C4—C5 | −0.1 (7) | N1—C1—C6—C5 | −177.5 (3) |
| C3—C4—C5—C6 | −0.3 (6) | C5—C6—O1—C7 | 5.6 (5) |
| C4—C5—C6—O1 | 179.9 (3) | C1—C6—O1—C7 | −174.3 (3) |
| C4—C5—C6—C1 | −0.1 (5) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| N1—H1A···O2Ai | 0.87 (4) | 2.35 (4) | 3.159 (17) | 154 (4) |
| N1—H1A···O5i | 0.87 (4) | 2.12 (4) | 2.825 (4) | 137 (3) |
| N1—H1B···O2Aii | 0.87 (4) | 2.07 (4) | 2.905 (16) | 162 (4) |
| N1—H1C···O2A | 0.87 (4) | 1.78 (4) | 2.634 (6) | 167 (5) |
| O4—H4A···O3iii | 0.82 (4) | 1.82 (4) | 2.634 (3) | 168 (4) |
| O5—H5A···O3iviv | 0.82 (3) | 1.78 (3) | 2.594 (4) | 173 (4) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) −x, −y+1, −z+1; (iii) −x, −y+2, −z+1; (iv) −x+1, −y+2, −z+1. |
The geometry of the C—H···π contact in the crystal of 1 top| D—H | CgI | d(D···CgI) (Å) | D—X···CgI (°) |
| C7—H7A | 1v | 3.664 (5) | 141 1 |
| Symmetry code: (v) x+1, y, z.
Cg1 denotes the geometric centre of gravity of the aromatic rings
delineated by the C1A–C6A and C1–C6 atoms (see Fig. 1). |
Percentage contribution of intermolecular interactions to the Hirshfeld
surface of the cation and the [H2AsO4]- anionic species containing the
major (Anionmajor) and minor (Anionminor) components of disordered O atom top| Cation | H···H | O···H | C···H | C···C | O···O | C···O | As···O |
| With Anionmajor | 42.2 | 32.5 | 20.2 | 2.5 | 2 | 0.4 | 0.2 |
| With Anionminor | 41.8 | 33.7 | 20.2 | 2.5 | 1.2 | 0.4 | 0.2 |
| | | | | | | |
| Anion | O···H | H···H | O···O | As···H | As···O | | |
| Anionmajor | 77.6 | 18.4 | 3.3 | 0.5 | 0.2 | | |
| Anionminor | 78.1 | 18.5 | 3.1 | 0 | 0.3 | | |
Mulliken, PABOON, Loewdin and Natural charges for 1 top| Atom | MULLIKEN charges | PABOON charges | LOEWDIN charges | Natural charges |
| As1 | 1.13455 | 1.4565 | 0.57836 | 2.47100 |
| H2 | 0.34114 | 0.1765 | 0.36002 | 0.48528 |
| H3 | 0.33803 | 0.1770 | 0.35825 | 0.48299 |
| O4 | -0.59195 | -0.5229 | -0.46736 | -1.00727 |
| O5 | -0.57098 | -0.5239 | -0.44135 | -1.02149 |
| O6 | -0.56659 | -0.5026 | -0.44495 | -0.98668 |
| O7 | -0.52259 | -0.6445 | -0.39272 | -1.01438 |
| C8 | 0.10726 | 0.0852 | -0.00934 | 0.13660 |
| C9 | 0.25465 | 0.1838 | 0.01906 | 0.30287 |
| C10 | -0.22389 | -0.0380 | -0.22698 | -0.29069 |
| H11 | 0.04251 | -0.0093 | 0.18483 | 0.22138 |
| C12 | -0.03740 | 0.0145 | -0.18506 | -0.21867 |
| H13 | 0.05275 | -0.0098 | 0.18055 | 0.21685 |
| C14 | -0.05538 | 0.0100 | -0.18452 | -0.22300 |
| H15 | 0.05380 | -0.0091 | 0.18096 | 0.21842 |
| C16 | -0.14554 | 0.0160 | -0.20111 | -0.23715 |
| H17 | 0.04624 | -0.0054 | 0.18559 | 0.22835 |
| C18 | -0.07267 | 0.0202 | -0.37276 | -0.21672 |
| H19 | 0.10704 | 0.0216 | 0.16543 | 0.17928 |
| H20 | 0.11830 | 0.0332 | 0.18280 | 0.20066 |
| H21 | 0.10343 | 0.0190 | 0.16544 | 0.17642 |
| N22 | -0.44534 | -0.1766 | -0.44812 | -0.85203 |
| H23 | 0.35065 | 0.1962 | 0.34751 | 0.51061 |
| H24 | 0.26484 | 0.1229 | 0.28765 | 0.40643 |
| H25 | 0.25047 | 0.1125 | 0.28614 | 0.39769 |
| O26 | -0.33334 | -0.2032 | -0.10833 | -0.56673 |
 H contacts play dominant roles in the crystal structure of the investigated system. An analysis of the electronic structure and molecular modelling using charge distribution confirms the good electrophilic reactivity of the title compound.
H contacts play dominant roles in the crystal structure of the investigated system. An analysis of the electronic structure and molecular modelling using charge distribution confirms the good electrophilic reactivity of the title compound.
 journal menu
journal menu











