In ferrocene-1,1′-diylbis(diphenylmethanol)–4,4′-ethylenedipyridine (1/1), [Fe(C
18H
15O)
2]·C
12H
12N
2, there is an intramolecular O—H

O hydrogen bond in the ferrocenediol component and a single O—H

N hydrogen bond linking the two components into a finite monomeric adduct. Ferrocene-1,1′-diylbis(diphenylmethanol)–ethylenediamine (1/1), [Fe(C
18H
15O)
2]·C
2H
8N
2, crystallizes with
Z′ = 2 in space group
P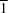
, and there are two independent four-component aggregates in the structure, both of which are centrosymmetric. In the first type of aggregate, the molecular components are linked by O—H

N and N—H

O hydrogen bonds, in which both diamine N atoms participate; in the second type of aggregate, the diamine component is disordered over two sets of sites, but only one N atom is involved in the hydrogen bonding.
Supporting information
CCDC references: 170175; 170176
For each adduct, stoichiometric quantities of the ferrocenediol and the
appropriate amine were separately dissolved in methanol: the component
solutions were mixed and the mixtures were set aside to crystallize producing
analytically pure (I) and (II), respectively. Analyses: compound (I), found C
78.5, H 5.9, N 3.7%; C48H42FeN2O2 requires C 78.5, H 5.8, N 3.8%;
compound (II), found C 74.2, H 6.3, N 4.6%; C38H38FeN2O2 requires C
74.7, H 6.3, N 4.6%. Crystals suitable for single-crystal X-ray diffraction
were selected directly from the analytical samples.
Compounds (I) and (II) both crystallize in the triclinic system; for each, space
group P1 was assumed and confirmed by the analysis. In (II), the
hydrogen-bonded N atoms of the two conformers of amine 2, N51 and N52, were
constrained to have identical coordinates and identical anisotropic
displacement parameters. H atoms were treated as riding atoms with C—H 0.95 Å (aryl and cyclopentadienyl) or 0.99 Å (aliphatic), N—H 0.91 Å and
O—H 0.84 Å.
For both compounds, data collection: Collect (Nonius, 1997-2000); cell refinement: DENZO-SMN (Otwinowski & Minor, 1997); data reduction: DENZO-SMN; program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: PLATON (Spek, 2001); software used to prepare material for publication: SHELXL97 and PRPKAPPA (Ferguson, 1999).
(I) Ferrocene-1,1'-diylbis(diphenylmethanol)–1,2-bis(4-pyridyl)ethane (1/1)
top
Crystal data top
| [Fe(C18H15O)]·C12H12N2 | Z = 2 |
| Mr = 734.69 | F(000) = 772 |
| Triclinic, P1 | Dx = 1.328 Mg m−3 |
| a = 9.7191 (3) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 11.3281 (4) Å | Cell parameters from 7609 reflections |
| c = 17.5549 (7) Å | θ = 2.7–27.2° |
| α = 78.6970 (16)° | µ = 0.45 mm−1 |
| β = 87.8100 (15)° | T = 150 K |
| γ = 75.772 (2)° | Block, orange |
| V = 1837.05 (11) Å3 | 0.20 × 0.18 × 0.18 mm |
Data collection top
Kappa-CCD
diffractometer | 8061 independent reflections |
| Radiation source: fine-focus sealed X-ray tube | 4882 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.094 |
| ϕ scans, and ω scans with κ offsets | θmax = 27.2°, θmin = 2.7° |
Absorption correction: multi-scan
(DENZO-SMN; Otwinowski & Minor, 1997) | h = 0→12 |
| Tmin = 0.915, Tmax = 0.923 | k = −13→14 |
| 23051 measured reflections | l = −22→22 |
Refinement top
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.051 | H-atom parameters constrained |
| wR(F2) = 0.125 | w = 1/[σ2(Fo2) + (0.0442P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 0.97 | (Δ/σ)max < 0.001 |
| 8061 reflections | Δρmax = 0.29 e Å−3 |
| 481 parameters | Δρmin = −0.40 e Å−3 |
| 0 restraints | Extinction correction: SHELXL97, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| Primary atom site location: structure-invariant direct methods | Extinction coefficient: 0.0031 (7) |
Crystal data top
| [Fe(C18H15O)]·C12H12N2 | γ = 75.772 (2)° |
| Mr = 734.69 | V = 1837.05 (11) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 9.7191 (3) Å | Mo Kα radiation |
| b = 11.3281 (4) Å | µ = 0.45 mm−1 |
| c = 17.5549 (7) Å | T = 150 K |
| α = 78.6970 (16)° | 0.20 × 0.18 × 0.18 mm |
| β = 87.8100 (15)° | |
Data collection top
Kappa-CCD
diffractometer | 8061 independent reflections |
Absorption correction: multi-scan
(DENZO-SMN; Otwinowski & Minor, 1997) | 4882 reflections with I > 2σ(I) |
| Tmin = 0.915, Tmax = 0.923 | Rint = 0.094 |
| 23051 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.051 | 0 restraints |
| wR(F2) = 0.125 | H-atom parameters constrained |
| S = 0.97 | Δρmax = 0.29 e Å−3 |
| 8061 reflections | Δρmin = −0.40 e Å−3 |
| 481 parameters | |
Special details top
Experimental. The program DENZO-SMN (Otwinowski & Minor, 1997) uses a scaling algorithm
[Fox, G·C. & Holmes, K·C. (1966). Acta Cryst. 20, 886–891] which
effectively corrects for absorption effects. High redundancy data were used in
the scaling program hence the 'multi-scan' code word was used. No transmission
coefficients are available from the program (only scale factors for each
frame). The scale factors in the experimental table are calculated from the
'size' command in the SHELXL97 input file. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Fe1 | 0.58246 (4) | 0.21972 (4) | 0.32690 (2) | 0.02559 (13) | |
| O1 | 0.32627 (19) | 0.16989 (16) | 0.19879 (10) | 0.0290 (4) | |
| O2 | 0.56163 (17) | −0.03784 (17) | 0.23881 (11) | 0.0299 (5) | |
| C1 | 0.2769 (3) | 0.2694 (2) | 0.24043 (15) | 0.0246 (6) | |
| C11 | 0.4045 (3) | 0.3133 (2) | 0.26100 (15) | 0.0239 (6) | |
| C12 | 0.4206 (3) | 0.3742 (2) | 0.32287 (16) | 0.0297 (6) | |
| C13 | 0.5531 (3) | 0.4061 (2) | 0.31567 (17) | 0.0323 (7) | |
| C14 | 0.6213 (3) | 0.3653 (3) | 0.24918 (17) | 0.0332 (7) | |
| C15 | 0.5307 (3) | 0.3092 (2) | 0.21527 (16) | 0.0282 (6) | |
| C31 | 0.1701 (3) | 0.3750 (2) | 0.18831 (15) | 0.0258 (6) | |
| C32 | 0.0514 (3) | 0.3485 (3) | 0.16009 (16) | 0.0319 (7) | |
| C33 | −0.0465 (3) | 0.4381 (3) | 0.11088 (17) | 0.0373 (7) | |
| C34 | −0.0283 (3) | 0.5576 (3) | 0.08990 (17) | 0.0410 (8) | |
| C35 | 0.0869 (3) | 0.5865 (3) | 0.11831 (18) | 0.0416 (8) | |
| C36 | 0.1853 (3) | 0.4951 (3) | 0.16740 (16) | 0.0326 (7) | |
| C41 | 0.2034 (3) | 0.2184 (2) | 0.31337 (15) | 0.0249 (6) | |
| C42 | 0.2149 (3) | 0.0909 (3) | 0.33677 (16) | 0.0299 (7) | |
| C43 | 0.1574 (3) | 0.0451 (3) | 0.40611 (16) | 0.0333 (7) | |
| C44 | 0.0866 (3) | 0.1241 (3) | 0.45359 (17) | 0.0382 (8) | |
| C45 | 0.0697 (3) | 0.2510 (3) | 0.42915 (16) | 0.0344 (7) | |
| C46 | 0.1264 (3) | 0.2976 (3) | 0.35983 (16) | 0.0310 (7) | |
| C2 | 0.6992 (3) | −0.0417 (2) | 0.26858 (16) | 0.0266 (6) | |
| C21 | 0.6802 (3) | 0.0371 (2) | 0.33112 (15) | 0.0255 (6) | |
| C22 | 0.5638 (3) | 0.0488 (2) | 0.38325 (16) | 0.0293 (6) | |
| C23 | 0.5875 (3) | 0.1191 (3) | 0.43750 (16) | 0.0307 (7) | |
| C24 | 0.7189 (3) | 0.1516 (3) | 0.41922 (16) | 0.0312 (7) | |
| C25 | 0.7762 (3) | 0.1011 (3) | 0.35407 (16) | 0.0296 (6) | |
| C51 | 0.7569 (3) | −0.1809 (3) | 0.30422 (17) | 0.0282 (6) | |
| C52 | 0.7502 (3) | −0.2266 (3) | 0.38375 (18) | 0.0352 (7) | |
| C53 | 0.7810 (3) | −0.3543 (3) | 0.41295 (19) | 0.0381 (8) | |
| C54 | 0.8226 (3) | −0.4368 (3) | 0.3628 (2) | 0.0407 (8) | |
| C55 | 0.8358 (3) | −0.3925 (3) | 0.2847 (2) | 0.0384 (8) | |
| C56 | 0.8026 (3) | −0.2649 (3) | 0.25523 (18) | 0.0332 (7) | |
| C61 | 0.7930 (3) | 0.0025 (2) | 0.20233 (16) | 0.0300 (7) | |
| C62 | 0.9381 (3) | −0.0143 (3) | 0.21361 (18) | 0.0352 (7) | |
| C63 | 1.0223 (3) | 0.0288 (3) | 0.1544 (2) | 0.0441 (8) | |
| C64 | 0.9633 (4) | 0.0887 (3) | 0.0825 (2) | 0.0498 (9) | |
| C65 | 0.8202 (4) | 0.1052 (3) | 0.06977 (19) | 0.0478 (9) | |
| C66 | 0.7355 (3) | 0.0629 (3) | 0.12927 (17) | 0.0353 (7) | |
| N71 | 0.3644 (3) | 0.2404 (2) | 0.04191 (14) | 0.0379 (6) | |
| C72 | 0.3967 (3) | 0.3484 (3) | 0.01159 (17) | 0.0431 (8) | |
| C73 | 0.4498 (3) | 0.3756 (3) | −0.06233 (17) | 0.0387 (7) | |
| C74 | 0.4717 (3) | 0.2889 (3) | −0.10968 (16) | 0.0290 (6) | |
| C75 | 0.4363 (3) | 0.1766 (3) | −0.07936 (17) | 0.0368 (7) | |
| C76 | 0.3853 (3) | 0.1572 (3) | −0.00449 (17) | 0.0393 (8) | |
| C77 | 0.5239 (3) | 0.3180 (3) | −0.19106 (16) | 0.0332 (7) | |
| N81 | 0.7512 (3) | 0.3633 (3) | −0.45181 (15) | 0.0462 (7) | |
| C82 | 0.6315 (3) | 0.3259 (3) | −0.43182 (18) | 0.0429 (8) | |
| C83 | 0.5999 (3) | 0.2765 (3) | −0.35746 (17) | 0.0364 (7) | |
| C84 | 0.6924 (3) | 0.2661 (3) | −0.29630 (17) | 0.0314 (7) | |
| C85 | 0.8146 (3) | 0.3056 (3) | −0.31637 (19) | 0.0384 (8) | |
| C86 | 0.8398 (3) | 0.3509 (3) | −0.3932 (2) | 0.0468 (9) | |
| C87 | 0.6532 (3) | 0.2231 (3) | −0.21328 (17) | 0.0346 (7) | |
| H1 | 0.3299 | 0.1993 | 0.1512 | 0.044* | |
| H2 | 0.5094 | 0.0342 | 0.2351 | 0.045* | |
| H12 | 0.3533 | 0.3906 | 0.3625 | 0.036* | |
| H13 | 0.5898 | 0.4474 | 0.3493 | 0.039* | |
| H14 | 0.7121 | 0.3742 | 0.2307 | 0.040* | |
| H15 | 0.5500 | 0.2746 | 0.1698 | 0.034* | |
| H32 | 0.0371 | 0.2671 | 0.1749 | 0.038* | |
| H33 | −0.1259 | 0.4175 | 0.0916 | 0.045* | |
| H34 | −0.0948 | 0.6193 | 0.0561 | 0.049* | |
| H35 | 0.0995 | 0.6685 | 0.1046 | 0.050* | |
| H36 | 0.2642 | 0.5161 | 0.1868 | 0.039* | |
| H42 | 0.2628 | 0.0355 | 0.3047 | 0.036* | |
| H43 | 0.1664 | −0.0418 | 0.4214 | 0.040* | |
| H44 | 0.0503 | 0.0919 | 0.5021 | 0.046* | |
| H45 | 0.0186 | 0.3064 | 0.4605 | 0.041* | |
| H46 | 0.1127 | 0.3848 | 0.3436 | 0.037* | |
| H22 | 0.4838 | 0.0151 | 0.3818 | 0.035* | |
| H23 | 0.5266 | 0.1407 | 0.4788 | 0.037* | |
| H24 | 0.7609 | 0.1992 | 0.4461 | 0.037* | |
| H25 | 0.8635 | 0.1084 | 0.3298 | 0.036* | |
| H52 | 0.7245 | −0.1703 | 0.4184 | 0.042* | |
| H53 | 0.7734 | −0.3844 | 0.4670 | 0.046* | |
| H54 | 0.8419 | −0.5237 | 0.3823 | 0.049* | |
| H55 | 0.8677 | −0.4491 | 0.2506 | 0.046* | |
| H56 | 0.8115 | −0.2354 | 0.2012 | 0.040* | |
| H62 | 0.9801 | −0.0558 | 0.2626 | 0.042* | |
| H63 | 1.1210 | 0.0171 | 0.1632 | 0.053* | |
| H64 | 1.0212 | 0.1185 | 0.0421 | 0.060* | |
| H65 | 0.7795 | 0.1455 | 0.0203 | 0.057* | |
| H66 | 0.6368 | 0.0753 | 0.1201 | 0.042* | |
| H72 | 0.3821 | 0.4100 | 0.0428 | 0.052* | |
| H73 | 0.4711 | 0.4538 | −0.0804 | 0.046* | |
| H75 | 0.4473 | 0.1143 | −0.1098 | 0.044* | |
| H76 | 0.3636 | 0.0797 | 0.0153 | 0.047* | |
| H77A | 0.5477 | 0.3995 | −0.1987 | 0.040* | |
| H77B | 0.4451 | 0.3263 | −0.2274 | 0.040* | |
| H82 | 0.5646 | 0.3343 | −0.4718 | 0.051* | |
| H83 | 0.5148 | 0.2492 | −0.3475 | 0.044* | |
| H85 | 0.8816 | 0.3015 | −0.2773 | 0.046* | |
| H86 | 0.9264 | 0.3749 | −0.4050 | 0.056* | |
| H87A | 0.7342 | 0.2144 | −0.1783 | 0.042* | |
| H87B | 0.6310 | 0.1411 | −0.2075 | 0.042* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Fe1 | 0.0260 (2) | 0.0237 (2) | 0.0258 (2) | −0.00525 (16) | −0.00004 (16) | −0.00246 (17) |
| O1 | 0.0327 (10) | 0.0272 (11) | 0.0249 (10) | −0.0037 (8) | 0.0019 (9) | −0.0043 (8) |
| O2 | 0.0264 (10) | 0.0247 (11) | 0.0380 (12) | −0.0032 (8) | −0.0068 (9) | −0.0073 (9) |
| C1 | 0.0280 (14) | 0.0220 (15) | 0.0216 (14) | −0.0007 (11) | 0.0007 (11) | −0.0063 (12) |
| C11 | 0.0255 (14) | 0.0198 (15) | 0.0235 (14) | −0.0037 (11) | 0.0007 (11) | 0.0006 (11) |
| C12 | 0.0316 (15) | 0.0251 (16) | 0.0303 (16) | −0.0029 (12) | 0.0006 (12) | −0.0051 (13) |
| C13 | 0.0346 (16) | 0.0216 (16) | 0.0398 (18) | −0.0073 (12) | −0.0051 (13) | −0.0026 (13) |
| C14 | 0.0306 (16) | 0.0278 (17) | 0.0380 (18) | −0.0096 (13) | −0.0020 (13) | 0.0046 (13) |
| C15 | 0.0301 (15) | 0.0246 (16) | 0.0263 (15) | −0.0040 (12) | −0.0001 (12) | 0.0004 (12) |
| C31 | 0.0274 (15) | 0.0254 (16) | 0.0220 (14) | −0.0027 (12) | 0.0012 (11) | −0.0035 (12) |
| C32 | 0.0342 (16) | 0.0297 (17) | 0.0320 (17) | −0.0092 (13) | −0.0017 (13) | −0.0044 (13) |
| C33 | 0.0337 (17) | 0.041 (2) | 0.0337 (18) | −0.0018 (14) | −0.0106 (13) | −0.0053 (15) |
| C34 | 0.0410 (18) | 0.037 (2) | 0.0349 (18) | 0.0047 (14) | −0.0094 (14) | 0.0009 (15) |
| C35 | 0.0471 (19) | 0.0298 (18) | 0.0420 (19) | −0.0038 (14) | −0.0076 (15) | 0.0017 (15) |
| C36 | 0.0326 (16) | 0.0293 (17) | 0.0341 (17) | −0.0057 (13) | −0.0054 (13) | −0.0029 (13) |
| C41 | 0.0229 (14) | 0.0278 (16) | 0.0224 (14) | −0.0062 (11) | −0.0021 (11) | −0.0006 (12) |
| C42 | 0.0222 (14) | 0.0314 (17) | 0.0334 (17) | −0.0034 (12) | −0.0016 (12) | −0.0039 (13) |
| C43 | 0.0256 (15) | 0.0341 (18) | 0.0366 (17) | −0.0105 (13) | −0.0024 (13) | 0.0063 (14) |
| C44 | 0.0308 (16) | 0.052 (2) | 0.0283 (17) | −0.0129 (15) | −0.0007 (13) | 0.0047 (15) |
| C45 | 0.0299 (16) | 0.044 (2) | 0.0266 (16) | −0.0053 (14) | 0.0035 (12) | −0.0049 (14) |
| C46 | 0.0290 (15) | 0.0329 (17) | 0.0278 (16) | −0.0039 (12) | 0.0009 (12) | −0.0027 (13) |
| C2 | 0.0232 (14) | 0.0228 (15) | 0.0335 (16) | −0.0059 (11) | −0.0029 (12) | −0.0038 (12) |
| C21 | 0.0260 (14) | 0.0214 (15) | 0.0264 (15) | −0.0040 (11) | −0.0014 (11) | −0.0004 (12) |
| C22 | 0.0268 (15) | 0.0265 (16) | 0.0318 (16) | −0.0059 (12) | −0.0019 (12) | 0.0010 (13) |
| C23 | 0.0339 (16) | 0.0315 (17) | 0.0248 (15) | −0.0056 (13) | 0.0018 (12) | −0.0039 (13) |
| C24 | 0.0342 (16) | 0.0300 (17) | 0.0278 (16) | −0.0063 (13) | −0.0062 (12) | −0.0027 (13) |
| C25 | 0.0245 (14) | 0.0304 (17) | 0.0318 (16) | −0.0054 (12) | −0.0030 (12) | −0.0019 (13) |
| C51 | 0.0185 (13) | 0.0243 (16) | 0.0407 (17) | −0.0043 (11) | −0.0013 (12) | −0.0043 (13) |
| C52 | 0.0299 (16) | 0.0308 (18) | 0.0426 (19) | −0.0044 (13) | −0.0026 (13) | −0.0052 (14) |
| C53 | 0.0319 (16) | 0.0293 (18) | 0.0458 (19) | −0.0042 (13) | −0.0064 (14) | 0.0070 (15) |
| C54 | 0.0317 (17) | 0.0238 (17) | 0.063 (2) | −0.0038 (13) | −0.0066 (15) | −0.0025 (16) |
| C55 | 0.0305 (16) | 0.0257 (17) | 0.060 (2) | −0.0045 (13) | −0.0033 (15) | −0.0134 (16) |
| C56 | 0.0300 (16) | 0.0285 (17) | 0.0425 (18) | −0.0062 (13) | −0.0021 (13) | −0.0107 (14) |
| C61 | 0.0339 (16) | 0.0225 (16) | 0.0340 (17) | −0.0048 (12) | 0.0022 (13) | −0.0096 (13) |
| C62 | 0.0328 (16) | 0.0280 (17) | 0.0443 (19) | −0.0061 (13) | 0.0024 (14) | −0.0079 (14) |
| C63 | 0.0360 (18) | 0.038 (2) | 0.061 (2) | −0.0107 (15) | 0.0123 (16) | −0.0152 (17) |
| C64 | 0.060 (2) | 0.042 (2) | 0.050 (2) | −0.0173 (17) | 0.0223 (18) | −0.0133 (17) |
| C65 | 0.062 (2) | 0.047 (2) | 0.0339 (19) | −0.0147 (17) | 0.0068 (16) | −0.0077 (16) |
| C66 | 0.0425 (18) | 0.0340 (18) | 0.0326 (17) | −0.0107 (14) | 0.0007 (14) | −0.0120 (14) |
| N71 | 0.0460 (15) | 0.0362 (16) | 0.0286 (14) | −0.0050 (12) | −0.0015 (11) | −0.0053 (12) |
| C72 | 0.062 (2) | 0.041 (2) | 0.0296 (18) | −0.0133 (16) | −0.0013 (15) | −0.0139 (15) |
| C73 | 0.0482 (19) | 0.0355 (19) | 0.0359 (18) | −0.0157 (15) | 0.0010 (14) | −0.0079 (15) |
| C74 | 0.0282 (15) | 0.0270 (16) | 0.0291 (16) | −0.0024 (12) | −0.0047 (12) | −0.0038 (13) |
| C75 | 0.0534 (19) | 0.0293 (17) | 0.0293 (17) | −0.0114 (14) | 0.0029 (14) | −0.0081 (13) |
| C76 | 0.054 (2) | 0.0271 (17) | 0.0338 (18) | −0.0080 (14) | 0.0066 (15) | −0.0026 (14) |
| C77 | 0.0331 (16) | 0.0315 (17) | 0.0331 (17) | −0.0067 (13) | 0.0017 (13) | −0.0035 (13) |
| N81 | 0.0501 (17) | 0.0551 (19) | 0.0402 (16) | −0.0214 (14) | 0.0156 (13) | −0.0179 (14) |
| C82 | 0.0421 (19) | 0.058 (2) | 0.0355 (19) | −0.0170 (16) | 0.0059 (14) | −0.0207 (17) |
| C83 | 0.0342 (17) | 0.046 (2) | 0.0353 (18) | −0.0183 (14) | 0.0028 (13) | −0.0127 (15) |
| C84 | 0.0321 (16) | 0.0272 (16) | 0.0364 (17) | −0.0071 (13) | 0.0070 (13) | −0.0109 (13) |
| C85 | 0.0254 (16) | 0.0394 (19) | 0.052 (2) | −0.0056 (13) | 0.0034 (14) | −0.0166 (16) |
| C86 | 0.0370 (18) | 0.057 (2) | 0.058 (2) | −0.0238 (16) | 0.0190 (17) | −0.0268 (19) |
| C87 | 0.0315 (16) | 0.0313 (17) | 0.0384 (18) | −0.0055 (13) | −0.0025 (13) | −0.0025 (14) |
Geometric parameters (Å, º) top
| Fe1—C14 | 2.027 (3) | C23—C24 | 1.422 (4) |
| Fe1—C13 | 2.030 (3) | C23—H23 | 0.95 |
| Fe1—C12 | 2.036 (3) | C24—C25 | 1.413 (4) |
| Fe1—C24 | 2.037 (3) | C24—H24 | 0.95 |
| Fe1—C25 | 2.038 (3) | C25—H25 | 0.95 |
| Fe1—C22 | 2.041 (3) | C51—C56 | 1.388 (4) |
| Fe1—C21 | 2.042 (3) | C51—C52 | 1.396 (4) |
| Fe1—C15 | 2.041 (3) | C52—C53 | 1.397 (4) |
| Fe1—C23 | 2.045 (3) | C52—H52 | 0.95 |
| Fe1—C11 | 2.055 (2) | C53—C54 | 1.386 (4) |
| O1—C1 | 1.437 (3) | C53—H53 | 0.95 |
| O1—H1 | 0.8400 | C54—C55 | 1.378 (4) |
| O2—C2 | 1.441 (3) | C54—H54 | 0.95 |
| O2—H2 | 0.84 | C55—C56 | 1.397 (4) |
| C1—C11 | 1.525 (4) | C55—H55 | 0.95 |
| C1—C41 | 1.525 (3) | C56—H56 | 0.95 |
| C1—C31 | 1.543 (3) | C61—C62 | 1.393 (4) |
| C11—C12 | 1.429 (4) | C61—C66 | 1.396 (4) |
| C11—C15 | 1.436 (3) | C62—C63 | 1.386 (4) |
| C12—C13 | 1.415 (4) | C62—H62 | 0.95 |
| C12—H12 | 0.95 | C63—C64 | 1.381 (4) |
| C13—C14 | 1.420 (4) | C63—H63 | 0.95 |
| C13—H13 | 0.95 | C64—C65 | 1.378 (4) |
| C14—C15 | 1.414 (4) | C64—H64 | 0.95 |
| C14—H14 | 0.95 | C65—C66 | 1.390 (4) |
| C15—H15 | 0.95 | C65—H65 | 0.95 |
| C31—C36 | 1.381 (4) | C66—H66 | 0.95 |
| C31—C32 | 1.393 (4) | N71—C76 | 1.337 (4) |
| C32—C33 | 1.388 (4) | N71—C72 | 1.341 (4) |
| C32—H32 | 0.95 | C72—C73 | 1.384 (4) |
| C33—C34 | 1.385 (4) | C72—H72 | 0.95 |
| C33—H33 | 0.95 | C73—C74 | 1.380 (4) |
| C34—C35 | 1.376 (4) | C73—H73 | 0.95 |
| C34—H34 | 0.95 | C74—C75 | 1.398 (4) |
| C35—C36 | 1.398 (4) | C74—C77 | 1.500 (4) |
| C35—H35 | 0.95 | C75—C76 | 1.383 (4) |
| C36—H36 | 0.95 | C75—H75 | 0.95 |
| C41—C46 | 1.393 (4) | C76—H76 | 0.95 |
| C41—C42 | 1.400 (4) | C77—C87 | 1.533 (4) |
| C42—C43 | 1.381 (4) | C77—H77A | 0.99 |
| C42—H42 | 0.95 | C77—H77B | 0.99 |
| C43—C44 | 1.384 (4) | N81—C86 | 1.331 (4) |
| C43—H43 | 0.95 | N81—C82 | 1.346 (4) |
| C44—C45 | 1.388 (4) | C82—C83 | 1.371 (4) |
| C44—H44 | 0.95 | C82—H82 | 0.95 |
| C45—C46 | 1.379 (4) | C83—C84 | 1.396 (4) |
| C45—H45 | 0.95 | C83—H83 | 0.95 |
| C46—H46 | 0.95 | C84—C85 | 1.379 (4) |
| C2—C21 | 1.524 (4) | C84—C87 | 1.509 (4) |
| C2—C61 | 1.533 (4) | C85—C86 | 1.381 (4) |
| C2—C51 | 1.549 (4) | C85—H85 | 0.95 |
| C21—C22 | 1.425 (4) | C86—H86 | 0.95 |
| C21—C25 | 1.426 (4) | C87—H87A | 0.99 |
| C22—C23 | 1.413 (4) | C87—H87B | 0.99 |
| C22—H22 | 0.95 | | |
| | | |
| C1—O1—H1 | 109.5 | C56—C51—C52 | 118.6 (3) |
| C2—O2—H2 | 109.5 | C56—C51—C2 | 119.2 (3) |
| O1—C1—C11 | 108.4 (2) | C52—C51—C2 | 121.8 (2) |
| O1—C1—C41 | 107.4 (2) | C51—C52—C53 | 120.7 (3) |
| C11—C1—C41 | 111.1 (2) | C51—C52—H52 | 119.6 |
| O1—C1—C31 | 108.6 (2) | C53—C52—H52 | 119.6 |
| C11—C1—C31 | 111.1 (2) | C54—C53—C52 | 119.8 (3) |
| C41—C1—C31 | 110.0 (2) | C54—C53—H53 | 120.1 |
| C12—C11—C15 | 106.3 (2) | C52—C53—H53 | 120.1 |
| C12—C11—C1 | 128.9 (2) | C55—C54—C53 | 119.8 (3) |
| C15—C11—C1 | 124.6 (2) | C55—C54—H54 | 120.1 |
| C13—C12—C11 | 109.0 (2) | C53—C54—H54 | 120.1 |
| C13—C12—H12 | 125.5 | C54—C55—C56 | 120.4 (3) |
| C11—C12—H12 | 125.5 | C54—C55—H55 | 119.8 |
| C12—C13—C14 | 107.8 (3) | C56—C55—H55 | 119.8 |
| C12—C13—H13 | 126.1 | C51—C56—C55 | 120.6 (3) |
| C14—C13—H13 | 126.1 | C51—C56—H56 | 119.7 |
| C15—C14—C13 | 108.1 (2) | C55—C56—H56 | 119.7 |
| C15—C14—H14 | 125.9 | C62—C61—C66 | 117.9 (3) |
| C13—C14—H14 | 125.9 | C62—C61—C2 | 121.0 (3) |
| C14—C15—C11 | 108.7 (2) | C66—C61—C2 | 121.1 (2) |
| C14—C15—H15 | 125.7 | C63—C62—C61 | 121.0 (3) |
| C11—C15—H15 | 125.7 | C63—C62—H62 | 119.5 |
| C36—C31—C32 | 117.6 (2) | C61—C62—H62 | 119.5 |
| C36—C31—C1 | 123.9 (2) | C64—C63—C62 | 120.3 (3) |
| C32—C31—C1 | 118.5 (2) | C64—C63—H63 | 119.8 |
| C33—C32—C31 | 121.5 (3) | C62—C63—H63 | 119.8 |
| C33—C32—H32 | 119.2 | C65—C64—C63 | 119.7 (3) |
| C31—C32—H32 | 119.2 | C65—C64—H64 | 120.1 |
| C34—C33—C32 | 119.8 (3) | C63—C64—H64 | 120.1 |
| C34—C33—H33 | 120.1 | C64—C65—C66 | 120.0 (3) |
| C32—C33—H33 | 120.1 | C64—C65—H65 | 120.0 |
| C35—C34—C33 | 119.6 (3) | C66—C65—H65 | 120.0 |
| C35—C34—H34 | 120.2 | C65—C66—C61 | 121.1 (3) |
| C33—C34—H34 | 120.2 | C65—C66—H66 | 119.5 |
| C34—C35—C36 | 120.0 (3) | C61—C66—H66 | 119.5 |
| C34—C35—H35 | 120.0 | C76—N71—C72 | 115.7 (3) |
| C36—C35—H35 | 120.0 | N71—C72—C73 | 124.0 (3) |
| C31—C36—C35 | 121.4 (3) | N71—C72—H72 | 118.0 |
| C31—C36—H36 | 119.3 | C73—C72—H72 | 118.0 |
| C35—C36—H36 | 119.3 | C74—C73—C72 | 119.8 (3) |
| C46—C41—C42 | 118.2 (3) | C74—C73—H73 | 120.1 |
| C46—C41—C1 | 120.7 (2) | C72—C73—H73 | 120.1 |
| C42—C41—C1 | 121.1 (2) | C73—C74—C75 | 116.8 (3) |
| C43—C42—C41 | 120.5 (3) | C73—C74—C77 | 120.8 (3) |
| C43—C42—H42 | 119.7 | C75—C74—C77 | 122.3 (3) |
| C41—C42—H42 | 119.7 | C76—C75—C74 | 119.2 (3) |
| C42—C43—C44 | 120.8 (3) | C76—C75—H75 | 120.4 |
| C42—C43—H43 | 119.6 | C74—C75—H75 | 120.4 |
| C44—C43—H43 | 119.6 | N71—C76—C75 | 124.4 (3) |
| C43—C44—C45 | 118.9 (3) | N71—C76—H76 | 117.8 |
| C43—C44—H44 | 120.5 | C75—C76—H76 | 117.8 |
| C45—C44—H44 | 120.5 | C74—C77—C87 | 116.0 (2) |
| C46—C45—C44 | 120.6 (3) | C74—C77—H77A | 108.3 |
| C46—C45—H45 | 119.7 | C87—C77—H77A | 108.3 |
| C44—C45—H45 | 119.7 | C74—C77—H77B | 108.3 |
| C45—C46—C41 | 120.8 (3) | C87—C77—H77B | 108.3 |
| C45—C46—H46 | 119.6 | H77A—C77—H77B | 107.4 |
| C41—C46—H46 | 119.6 | C86—N81—C82 | 115.1 (3) |
| O2—C2—C21 | 109.1 (2) | N81—C82—C83 | 124.2 (3) |
| O2—C2—C61 | 110.2 (2) | N81—C82—H82 | 117.9 |
| C21—C2—C61 | 111.9 (2) | C83—C82—H82 | 117.9 |
| O2—C2—C51 | 102.9 (2) | C82—C83—C84 | 120.1 (3) |
| C21—C2—C51 | 110.9 (2) | C82—C83—H83 | 119.9 |
| C61—C2—C51 | 111.6 (2) | C84—C83—H83 | 119.9 |
| C22—C21—C25 | 107.3 (2) | C85—C84—C83 | 115.9 (3) |
| C22—C21—C2 | 124.0 (2) | C85—C84—C87 | 123.0 (3) |
| C25—C21—C2 | 128.4 (2) | C83—C84—C87 | 120.9 (2) |
| C23—C22—C21 | 108.5 (2) | C84—C85—C86 | 120.1 (3) |
| C23—C22—H22 | 125.8 | C84—C85—H85 | 120.0 |
| C21—C22—H22 | 125.8 | C86—C85—H85 | 120.0 |
| C22—C23—C24 | 107.8 (2) | N81—C86—C85 | 124.6 (3) |
| C22—C23—H23 | 126.1 | N81—C86—H86 | 117.7 |
| C24—C23—H23 | 126.1 | C85—C86—H86 | 117.7 |
| C25—C24—C23 | 108.2 (3) | C84—C87—C77 | 109.4 (2) |
| C25—C24—H24 | 125.9 | C84—C87—H87A | 109.8 |
| C23—C24—H24 | 125.9 | C77—C87—H87A | 109.8 |
| C24—C25—C21 | 108.2 (2) | C84—C87—H87B | 109.8 |
| C24—C25—H25 | 125.9 | C77—C87—H87B | 109.8 |
| C21—C25—H25 | 125.9 | H87A—C87—H87B | 108.2 |
| | | |
| O1—C1—C11—C12 | −155.0 (2) | C22—C23—C24—C25 | −0.3 (3) |
| C31—C1—C11—C12 | 85.7 (3) | C23—C24—C25—C21 | 0.3 (3) |
| C41—C1—C11—C12 | −37.2 (4) | C22—C21—C25—C24 | −0.2 (3) |
| O1—C1—C11—C15 | 30.6 (3) | C2—C21—C25—C24 | −174.4 (3) |
| C41—C1—C11—C15 | 148.5 (2) | O2—C2—C51—C56 | 73.9 (3) |
| C31—C1—C11—C15 | −88.6 (3) | C21—C2—C51—C56 | −169.7 (2) |
| C15—C11—C12—C13 | −0.3 (3) | C61—C2—C51—C56 | −44.3 (3) |
| C1—C11—C12—C13 | −175.4 (2) | O2—C2—C51—C52 | −99.2 (3) |
| C11—C12—C13—C14 | −0.1 (3) | C21—C2—C51—C52 | 17.3 (3) |
| C12—C13—C14—C15 | 0.4 (3) | C61—C2—C51—C52 | 142.7 (2) |
| C13—C14—C15—C11 | −0.6 (3) | C56—C51—C52—C53 | −3.7 (4) |
| C12—C11—C15—C14 | 0.5 (3) | C2—C51—C52—C53 | 169.4 (2) |
| C1—C11—C15—C14 | 175.9 (2) | C51—C52—C53—C54 | 1.9 (4) |
| O1—C1—C31—C36 | −122.8 (3) | C52—C53—C54—C55 | 1.1 (4) |
| C11—C1—C31—C36 | −3.7 (4) | C53—C54—C55—C56 | −2.3 (4) |
| C41—C1—C31—C36 | 119.9 (3) | C52—C51—C56—C55 | 2.5 (4) |
| O1—C1—C31—C32 | 56.9 (3) | C2—C51—C56—C55 | −170.7 (2) |
| C11—C1—C31—C32 | 176.0 (2) | C54—C55—C56—C51 | 0.4 (4) |
| C41—C1—C31—C32 | −60.4 (3) | O2—C2—C61—C62 | −168.0 (2) |
| C36—C31—C32—C33 | 1.8 (4) | C21—C2—C61—C62 | 70.5 (3) |
| C1—C31—C32—C33 | −177.9 (3) | C51—C2—C61—C62 | −54.3 (3) |
| C31—C32—C33—C34 | −1.1 (4) | O2—C2—C61—C66 | 13.2 (3) |
| C32—C33—C34—C35 | −0.2 (5) | C21—C2—C61—C66 | −108.3 (3) |
| C33—C34—C35—C36 | 0.6 (5) | C51—C2—C61—C66 | 126.9 (3) |
| C32—C31—C36—C35 | −1.4 (4) | C66—C61—C62—C63 | 0.7 (4) |
| C1—C31—C36—C35 | 178.3 (3) | C2—C61—C62—C63 | −178.2 (3) |
| C34—C35—C36—C31 | 0.2 (5) | C61—C62—C63—C64 | −0.4 (5) |
| O1—C1—C41—C46 | −170.6 (2) | C62—C63—C64—C65 | −0.3 (5) |
| C11—C1—C41—C46 | 70.9 (3) | C63—C64—C65—C66 | 0.8 (5) |
| C31—C1—C41—C46 | −52.6 (3) | C64—C65—C66—C61 | −0.5 (5) |
| O1—C1—C41—C42 | 11.4 (3) | C62—C61—C66—C65 | −0.2 (4) |
| C11—C1—C41—C42 | −107.1 (3) | C2—C61—C66—C65 | 178.7 (3) |
| C31—C1—C41—C42 | 129.4 (2) | C76—N71—C72—C73 | −0.7 (4) |
| C46—C41—C42—C43 | −3.2 (4) | N71—C72—C73—C74 | 0.4 (5) |
| C1—C41—C42—C43 | 174.9 (2) | C72—C73—C74—C75 | 0.7 (4) |
| C41—C42—C43—C44 | 0.3 (4) | C72—C73—C74—C77 | 177.7 (2) |
| C42—C43—C44—C45 | 2.4 (4) | C73—C74—C75—C76 | −1.4 (4) |
| C43—C44—C45—C46 | −2.1 (4) | C77—C74—C75—C76 | −178.3 (2) |
| C44—C45—C46—C41 | −0.9 (4) | C72—N71—C76—C75 | −0.1 (4) |
| C42—C41—C46—C45 | 3.5 (4) | C74—C75—C76—N71 | 1.1 (5) |
| C1—C41—C46—C45 | −174.6 (2) | C75—C74—C77—C87 | −54.6 (4) |
| C73—C74—C77—C87 | 128.6 (3) | C86—N81—C82—C83 | −0.9 (5) |
| C83—C84—C87—C77 | −64.3 (3) | N81—C82—C83—C84 | 2.2 (5) |
| O2—C2—C21—C22 | 34.4 (3) | C82—C83—C84—C85 | −1.3 (4) |
| C51—C2—C21—C22 | −78.2 (3) | C82—C83—C84—C87 | 174.3 (3) |
| C61—C2—C21—C22 | 156.6 (2) | C83—C84—C85—C86 | −0.5 (4) |
| O2—C2—C21—C25 | −152.2 (2) | C87—C84—C85—C86 | −176.0 (3) |
| C61—C2—C21—C25 | −30.0 (4) | C82—N81—C86—C85 | −1.1 (5) |
| C51—C2—C21—C25 | 95.2 (3) | C84—C85—C86—N81 | 1.8 (5) |
| C25—C21—C22—C23 | 0.0 (3) | C85—C84—C87—C77 | 110.9 (3) |
| C2—C21—C22—C23 | 174.6 (2) | C74—C77—C87—C84 | −179.8 (2) |
| C21—C22—C23—C24 | 0.1 (3) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1···N71 | 0.84 | 1.92 | 2.751 (3) | 170 |
| O2—H2···O1 | 0.84 | 2.07 | 2.845 (2) | 153 |
(II) Ferrocene-1,1'-diylbis(diphenylmethanol)–1,2-diaminoethane (1/1)
top
Crystal data top
| [Fe(C18H15O)]·C2H8N2 | Z = 4 |
| Mr = 610.55 | F(000) = 1288 |
| Triclinic, P1 | Dx = 1.338 Mg m−3 |
| a = 9.8551 (2) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 11.7183 (2) Å | Cell parameters from 12683 reflections |
| c = 27.6810 (5) Å | θ = 2.7–27.5° |
| α = 82.3495 (8)° | µ = 0.54 mm−1 |
| β = 84.4918 (8)° | T = 150 K |
| γ = 73.4846 (8)° | Block, orange |
| V = 3031.88 (10) Å3 | 0.32 × 0.28 × 0.26 mm |
Data collection top
Kappa-CCD
diffractometer | 13826 independent reflections |
| Radiation source: fine-focus sealed X-ray tube | 8093 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.088 |
| ϕ scans, and ω scans with κ offsets | θmax = 27.5°, θmin = 2.7° |
Absorption correction: multi-scan
(DENZO-SMN; Otwinowski & Minor, 1997) | h = 0→12 |
| Tmin = 0.848, Tmax = 0.873 | k = −14→15 |
| 39122 measured reflections | l = −35→35 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.050 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.123 | H-atom parameters constrained |
| S = 0.98 | w = 1/[σ2(Fo2) + (0.043P)2]
where P = (Fo2 + 2Fc2)/3 |
| 13826 reflections | (Δ/σ)max < 0.001 |
| 795 parameters | Δρmax = 0.38 e Å−3 |
| 9 restraints | Δρmin = −0.42 e Å−3 |
Crystal data top
| [Fe(C18H15O)]·C2H8N2 | γ = 73.4846 (8)° |
| Mr = 610.55 | V = 3031.88 (10) Å3 |
| Triclinic, P1 | Z = 4 |
| a = 9.8551 (2) Å | Mo Kα radiation |
| b = 11.7183 (2) Å | µ = 0.54 mm−1 |
| c = 27.6810 (5) Å | T = 150 K |
| α = 82.3495 (8)° | 0.32 × 0.28 × 0.26 mm |
| β = 84.4918 (8)° | |
Data collection top
Kappa-CCD
diffractometer | 13826 independent reflections |
Absorption correction: multi-scan
(DENZO-SMN; Otwinowski & Minor, 1997) | 8093 reflections with I > 2σ(I) |
| Tmin = 0.848, Tmax = 0.873 | Rint = 0.088 |
| 39122 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.050 | 9 restraints |
| wR(F2) = 0.123 | H-atom parameters constrained |
| S = 0.98 | Δρmax = 0.38 e Å−3 |
| 13826 reflections | Δρmin = −0.42 e Å−3 |
| 795 parameters | |
Special details top
Experimental. The program DENZO-SMN (Otwinowski & Minor, 1997) uses a scaling algorithm
[Fox, G·C. & Holmes, K·C. (1966). Acta Cryst. 20, 886–891] which
effectively corrects for absorption effects. High redundancy data were used in
the scaling program hence the 'multi-scan' code word was used. No transmission
coefficients are available from the program (only scale factors for each
frame). The scale factors in the experimental table are calculated from the
'size' command in the SHELXL97 input file. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Fe1 | 0.40357 (4) | 0.36346 (3) | 0.347750 (14) | 0.02290 (11) | |
| O11 | 0.1126 (2) | 0.37855 (14) | 0.44166 (7) | 0.0262 (4) | |
| O12 | 0.0750 (2) | 0.58204 (15) | 0.38063 (7) | 0.0294 (5) | |
| C11 | 0.1845 (3) | 0.2618 (2) | 0.42763 (10) | 0.0230 (6) | |
| C12 | 0.1783 (3) | 0.6316 (2) | 0.35284 (10) | 0.0245 (6) | |
| C111 | 0.3327 (3) | 0.2615 (2) | 0.40629 (10) | 0.0229 (6) | |
| C112 | 0.4194 (3) | 0.1877 (2) | 0.37181 (10) | 0.0257 (6) | |
| C113 | 0.5558 (3) | 0.2080 (2) | 0.36650 (10) | 0.0295 (7) | |
| C114 | 0.5550 (3) | 0.2953 (2) | 0.39781 (10) | 0.0308 (7) | |
| C115 | 0.4179 (3) | 0.3286 (2) | 0.42184 (10) | 0.0264 (6) | |
| C121 | 0.2846 (3) | 0.5360 (2) | 0.32572 (10) | 0.0239 (6) | |
| C122 | 0.2445 (3) | 0.4498 (2) | 0.30225 (10) | 0.0283 (7) | |
| C123 | 0.3656 (3) | 0.3851 (2) | 0.27576 (10) | 0.0321 (7) | |
| C124 | 0.4803 (3) | 0.4294 (2) | 0.28212 (10) | 0.0320 (7) | |
| C125 | 0.4314 (3) | 0.5225 (2) | 0.31309 (10) | 0.0258 (6) | |
| C131 | 0.1894 (3) | 0.1718 (2) | 0.47375 (10) | 0.0239 (6) | |
| C132 | 0.0628 (3) | 0.1550 (3) | 0.49574 (11) | 0.0319 (7) | |
| C133 | 0.0578 (3) | 0.0811 (3) | 0.53873 (11) | 0.0380 (8) | |
| C134 | 0.1832 (3) | 0.0213 (3) | 0.56087 (11) | 0.0377 (8) | |
| C135 | 0.3093 (3) | 0.0384 (3) | 0.54034 (11) | 0.0397 (8) | |
| C136 | 0.3131 (3) | 0.1133 (2) | 0.49686 (10) | 0.0317 (7) | |
| C141 | 0.0981 (3) | 0.2345 (2) | 0.38947 (10) | 0.0236 (6) | |
| C142 | −0.0079 (3) | 0.3232 (2) | 0.36603 (10) | 0.0285 (7) | |
| C143 | −0.0818 (3) | 0.2971 (3) | 0.33056 (11) | 0.0344 (7) | |
| C144 | −0.0497 (3) | 0.1824 (3) | 0.31768 (11) | 0.0349 (7) | |
| C145 | 0.0543 (3) | 0.0924 (3) | 0.34149 (11) | 0.0325 (7) | |
| C146 | 0.1253 (3) | 0.1187 (2) | 0.37717 (10) | 0.0264 (6) | |
| C151 | 0.0921 (3) | 0.7293 (2) | 0.31591 (10) | 0.0258 (6) | |
| C152 | 0.1340 (3) | 0.7433 (2) | 0.26762 (11) | 0.0324 (7) | |
| C153 | 0.0523 (3) | 0.8327 (3) | 0.23519 (12) | 0.0406 (8) | |
| C154 | −0.0734 (3) | 0.9083 (3) | 0.25216 (13) | 0.0412 (9) | |
| C155 | −0.1164 (3) | 0.8951 (2) | 0.30077 (13) | 0.0389 (8) | |
| C156 | −0.0341 (3) | 0.8072 (2) | 0.33319 (11) | 0.0323 (7) | |
| C161 | 0.2509 (3) | 0.6900 (2) | 0.38555 (10) | 0.0270 (7) | |
| C162 | 0.3241 (3) | 0.7719 (2) | 0.36530 (11) | 0.0309 (7) | |
| C163 | 0.3839 (3) | 0.8287 (2) | 0.39506 (12) | 0.0395 (8) | |
| C164 | 0.3716 (3) | 0.8046 (3) | 0.44523 (13) | 0.0414 (9) | |
| C165 | 0.3025 (3) | 0.7210 (3) | 0.46562 (12) | 0.0437 (9) | |
| C166 | 0.2418 (3) | 0.6637 (3) | 0.43631 (11) | 0.0340 (7) | |
| Fe2 | 0.37640 (4) | 0.06863 (3) | 0.195217 (14) | 0.02341 (11) | |
| O21 | 0.4933 (2) | 0.10212 (15) | 0.06452 (6) | 0.0254 (4) | |
| O22 | 0.63074 (19) | −0.11735 (15) | 0.11437 (6) | 0.0271 (4) | |
| C21 | 0.4444 (3) | 0.2080 (2) | 0.08943 (10) | 0.0246 (6) | |
| C22 | 0.5795 (3) | −0.1800 (2) | 0.15691 (10) | 0.0242 (6) | |
| C211 | 0.3431 (3) | 0.1848 (2) | 0.13210 (10) | 0.0244 (6) | |
| C212 | 0.2990 (3) | 0.2481 (2) | 0.17409 (10) | 0.0258 (6) | |
| C213 | 0.1929 (3) | 0.2028 (2) | 0.20202 (11) | 0.0313 (7) | |
| C214 | 0.1700 (3) | 0.1115 (2) | 0.17777 (10) | 0.0312 (7) | |
| C215 | 0.2620 (3) | 0.1007 (2) | 0.13465 (10) | 0.0272 (7) | |
| C221 | 0.5254 (3) | −0.0952 (2) | 0.19609 (10) | 0.0243 (6) | |
| C222 | 0.5892 (3) | −0.0050 (2) | 0.20404 (10) | 0.0267 (7) | |
| C223 | 0.5210 (3) | 0.0481 (2) | 0.24620 (10) | 0.0287 (7) | |
| C224 | 0.4150 (3) | −0.0082 (2) | 0.26501 (10) | 0.0300 (7) | |
| C225 | 0.4168 (3) | −0.0970 (2) | 0.23399 (9) | 0.0263 (6) | |
| C231 | 0.3697 (3) | 0.3136 (2) | 0.05295 (10) | 0.0270 (7) | |
| C232 | 0.4432 (3) | 0.3397 (3) | 0.00960 (11) | 0.0360 (8) | |
| C233 | 0.3806 (4) | 0.4317 (3) | −0.02505 (12) | 0.0438 (9) | |
| C234 | 0.2440 (4) | 0.5009 (3) | −0.01618 (12) | 0.0489 (10) | |
| C235 | 0.1699 (4) | 0.4762 (3) | 0.02674 (12) | 0.0522 (10) | |
| C236 | 0.2314 (4) | 0.3823 (3) | 0.06141 (11) | 0.0427 (9) | |
| C241 | 0.5748 (3) | 0.2350 (2) | 0.10606 (10) | 0.0251 (6) | |
| C242 | 0.5609 (3) | 0.3409 (2) | 0.12663 (10) | 0.0307 (7) | |
| C243 | 0.6757 (3) | 0.3632 (3) | 0.14466 (11) | 0.0377 (8) | |
| C244 | 0.8093 (3) | 0.2817 (3) | 0.14131 (12) | 0.0424 (8) | |
| C245 | 0.8248 (3) | 0.1795 (3) | 0.11934 (12) | 0.0388 (8) | |
| C246 | 0.7093 (3) | 0.1551 (2) | 0.10208 (11) | 0.0314 (7) | |
| C251 | 0.7098 (3) | −0.2816 (2) | 0.17373 (10) | 0.0261 (6) | |
| C252 | 0.7930 (3) | −0.3504 (2) | 0.13907 (11) | 0.0321 (7) | |
| C253 | 0.9109 (3) | −0.4426 (2) | 0.15229 (12) | 0.0381 (8) | |
| C254 | 0.9469 (3) | −0.4685 (3) | 0.20033 (13) | 0.0425 (9) | |
| C255 | 0.8630 (4) | −0.4018 (3) | 0.23542 (12) | 0.0435 (9) | |
| C256 | 0.7454 (3) | −0.3089 (2) | 0.22204 (11) | 0.0356 (8) | |
| C261 | 0.4661 (3) | −0.2356 (2) | 0.14416 (10) | 0.0244 (6) | |
| C262 | 0.3970 (3) | −0.1963 (3) | 0.10060 (11) | 0.0331 (7) | |
| C263 | 0.2947 (3) | −0.2467 (3) | 0.08924 (13) | 0.0438 (9) | |
| C264 | 0.2573 (3) | −0.3360 (3) | 0.12106 (13) | 0.0457 (9) | |
| C265 | 0.3260 (3) | −0.3762 (3) | 0.16431 (12) | 0.0389 (8) | |
| C266 | 0.4293 (3) | −0.3264 (2) | 0.17523 (11) | 0.0320 (7) | |
| N1 | 0.1551 (2) | 0.4189 (2) | 0.53263 (8) | 0.0315 (6) | |
| C2 | 0.3019 (3) | 0.4024 (3) | 0.54452 (11) | 0.0373 (8) | |
| C3 | 0.3210 (3) | 0.4354 (3) | 0.59423 (10) | 0.0355 (7) | |
| N4 | 0.2705 (3) | 0.3680 (3) | 0.63668 (9) | 0.0533 (8) | |
| N51 | 0.2997 (3) | 0.0977 (2) | 0.00037 (9) | 0.0405 (7) | 0.744 (5) |
| C61 | 0.1492 (4) | 0.1211 (16) | 0.0148 (2) | 0.0523 (10) | 0.744 (5) |
| C71 | 0.0470 (5) | 0.1870 (5) | −0.02434 (15) | 0.0460 (14) | 0.744 (5) |
| N81 | 0.0708 (4) | 0.1159 (3) | −0.06569 (12) | 0.0435 (12) | 0.744 (5) |
| N52 | 0.2997 (3) | 0.0977 (2) | 0.00037 (9) | 0.0405 (7) | 0.256 (5) |
| C62 | 0.1520 (9) | 0.120 (5) | 0.0190 (6) | 0.0523 (10) | 0.256 (5) |
| C72 | 0.0533 (12) | 0.1161 (14) | −0.0195 (5) | 0.040* | 0.256 (5) |
| N82 | −0.0905 (12) | 0.1385 (11) | 0.0032 (4) | 0.070 (4)* | 0.256 (5) |
| H11 | 0.1359 | 0.3842 | 0.4695 | 0.039* | |
| H12 | 0.1159 | 0.5172 | 0.3966 | 0.044* | |
| H112 | 0.3906 | 0.1336 | 0.3551 | 0.031* | |
| H113 | 0.6335 | 0.1701 | 0.3458 | 0.035* | |
| H114 | 0.6323 | 0.3258 | 0.4019 | 0.037* | |
| H115 | 0.3877 | 0.3862 | 0.4446 | 0.032* | |
| H122 | 0.1525 | 0.4381 | 0.3041 | 0.034* | |
| H123 | 0.3688 | 0.3223 | 0.2569 | 0.039* | |
| H124 | 0.5741 | 0.4020 | 0.2682 | 0.038* | |
| H125 | 0.4871 | 0.5678 | 0.3235 | 0.031* | |
| H132 | −0.0232 | 0.1956 | 0.4807 | 0.038* | |
| H133 | −0.0303 | 0.0712 | 0.5530 | 0.046* | |
| H134 | 0.1816 | −0.0311 | 0.5901 | 0.045* | |
| H135 | 0.3947 | −0.0010 | 0.5559 | 0.048* | |
| H136 | 0.4012 | 0.1242 | 0.4830 | 0.038* | |
| H142 | −0.0301 | 0.4028 | 0.3743 | 0.034* | |
| H143 | −0.1548 | 0.3587 | 0.3151 | 0.041* | |
| H144 | −0.0983 | 0.1651 | 0.2928 | 0.042* | |
| H145 | 0.0764 | 0.0128 | 0.3332 | 0.039* | |
| H146 | 0.1947 | 0.0560 | 0.3938 | 0.032* | |
| H152 | 0.2201 | 0.6914 | 0.2558 | 0.039* | |
| H153 | 0.0831 | 0.8415 | 0.2017 | 0.049* | |
| H154 | −0.1295 | 0.9690 | 0.2303 | 0.049* | |
| H155 | −0.2032 | 0.9463 | 0.3124 | 0.047* | |
| H156 | −0.0635 | 0.8001 | 0.3669 | 0.039* | |
| H162 | 0.3334 | 0.7893 | 0.3308 | 0.037* | |
| H163 | 0.4337 | 0.8846 | 0.3808 | 0.047* | |
| H164 | 0.4104 | 0.8452 | 0.4655 | 0.050* | |
| H165 | 0.2962 | 0.7023 | 0.5001 | 0.052* | |
| H166 | 0.1941 | 0.6065 | 0.4508 | 0.041* | |
| H21 | 0.4326 | 0.1013 | 0.0453 | 0.038* | |
| H22 | 0.5708 | −0.0515 | 0.1074 | 0.041* | |
| H212 | 0.3346 | 0.3102 | 0.1821 | 0.031* | |
| H213 | 0.1456 | 0.2293 | 0.2317 | 0.038* | |
| H214 | 0.1050 | 0.0657 | 0.1883 | 0.037* | |
| H215 | 0.2682 | 0.0464 | 0.1114 | 0.033* | |
| H222 | 0.6648 | 0.0161 | 0.1844 | 0.032* | |
| H223 | 0.5428 | 0.1109 | 0.2596 | 0.034* | |
| H224 | 0.3536 | 0.0097 | 0.2932 | 0.036* | |
| H225 | 0.3562 | −0.1482 | 0.2379 | 0.032* | |
| H232 | 0.5382 | 0.2937 | 0.0035 | 0.043* | |
| H233 | 0.4319 | 0.4471 | −0.0549 | 0.053* | |
| H234 | 0.2014 | 0.5650 | −0.0395 | 0.059* | |
| H235 | 0.0756 | 0.5236 | 0.0329 | 0.063* | |
| H236 | 0.1787 | 0.3655 | 0.0907 | 0.051* | |
| H242 | 0.4710 | 0.3985 | 0.1282 | 0.037* | |
| H243 | 0.6634 | 0.4344 | 0.1594 | 0.045* | |
| H244 | 0.8884 | 0.2963 | 0.1539 | 0.051* | |
| H245 | 0.9162 | 0.1247 | 0.1159 | 0.047* | |
| H246 | 0.7221 | 0.0836 | 0.0875 | 0.038* | |
| H252 | 0.7688 | −0.3341 | 0.1060 | 0.038* | |
| H253 | 0.9676 | −0.4885 | 0.1281 | 0.046* | |
| H254 | 1.0284 | −0.5315 | 0.2092 | 0.051* | |
| H255 | 0.8860 | −0.4196 | 0.2686 | 0.052* | |
| H256 | 0.6884 | −0.2634 | 0.2463 | 0.043* | |
| H262 | 0.4206 | −0.1344 | 0.0786 | 0.040* | |
| H263 | 0.2496 | −0.2198 | 0.0594 | 0.053* | |
| H264 | 0.1857 | −0.3694 | 0.1135 | 0.055* | |
| H265 | 0.3019 | −0.4379 | 0.1863 | 0.047* | |
| H266 | 0.4761 | −0.3551 | 0.2047 | 0.038* | |
| H1A | 0.1040 | 0.4960 | 0.5355 | 0.047* | |
| H1B | 0.1162 | 0.3684 | 0.5536 | 0.047* | |
| H2A | 0.3459 | 0.4511 | 0.5192 | 0.045* | |
| H2B | 0.3547 | 0.3174 | 0.5426 | 0.045* | |
| H3A | 0.4233 | 0.4254 | 0.5969 | 0.043* | |
| H3B | 0.2716 | 0.5213 | 0.5955 | 0.043* | |
| H4B | 0.3183 | 0.2890 | 0.6371 | 0.080* | |
| H4A | 0.1762 | 0.3771 | 0.6352 | 0.080* | |
| H51B | 0.3155 | 0.1626 | −0.0188 | 0.061* | |
| H51A | 0.3276 | 0.0327 | −0.0166 | 0.061* | |
| H61A | 0.1288 | 0.1685 | 0.0430 | 0.063* | 0.744 (5) |
| H61B | 0.1290 | 0.0434 | 0.0262 | 0.063* | 0.744 (5) |
| H71A | −0.0520 | 0.1995 | −0.0107 | 0.055* | 0.744 (5) |
| H71B | 0.0627 | 0.2663 | −0.0355 | 0.055* | 0.744 (5) |
| H81A | 0.0096 | 0.1548 | −0.0890 | 0.065* | 0.744 (5) |
| H81B | 0.0559 | 0.0432 | −0.0553 | 0.065* | 0.744 (5) |
| H62A | 0.1208 | 0.1998 | 0.0312 | 0.063* | 0.256 (5) |
| H62B | 0.1448 | 0.0594 | 0.0469 | 0.063* | 0.256 (5) |
| H72A | 0.0572 | 0.1778 | −0.0473 | 0.048* | 0.256 (5) |
| H72B | 0.0825 | 0.0366 | −0.0318 | 0.048* | 0.256 (5) |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Fe1 | 0.0247 (2) | 0.0207 (2) | 0.0224 (2) | −0.00531 (17) | −0.00219 (17) | −0.00097 (16) |
| O11 | 0.0320 (11) | 0.0202 (9) | 0.0250 (11) | −0.0038 (8) | −0.0027 (9) | −0.0046 (8) |
| O12 | 0.0313 (11) | 0.0211 (10) | 0.0336 (12) | −0.0067 (9) | 0.0018 (9) | 0.0013 (8) |
| C11 | 0.0236 (15) | 0.0201 (13) | 0.0248 (15) | −0.0046 (11) | −0.0003 (12) | −0.0043 (11) |
| C12 | 0.0245 (15) | 0.0220 (13) | 0.0267 (15) | −0.0073 (12) | −0.0021 (12) | 0.0009 (11) |
| C111 | 0.0238 (15) | 0.0199 (13) | 0.0230 (15) | −0.0038 (11) | −0.0016 (12) | 0.0004 (11) |
| C112 | 0.0270 (16) | 0.0206 (14) | 0.0277 (16) | −0.0041 (12) | −0.0026 (13) | −0.0010 (12) |
| C113 | 0.0264 (16) | 0.0237 (14) | 0.0348 (17) | −0.0047 (12) | −0.0006 (13) | 0.0040 (12) |
| C114 | 0.0294 (17) | 0.0352 (16) | 0.0290 (16) | −0.0136 (13) | −0.0080 (13) | 0.0074 (13) |
| C115 | 0.0285 (16) | 0.0293 (15) | 0.0227 (15) | −0.0106 (13) | −0.0037 (12) | −0.0001 (12) |
| C121 | 0.0276 (16) | 0.0203 (13) | 0.0219 (15) | −0.0041 (12) | −0.0042 (12) | 0.0004 (11) |
| C122 | 0.0297 (16) | 0.0263 (15) | 0.0272 (16) | −0.0052 (13) | −0.0061 (13) | 0.0004 (12) |
| C123 | 0.0447 (19) | 0.0230 (14) | 0.0243 (16) | −0.0007 (14) | −0.0066 (14) | −0.0032 (12) |
| C124 | 0.0311 (17) | 0.0305 (16) | 0.0251 (16) | 0.0010 (13) | 0.0052 (13) | 0.0041 (12) |
| C125 | 0.0294 (16) | 0.0247 (14) | 0.0230 (15) | −0.0090 (12) | −0.0006 (12) | 0.0009 (11) |
| C131 | 0.0280 (16) | 0.0210 (13) | 0.0222 (15) | −0.0057 (12) | −0.0009 (12) | −0.0037 (11) |
| C132 | 0.0266 (16) | 0.0348 (16) | 0.0312 (17) | −0.0063 (13) | 0.0001 (13) | 0.0016 (13) |
| C133 | 0.0329 (18) | 0.0414 (18) | 0.0382 (19) | −0.0138 (15) | 0.0034 (14) | 0.0053 (14) |
| C134 | 0.046 (2) | 0.0301 (16) | 0.0286 (17) | −0.0037 (15) | 0.0030 (15) | 0.0059 (13) |
| C135 | 0.0358 (19) | 0.0408 (18) | 0.0314 (18) | 0.0018 (15) | −0.0005 (15) | 0.0070 (14) |
| C136 | 0.0250 (16) | 0.0358 (16) | 0.0323 (17) | −0.0068 (13) | 0.0006 (13) | −0.0017 (13) |
| C141 | 0.0241 (15) | 0.0228 (14) | 0.0241 (15) | −0.0077 (12) | 0.0017 (12) | −0.0029 (11) |
| C142 | 0.0253 (15) | 0.0281 (15) | 0.0325 (17) | −0.0072 (12) | −0.0009 (13) | −0.0059 (12) |
| C143 | 0.0298 (17) | 0.0377 (17) | 0.0362 (18) | −0.0118 (14) | −0.0055 (14) | 0.0022 (14) |
| C144 | 0.0329 (17) | 0.0451 (18) | 0.0327 (17) | −0.0189 (15) | −0.0007 (14) | −0.0088 (14) |
| C145 | 0.0358 (18) | 0.0325 (16) | 0.0347 (17) | −0.0173 (14) | 0.0059 (14) | −0.0114 (13) |
| C146 | 0.0243 (15) | 0.0220 (14) | 0.0316 (16) | −0.0062 (12) | 0.0008 (13) | −0.0011 (12) |
| C151 | 0.0284 (16) | 0.0183 (13) | 0.0312 (16) | −0.0070 (12) | −0.0073 (13) | 0.0006 (12) |
| C152 | 0.0351 (18) | 0.0300 (15) | 0.0314 (17) | −0.0104 (14) | −0.0026 (14) | 0.0023 (13) |
| C153 | 0.044 (2) | 0.0417 (18) | 0.0360 (19) | −0.0150 (16) | −0.0123 (16) | 0.0099 (14) |
| C154 | 0.0374 (19) | 0.0318 (17) | 0.055 (2) | −0.0103 (15) | −0.0253 (18) | 0.0089 (15) |
| C155 | 0.0258 (17) | 0.0267 (16) | 0.064 (2) | −0.0043 (13) | −0.0135 (16) | −0.0025 (15) |
| C156 | 0.0315 (17) | 0.0255 (15) | 0.0394 (18) | −0.0075 (13) | −0.0021 (14) | −0.0021 (13) |
| C161 | 0.0246 (15) | 0.0220 (14) | 0.0305 (16) | 0.0023 (12) | −0.0054 (13) | −0.0060 (12) |
| C162 | 0.0348 (17) | 0.0233 (14) | 0.0360 (18) | −0.0094 (13) | −0.0099 (14) | 0.0003 (13) |
| C163 | 0.0374 (19) | 0.0236 (15) | 0.058 (2) | −0.0058 (14) | −0.0171 (17) | −0.0028 (15) |
| C164 | 0.0306 (18) | 0.0359 (18) | 0.057 (2) | 0.0030 (15) | −0.0163 (17) | −0.0198 (16) |
| C165 | 0.041 (2) | 0.056 (2) | 0.0324 (19) | −0.0023 (17) | −0.0070 (16) | −0.0190 (16) |
| C166 | 0.0300 (17) | 0.0379 (17) | 0.0304 (17) | −0.0031 (14) | −0.0023 (13) | −0.0040 (13) |
| Fe2 | 0.0233 (2) | 0.0227 (2) | 0.0225 (2) | −0.00375 (17) | −0.00159 (17) | −0.00177 (16) |
| O21 | 0.0287 (11) | 0.0230 (9) | 0.0230 (10) | −0.0026 (8) | −0.0027 (8) | −0.0062 (8) |
| O22 | 0.0289 (11) | 0.0237 (10) | 0.0238 (10) | −0.0021 (8) | 0.0013 (9) | 0.0010 (8) |
| C21 | 0.0264 (15) | 0.0215 (13) | 0.0238 (15) | −0.0017 (12) | −0.0042 (12) | −0.0036 (11) |
| C22 | 0.0252 (15) | 0.0228 (14) | 0.0231 (15) | −0.0052 (12) | 0.0004 (12) | −0.0012 (11) |
| C211 | 0.0221 (15) | 0.0225 (14) | 0.0260 (15) | −0.0003 (12) | −0.0064 (12) | −0.0028 (11) |
| C212 | 0.0279 (16) | 0.0234 (14) | 0.0233 (15) | −0.0026 (12) | −0.0023 (12) | −0.0019 (11) |
| C213 | 0.0269 (16) | 0.0300 (15) | 0.0289 (16) | 0.0021 (13) | 0.0023 (13) | 0.0003 (13) |
| C214 | 0.0227 (16) | 0.0334 (16) | 0.0342 (18) | −0.0062 (13) | −0.0016 (13) | 0.0042 (13) |
| C215 | 0.0234 (15) | 0.0305 (15) | 0.0263 (16) | −0.0050 (12) | −0.0063 (12) | 0.0000 (12) |
| C221 | 0.0252 (15) | 0.0212 (13) | 0.0239 (15) | −0.0016 (12) | −0.0058 (12) | −0.0008 (11) |
| C222 | 0.0265 (16) | 0.0264 (14) | 0.0260 (16) | −0.0055 (12) | −0.0055 (12) | 0.0007 (12) |
| C223 | 0.0302 (16) | 0.0270 (15) | 0.0281 (16) | −0.0027 (13) | −0.0104 (13) | −0.0057 (12) |
| C224 | 0.0351 (18) | 0.0289 (15) | 0.0212 (15) | −0.0018 (13) | −0.0042 (13) | 0.0009 (12) |
| C225 | 0.0297 (16) | 0.0250 (14) | 0.0230 (15) | −0.0076 (12) | −0.0030 (12) | 0.0027 (12) |
| C231 | 0.0344 (17) | 0.0240 (14) | 0.0228 (15) | −0.0071 (13) | −0.0061 (13) | −0.0023 (12) |
| C232 | 0.0340 (18) | 0.0382 (17) | 0.0331 (18) | −0.0079 (14) | −0.0032 (14) | 0.0019 (14) |
| C233 | 0.058 (2) | 0.0393 (18) | 0.0306 (18) | −0.0120 (17) | −0.0072 (16) | 0.0081 (14) |
| C234 | 0.072 (3) | 0.0334 (18) | 0.033 (2) | −0.0020 (18) | −0.0179 (19) | 0.0073 (15) |
| C235 | 0.049 (2) | 0.050 (2) | 0.039 (2) | 0.0146 (18) | −0.0098 (17) | 0.0000 (16) |
| C236 | 0.047 (2) | 0.0439 (19) | 0.0239 (16) | 0.0071 (16) | −0.0033 (15) | 0.0000 (14) |
| C241 | 0.0250 (15) | 0.0250 (14) | 0.0246 (15) | −0.0070 (12) | −0.0024 (12) | 0.0006 (11) |
| C242 | 0.0309 (17) | 0.0274 (15) | 0.0330 (17) | −0.0073 (13) | −0.0049 (14) | −0.0001 (13) |
| C243 | 0.043 (2) | 0.0333 (17) | 0.0397 (19) | −0.0140 (15) | −0.0078 (16) | −0.0028 (14) |
| C244 | 0.0380 (19) | 0.0422 (19) | 0.052 (2) | −0.0173 (16) | −0.0154 (17) | 0.0012 (16) |
| C245 | 0.0234 (17) | 0.0384 (18) | 0.053 (2) | −0.0077 (14) | −0.0052 (15) | −0.0006 (16) |
| C246 | 0.0268 (16) | 0.0290 (15) | 0.0387 (18) | −0.0079 (13) | 0.0001 (14) | −0.0055 (13) |
| C251 | 0.0235 (15) | 0.0217 (14) | 0.0329 (16) | −0.0061 (12) | −0.0041 (13) | −0.0006 (12) |
| C252 | 0.0294 (17) | 0.0282 (15) | 0.0364 (18) | −0.0051 (13) | 0.0009 (14) | −0.0040 (13) |
| C253 | 0.0335 (18) | 0.0250 (15) | 0.052 (2) | −0.0026 (14) | 0.0064 (15) | −0.0082 (14) |
| C254 | 0.0345 (19) | 0.0259 (16) | 0.060 (2) | 0.0024 (14) | −0.0123 (17) | 0.0020 (15) |
| C255 | 0.046 (2) | 0.0357 (18) | 0.045 (2) | −0.0033 (16) | −0.0171 (17) | −0.0012 (15) |
| C256 | 0.0402 (19) | 0.0315 (16) | 0.0319 (17) | −0.0030 (14) | −0.0063 (15) | −0.0051 (13) |
| C261 | 0.0215 (15) | 0.0207 (13) | 0.0286 (16) | −0.0015 (12) | 0.0016 (12) | −0.0060 (12) |
| C262 | 0.0322 (17) | 0.0344 (16) | 0.0323 (17) | −0.0067 (14) | −0.0043 (14) | −0.0060 (13) |
| C263 | 0.039 (2) | 0.048 (2) | 0.047 (2) | −0.0107 (16) | −0.0160 (17) | −0.0109 (17) |
| C264 | 0.0334 (19) | 0.047 (2) | 0.063 (2) | −0.0142 (16) | −0.0018 (17) | −0.0199 (18) |
| C265 | 0.042 (2) | 0.0335 (17) | 0.044 (2) | −0.0140 (15) | 0.0047 (16) | −0.0113 (15) |
| C266 | 0.0368 (18) | 0.0269 (15) | 0.0323 (17) | −0.0076 (14) | −0.0008 (14) | −0.0066 (13) |
| N1 | 0.0291 (14) | 0.0323 (13) | 0.0321 (14) | −0.0053 (11) | −0.0003 (11) | −0.0080 (11) |
| C2 | 0.0307 (18) | 0.0469 (18) | 0.0324 (18) | −0.0053 (15) | −0.0020 (14) | −0.0088 (14) |
| C3 | 0.0301 (17) | 0.0407 (17) | 0.0355 (18) | −0.0071 (14) | −0.0012 (14) | −0.0104 (14) |
| N4 | 0.056 (2) | 0.069 (2) | 0.0363 (17) | −0.0219 (17) | −0.0049 (15) | 0.0027 (15) |
| N51 | 0.0376 (16) | 0.0522 (16) | 0.0322 (15) | −0.0103 (13) | −0.0037 (12) | −0.0103 (13) |
| C61 | 0.044 (2) | 0.081 (3) | 0.032 (2) | −0.016 (2) | −0.0002 (17) | −0.015 (2) |
| C71 | 0.040 (3) | 0.052 (3) | 0.040 (3) | 0.001 (2) | −0.011 (2) | −0.010 (2) |
| N81 | 0.047 (2) | 0.045 (2) | 0.038 (2) | −0.0083 (18) | −0.0078 (18) | −0.0097 (17) |
| N52 | 0.0376 (16) | 0.0522 (16) | 0.0322 (15) | −0.0103 (13) | −0.0037 (12) | −0.0103 (13) |
| C62 | 0.044 (2) | 0.081 (3) | 0.032 (2) | −0.016 (2) | −0.0002 (17) | −0.015 (2) |
Geometric parameters (Å, º) top
| Fe1—C123 | 2.032 (3) | C21—C211 | 1.519 (4) |
| Fe1—C124 | 2.041 (3) | C21—C241 | 1.532 (4) |
| Fe1—C113 | 2.045 (2) | C21—C231 | 1.544 (3) |
| Fe1—C112 | 2.046 (3) | C22—C221 | 1.524 (4) |
| Fe1—C115 | 2.047 (3) | C22—C261 | 1.532 (4) |
| Fe1—C122 | 2.051 (2) | C22—C251 | 1.544 (3) |
| Fe1—C114 | 2.052 (3) | C211—C215 | 1.427 (4) |
| Fe1—C125 | 2.057 (3) | C211—C212 | 1.428 (4) |
| Fe1—C111 | 2.069 (3) | C212—C213 | 1.423 (4) |
| Fe1—C121 | 2.073 (2) | C212—H212 | 0.95 |
| O11—C11 | 1.436 (3) | C213—C214 | 1.415 (4) |
| O11—H11 | 0.84 | C213—H213 | 0.95 |
| O12—C12 | 1.425 (3) | C214—C215 | 1.425 (4) |
| O12—H12 | 0.84 | C214—H214 | 0.95 |
| C11—C111 | 1.522 (4) | C215—H215 | 0.95 |
| C11—C141 | 1.536 (4) | C221—C222 | 1.425 (4) |
| C11—C131 | 1.539 (4) | C221—C225 | 1.428 (4) |
| C12—C121 | 1.523 (4) | C222—C223 | 1.416 (4) |
| C12—C161 | 1.539 (4) | C222—H222 | 0.95 |
| C12—C151 | 1.543 (3) | C223—C224 | 1.412 (4) |
| C111—C112 | 1.426 (4) | C223—H223 | 0.95 |
| C111—C115 | 1.429 (4) | C224—C225 | 1.430 (4) |
| C112—C113 | 1.422 (4) | C224—H224 | 0.95 |
| C112—H112 | 0.95 | C225—H225 | 0.95 |
| C113—C114 | 1.424 (4) | C231—C232 | 1.386 (4) |
| C113—H113 | 0.95 | C231—C236 | 1.388 (4) |
| C114—C115 | 1.420 (4) | C232—C233 | 1.387 (4) |
| C114—H114 | 0.95 | C232—H232 | 0.95 |
| C115—H115 | 0.95 | C233—C234 | 1.378 (5) |
| C121—C125 | 1.424 (4) | C233—H233 | 0.95 |
| C121—C122 | 1.431 (4) | C234—C235 | 1.375 (5) |
| C122—C123 | 1.415 (4) | C234—H234 | 0.95 |
| C122—H122 | 0.95 | C235—C236 | 1.396 (4) |
| C123—C124 | 1.405 (4) | C235—H235 | 0.95 |
| C123—H123 | 0.95 | C236—H236 | 0.95 |
| C124—C125 | 1.424 (4) | C241—C246 | 1.392 (4) |
| C124—H124 | 0.95 | C241—C242 | 1.400 (4) |
| C125—H125 | 0.95 | C242—C243 | 1.381 (4) |
| C131—C136 | 1.387 (3) | C242—H242 | 0.95 |
| C131—C132 | 1.389 (4) | C243—C244 | 1.393 (4) |
| C132—C133 | 1.381 (4) | C243—H243 | 0.95 |
| C132—H132 | 0.95 | C244—C245 | 1.378 (4) |
| C133—C134 | 1.390 (4) | C244—H244 | 0.95 |
| C133—H133 | 0.95 | C245—C246 | 1.390 (4) |
| C134—C135 | 1.373 (5) | C245—H245 | 0.95 |
| C134—H134 | 0.95 | C246—H246 | 0.95 |
| C135—C136 | 1.396 (4) | C251—C252 | 1.386 (4) |
| C135—H135 | 0.95 | C251—C256 | 1.388 (4) |
| C136—H136 | 0.95 | C252—C253 | 1.383 (4) |
| C141—C142 | 1.387 (3) | C252—H252 | 0.95 |
| C141—C146 | 1.389 (4) | C253—C254 | 1.382 (4) |
| C142—C143 | 1.390 (4) | C253—H253 | 0.95 |
| C142—H142 | 0.95 | C254—C255 | 1.386 (5) |
| C143—C144 | 1.378 (4) | C254—H254 | 0.95 |
| C143—H143 | 0.95 | C255—C256 | 1.386 (4) |
| C144—C145 | 1.388 (4) | C255—H255 | 0.95 |
| C144—H144 | 0.95 | C256—H256 | 0.95 |
| C145—C146 | 1.377 (4) | C261—C266 | 1.386 (4) |
| C145—H145 | 0.95 | C261—C262 | 1.401 (4) |
| C146—H146 | 0.95 | C262—C263 | 1.383 (4) |
| C151—C152 | 1.363 (4) | C262—H262 | 0.95 |
| C151—C156 | 1.402 (4) | C263—C264 | 1.385 (5) |
| C152—C153 | 1.400 (4) | C263—H263 | 0.95 |
| C152—H152 | 0.95 | C264—C265 | 1.392 (4) |
| C153—C154 | 1.384 (4) | C264—H264 | 0.95 |
| C153—H153 | 0.95 | C265—C266 | 1.383 (4) |
| C154—C155 | 1.374 (5) | C265—H265 | 0.95 |
| C154—H154 | 0.95 | C266—H266 | 0.95 |
| C155—C156 | 1.394 (4) | N1—C2 | 1.468 (3) |
| C155—H155 | 0.95 | N1—H1A | 0.91 |
| C156—H156 | 0.95 | N1—H1B | 0.91 |
| C161—C162 | 1.390 (4) | C2—C3 | 1.520 (4) |
| C161—C166 | 1.398 (4) | C2—H2A | 0.99 |
| C162—C163 | 1.389 (4) | C2—H2B | 0.99 |
| C162—H162 | 0.95 | C3—N4 | 1.455 (3) |
| C163—C164 | 1.381 (5) | C3—H3A | 0.99 |
| C163—H163 | 0.95 | C3—H3B | 0.99 |
| C164—C165 | 1.379 (5) | N4—H4B | 0.91 |
| C164—H164 | 0.95 | N4—H4A | 0.91 |
| C165—C166 | 1.391 (4) | N51—C61 | 1.456 (4) |
| C165—H165 | 0.95 | N51—H51B | 0.91 |
| C166—H166 | 0.95 | N51—H51A | 0.91 |
| Fe2—C225 | 2.041 (3) | C61—C71 | 1.526 (7) |
| Fe2—C214 | 2.041 (3) | C61—H61A | 0.99 |
| Fe2—C224 | 2.042 (3) | C61—H61B | 0.99 |
| Fe2—C213 | 2.044 (3) | C71—N81 | 1.466 (4) |
| Fe2—C215 | 2.044 (2) | C71—H71A | 0.99 |
| Fe2—C223 | 2.045 (3) | C71—H71B | 0.99 |
| Fe2—C212 | 2.048 (2) | N81—H81A | 0.91 |
| Fe2—C222 | 2.053 (3) | N81—H81B | 0.91 |
| Fe2—C221 | 2.058 (2) | C62—C72 | 1.524 (7) |
| Fe2—C211 | 2.059 (3) | C62—H62A | 0.99 |
| O21—C21 | 1.439 (3) | C62—H62B | 0.99 |
| O21—H21 | 0.84 | C72—N82 | 1.461 (6) |
| O22—C22 | 1.431 (3) | C72—H72A | 0.99 |
| O22—H22 | 0.84 | C72—H72B | 0.99 |
| | | |
| C11—O11—H11 | 109.5 | C214—C213—H213 | 126.0 |
| C12—O12—H12 | 109.5 | C212—C213—H213 | 126.0 |
| O11—C11—C111 | 109.0 (2) | C213—C214—C215 | 107.7 (3) |
| O11—C11—C141 | 107.93 (19) | C213—C214—H214 | 126.1 |
| C111—C11—C141 | 110.7 (2) | C215—C214—H214 | 126.1 |
| O11—C11—C131 | 107.3 (2) | C214—C215—C211 | 108.8 (3) |
| C111—C11—C131 | 111.6 (2) | C214—C215—H215 | 125.6 |
| C141—C11—C131 | 110.2 (2) | C211—C215—H215 | 125.6 |
| O12—C12—C121 | 110.2 (2) | C222—C221—C225 | 107.2 (2) |
| O12—C12—C161 | 110.9 (2) | C222—C221—C22 | 123.8 (3) |
| C121—C12—C161 | 112.2 (2) | C225—C221—C22 | 128.7 (3) |
| O12—C12—C151 | 104.3 (2) | C223—C222—C221 | 108.5 (3) |
| C121—C12—C151 | 109.8 (2) | C223—C222—H222 | 125.7 |
| C161—C12—C151 | 109.2 (2) | C221—C222—H222 | 125.7 |
| C112—C111—C115 | 106.6 (2) | C224—C223—C222 | 108.4 (3) |
| C112—C111—C11 | 128.1 (2) | C224—C223—H223 | 125.8 |
| C115—C111—C11 | 125.0 (2) | C222—C223—H223 | 125.8 |
| C113—C112—C111 | 109.0 (2) | C223—C224—C225 | 107.8 (3) |
| C113—C112—H112 | 125.5 | C223—C224—H224 | 126.1 |
| C111—C112—H112 | 125.5 | C225—C224—H224 | 126.1 |
| C112—C113—C114 | 107.7 (3) | C221—C225—C224 | 108.1 (3) |
| C112—C113—H113 | 126.1 | C221—C225—H225 | 126.0 |
| C114—C113—H113 | 126.1 | C224—C225—H225 | 126.0 |
| C115—C114—C113 | 107.7 (3) | C232—C231—C236 | 118.6 (2) |
| C115—C114—H114 | 126.1 | C232—C231—C21 | 118.6 (2) |
| C113—C114—H114 | 126.1 | C236—C231—C21 | 122.8 (3) |
| C114—C115—C111 | 109.0 (3) | C231—C232—C233 | 121.2 (3) |
| C114—C115—H115 | 125.5 | C231—C232—H232 | 119.4 |
| C111—C115—H115 | 125.5 | C233—C232—H232 | 119.4 |
| C125—C121—C122 | 107.1 (2) | C234—C233—C232 | 120.0 (3) |
| C125—C121—C12 | 129.4 (2) | C234—C233—H233 | 120.0 |
| C122—C121—C12 | 123.1 (2) | C232—C233—H233 | 120.0 |
| C123—C122—C121 | 108.2 (3) | C235—C234—C233 | 119.4 (3) |
| C123—C122—H122 | 125.9 | C235—C234—H234 | 120.3 |
| C121—C122—H122 | 125.9 | C233—C234—H234 | 120.3 |
| C124—C123—C122 | 108.5 (2) | C234—C235—C236 | 120.9 (3) |
| C124—C123—H123 | 125.8 | C234—C235—H235 | 119.6 |
| C122—C123—H123 | 125.8 | C236—C235—H235 | 119.6 |
| C123—C124—C125 | 108.1 (3) | C231—C236—C235 | 119.9 (3) |
| C123—C124—H124 | 125.9 | C231—C236—H236 | 120.1 |
| C125—C124—H124 | 125.9 | C235—C236—H236 | 120.1 |
| C121—C125—C124 | 108.2 (3) | C246—C241—C242 | 118.1 (3) |
| C121—C125—H125 | 125.9 | C246—C241—C21 | 121.6 (2) |
| C124—C125—H125 | 125.9 | C242—C241—C21 | 120.4 (2) |
| C136—C131—C132 | 117.8 (3) | C243—C242—C241 | 121.1 (3) |
| C136—C131—C11 | 123.2 (3) | C243—C242—H242 | 119.4 |
| C132—C131—C11 | 118.7 (2) | C241—C242—H242 | 119.4 |
| C133—C132—C131 | 122.1 (3) | C242—C243—C244 | 120.3 (3) |
| C133—C132—H132 | 118.9 | C242—C243—H243 | 119.8 |
| C131—C132—H132 | 118.9 | C244—C243—H243 | 119.8 |
| C132—C133—C134 | 119.2 (3) | C245—C244—C243 | 118.8 (3) |
| C132—C133—H133 | 120.4 | C245—C244—H244 | 120.6 |
| C134—C133—H133 | 120.4 | C243—C244—H244 | 120.6 |
| C135—C134—C133 | 119.7 (3) | C244—C245—C246 | 121.2 (3) |
| C135—C134—H134 | 120.2 | C244—C245—H245 | 119.4 |
| C133—C134—H134 | 120.2 | C246—C245—H245 | 119.4 |
| C134—C135—C136 | 120.6 (3) | C245—C246—C241 | 120.4 (3) |
| C134—C135—H135 | 119.7 | C245—C246—H246 | 119.8 |
| C136—C135—H135 | 119.7 | C241—C246—H246 | 119.8 |
| C131—C136—C135 | 120.5 (3) | C252—C251—C256 | 118.7 (2) |
| C131—C136—H136 | 119.7 | C252—C251—C22 | 118.3 (2) |
| C135—C136—H136 | 119.7 | C256—C251—C22 | 123.0 (2) |
| C142—C141—C146 | 117.9 (2) | C253—C252—C251 | 120.5 (3) |
| C142—C141—C11 | 121.9 (2) | C253—C252—H252 | 119.7 |
| C146—C141—C11 | 120.2 (2) | C251—C252—H252 | 119.7 |
| C141—C142—C143 | 120.9 (3) | C254—C253—C252 | 120.6 (3) |
| C141—C142—H142 | 119.6 | C254—C253—H253 | 119.7 |
| C143—C142—H142 | 119.6 | C252—C253—H253 | 119.7 |
| C144—C143—C142 | 120.3 (3) | C253—C254—C255 | 119.3 (3) |
| C144—C143—H143 | 119.8 | C253—C254—H254 | 120.3 |
| C142—C143—H143 | 119.8 | C255—C254—H254 | 120.3 |
| C143—C144—C145 | 119.3 (3) | C256—C255—C254 | 120.0 (3) |
| C143—C144—H144 | 120.3 | C256—C255—H255 | 120.0 |
| C145—C144—H144 | 120.3 | C254—C255—H255 | 120.0 |
| C146—C145—C144 | 120.0 (3) | C255—C256—C251 | 120.9 (3) |
| C146—C145—H145 | 120.0 | C255—C256—H256 | 119.6 |
| C144—C145—H145 | 120.0 | C251—C256—H256 | 119.6 |
| C145—C146—C141 | 121.6 (2) | C266—C261—C262 | 118.0 (3) |
| C145—C146—H146 | 119.2 | C266—C261—C22 | 121.1 (2) |
| C141—C146—H146 | 119.2 | C262—C261—C22 | 120.9 (3) |
| C152—C151—C156 | 119.1 (2) | C263—C262—C261 | 120.7 (3) |
| C152—C151—C12 | 122.8 (3) | C263—C262—H262 | 119.6 |
| C156—C151—C12 | 118.1 (3) | C261—C262—H262 | 119.6 |
| C151—C152—C153 | 121.1 (3) | C262—C263—C264 | 120.6 (3) |
| C151—C152—H152 | 119.5 | C262—C263—H263 | 119.7 |
| C153—C152—H152 | 119.5 | C264—C263—H263 | 119.7 |
| C154—C153—C152 | 119.8 (3) | C263—C264—C265 | 119.2 (3) |
| C154—C153—H153 | 120.1 | C263—C264—H264 | 120.4 |
| C152—C153—H153 | 120.1 | C265—C264—H264 | 120.4 |
| C155—C154—C153 | 119.6 (3) | C266—C265—C264 | 120.0 (3) |
| C155—C154—H154 | 120.2 | C266—C265—H265 | 120.0 |
| C153—C154—H154 | 120.2 | C264—C265—H265 | 120.0 |
| C154—C155—C156 | 120.7 (3) | C265—C266—C261 | 121.5 (3) |
| C154—C155—H155 | 119.7 | C265—C266—H266 | 119.2 |
| C156—C155—H155 | 119.7 | C261—C266—H266 | 119.2 |
| C155—C156—C151 | 119.8 (3) | C2—N1—H1A | 109.5 |
| C155—C156—H156 | 120.1 | C2—N1—H1B | 109.5 |
| C151—C156—H156 | 120.1 | H1A—N1—H1B | 109.5 |
| C162—C161—C166 | 118.6 (3) | N1—C2—C3 | 116.0 (2) |
| C162—C161—C12 | 120.7 (3) | N1—C2—H2A | 108.3 |
| C166—C161—C12 | 120.6 (3) | C3—C2—H2A | 108.3 |
| C163—C162—C161 | 120.5 (3) | N1—C2—H2B | 108.3 |
| C163—C162—H162 | 119.7 | C3—C2—H2B | 108.3 |
| C161—C162—H162 | 119.7 | H2A—C2—H2B | 107.4 |
| C164—C163—C162 | 120.6 (3) | N4—C3—C2 | 116.7 (3) |
| C164—C163—H163 | 119.7 | N4—C3—H3A | 108.1 |
| C162—C163—H163 | 119.7 | C2—C3—H3A | 108.1 |
| C165—C164—C163 | 119.3 (3) | N4—C3—H3B | 108.1 |
| C165—C164—H164 | 120.3 | C2—C3—H3B | 108.1 |
| C163—C164—H164 | 120.3 | H3A—C3—H3B | 107.3 |
| C164—C165—C166 | 120.7 (3) | C3—N4—H4B | 109.5 |
| C164—C165—H165 | 119.6 | C3—N4—H4A | 109.5 |
| C166—C165—H165 | 119.6 | H4B—N4—H4A | 109.5 |
| C165—C166—C161 | 120.1 (3) | N51—C61—C71 | 116.3 (6) |
| C165—C166—H166 | 119.9 | N51—C61—H61A | 108.2 |
| C161—C166—H166 | 119.9 | C71—C61—H61A | 108.2 |
| C21—O21—H21 | 109.5 | N51—C61—H61B | 108.2 |
| C22—O22—H22 | 109.5 | C71—C61—H61B | 108.2 |
| O21—C21—C211 | 108.5 (2) | H61A—C61—H61B | 107.4 |
| O21—C21—C241 | 107.7 (2) | N81—C71—C61 | 109.3 (6) |
| C211—C21—C241 | 112.2 (2) | N81—C71—H71A | 109.8 |
| O21—C21—C231 | 108.6 (2) | C61—C71—H71A | 109.8 |
| C211—C21—C231 | 111.1 (2) | N81—C71—H71B | 109.8 |
| C241—C21—C231 | 108.7 (2) | C61—C71—H71B | 109.8 |
| O22—C22—C221 | 109.4 (2) | H71A—C71—H71B | 108.3 |
| O22—C22—C261 | 110.8 (2) | C71—N81—H81A | 109.5 |
| C221—C22—C261 | 112.4 (2) | C71—N81—H81B | 109.5 |
| O22—C22—C251 | 104.9 (2) | H81A—N81—H81B | 109.5 |
| C221—C22—C251 | 110.4 (2) | C72—C62—H62A | 109.0 |
| C261—C22—C251 | 108.8 (2) | C72—C62—H62B | 109.0 |
| C215—C211—C212 | 106.7 (3) | H62A—C62—H62B | 107.8 |
| C215—C211—C21 | 124.8 (2) | N82—C72—C62 | 108.0 (9) |
| C212—C211—C21 | 128.2 (3) | N82—C72—H72A | 110.1 |
| C213—C212—C211 | 108.7 (3) | C62—C72—H72A | 110.1 |
| C213—C212—H212 | 125.7 | N82—C72—H72B | 110.1 |
| C211—C212—H212 | 125.7 | C62—C72—H72B | 110.1 |
| C214—C213—C212 | 108.1 (3) | H72A—C72—H72B | 108.4 |
| | | |
| O11—C11—C111—C112 | −153.6 (2) | O21—C21—C211—C215 | −25.6 (3) |
| C131—C11—C111—C112 | 88.1 (3) | C241—C21—C211—C215 | −144.4 (2) |
| C141—C11—C111—C112 | −35.0 (3) | C231—C21—C211—C215 | 93.7 (3) |
| O11—C11—C111—C115 | 33.8 (3) | O21—C21—C211—C212 | 161.3 (2) |
| C141—C11—C111—C115 | 152.4 (2) | C231—C21—C211—C212 | −79.4 (3) |
| C131—C11—C111—C115 | −84.5 (3) | C241—C21—C211—C212 | 42.5 (3) |
| C115—C111—C112—C113 | 0.5 (3) | C215—C211—C212—C213 | 0.3 (3) |
| C11—C111—C112—C113 | −173.2 (2) | C21—C211—C212—C213 | 174.3 (2) |
| C111—C112—C113—C114 | 0.0 (3) | C211—C212—C213—C214 | −0.1 (3) |
| C112—C113—C114—C115 | −0.4 (3) | C212—C213—C214—C215 | −0.2 (3) |
| C113—C114—C115—C111 | 0.7 (3) | C213—C214—C215—C211 | 0.4 (3) |
| C112—C111—C115—C114 | −0.8 (3) | C212—C211—C215—C214 | −0.4 (3) |
| C11—C111—C115—C114 | 173.2 (2) | C21—C211—C215—C214 | −174.7 (2) |
| O12—C12—C121—C125 | −147.8 (3) | O22—C22—C221—C222 | −36.2 (3) |
| C161—C12—C121—C125 | −23.8 (4) | C251—C22—C221—C222 | 78.7 (3) |
| C151—C12—C121—C125 | 97.8 (3) | C261—C22—C221—C222 | −159.7 (2) |
| O12—C12—C121—C122 | 41.0 (3) | O22—C22—C221—C225 | 151.1 (2) |
| C151—C12—C121—C122 | −73.4 (3) | C261—C22—C221—C225 | 27.6 (3) |
| C161—C12—C121—C122 | 165.0 (2) | C251—C22—C221—C225 | −94.0 (3) |
| C125—C121—C122—C123 | 0.0 (3) | C225—C221—C222—C223 | 0.0 (3) |
| C12—C121—C122—C123 | 172.8 (2) | C22—C221—C222—C223 | −174.0 (2) |
| C121—C122—C123—C124 | −0.2 (3) | C221—C222—C223—C224 | 0.2 (3) |
| C122—C123—C124—C125 | 0.3 (3) | C222—C223—C224—C225 | −0.3 (3) |
| C122—C121—C125—C124 | 0.2 (3) | C222—C221—C225—C224 | −0.2 (3) |
| C12—C121—C125—C124 | −172.1 (2) | C22—C221—C225—C224 | 173.4 (2) |
| C123—C124—C125—C121 | −0.3 (3) | C223—C224—C225—C221 | 0.3 (3) |
| O11—C11—C131—C136 | −109.6 (3) | O21—C21—C231—C232 | −54.9 (3) |
| C111—C11—C131—C136 | 9.7 (4) | C211—C21—C231—C232 | −174.1 (3) |
| C141—C11—C131—C136 | 133.1 (3) | C241—C21—C231—C232 | 62.0 (3) |
| O11—C11—C131—C132 | 65.1 (3) | O21—C21—C231—C236 | 124.1 (3) |
| C111—C11—C131—C132 | −175.5 (2) | C211—C21—C231—C236 | 4.9 (4) |
| C141—C11—C131—C132 | −52.1 (3) | C241—C21—C231—C236 | −119.0 (3) |
| C136—C131—C132—C133 | −1.2 (4) | C236—C231—C232—C233 | −0.5 (5) |
| C11—C131—C132—C133 | −176.2 (3) | C21—C231—C232—C233 | 178.6 (3) |
| C131—C132—C133—C134 | 0.0 (5) | C231—C232—C233—C234 | 1.5 (5) |
| C132—C133—C134—C135 | 1.3 (5) | C232—C233—C234—C235 | −1.3 (5) |
| C133—C134—C135—C136 | −1.4 (5) | C233—C234—C235—C236 | 0.1 (6) |
| C132—C131—C136—C135 | 1.1 (4) | C232—C231—C236—C235 | −0.6 (5) |
| C11—C131—C136—C135 | 175.9 (3) | C21—C231—C236—C235 | −179.7 (3) |
| C134—C135—C136—C131 | 0.1 (5) | C234—C235—C236—C231 | 0.8 (5) |
| O11—C11—C141—C142 | 14.3 (4) | O21—C21—C241—C246 | −8.2 (3) |
| C111—C11—C141—C142 | −104.9 (3) | C211—C21—C241—C246 | 111.1 (3) |
| C131—C11—C141—C142 | 131.2 (3) | C231—C21—C241—C246 | −125.7 (3) |
| O11—C11—C141—C146 | −166.2 (3) | O21—C21—C241—C242 | 172.9 (2) |
| C111—C11—C141—C146 | 74.6 (3) | C211—C21—C241—C242 | −67.8 (3) |
| C131—C11—C141—C146 | −49.3 (3) | C231—C21—C241—C242 | 55.4 (3) |
| C146—C141—C142—C143 | −1.5 (4) | C246—C241—C242—C243 | −3.1 (4) |
| C11—C141—C142—C143 | 178.0 (3) | C21—C241—C242—C243 | 175.8 (3) |
| C141—C142—C143—C144 | −0.7 (5) | C241—C242—C243—C244 | 2.0 (5) |
| C142—C143—C144—C145 | 1.9 (5) | C242—C243—C244—C245 | 0.6 (5) |
| C143—C144—C145—C146 | −0.8 (5) | C243—C244—C245—C246 | −2.1 (5) |
| C144—C145—C146—C141 | −1.5 (5) | C244—C245—C246—C241 | 0.9 (5) |
| C142—C141—C146—C145 | 2.6 (5) | C242—C241—C246—C245 | 1.7 (4) |
| C11—C141—C146—C145 | −176.9 (3) | C21—C241—C246—C245 | −177.3 (3) |
| O12—C12—C151—C152 | −134.9 (3) | O22—C22—C251—C252 | −47.4 (3) |
| C121—C12—C151—C152 | −16.8 (4) | C221—C22—C251—C252 | −165.1 (3) |
| C161—C12—C151—C152 | 106.5 (3) | C261—C22—C251—C252 | 71.1 (3) |
| O12—C12—C151—C156 | 45.5 (3) | O22—C22—C251—C256 | 134.2 (3) |
| C121—C12—C151—C156 | 163.6 (2) | C221—C22—C251—C256 | 16.5 (4) |
| C161—C12—C151—C156 | −73.1 (3) | C261—C22—C251—C256 | −107.3 (3) |
| C156—C151—C152—C153 | −0.8 (4) | C256—C251—C252—C253 | −1.5 (4) |
| C12—C151—C152—C153 | 179.6 (3) | C22—C251—C252—C253 | −180.0 (3) |
| C151—C152—C153—C154 | −0.2 (5) | C251—C252—C253—C254 | 0.7 (5) |
| C152—C153—C154—C155 | 0.3 (5) | C252—C253—C254—C255 | 0.5 (5) |
| C153—C154—C155—C156 | 0.7 (5) | C253—C254—C255—C256 | −0.9 (5) |
| C154—C155—C156—C151 | −1.7 (4) | C254—C255—C256—C251 | 0.1 (5) |
| C152—C151—C156—C155 | 1.7 (4) | C252—C251—C256—C255 | 1.0 (5) |
| C12—C151—C156—C155 | −178.7 (3) | C22—C251—C256—C255 | 179.5 (3) |
| O12—C12—C161—C162 | −161.7 (2) | O22—C22—C261—C266 | 161.9 (2) |
| C121—C12—C161—C162 | 74.7 (3) | C221—C22—C261—C266 | −75.4 (3) |
| C151—C12—C161—C162 | −47.3 (3) | C251—C22—C261—C266 | 47.2 (3) |
| O12—C12—C161—C166 | 16.9 (3) | O22—C22—C261—C262 | −17.8 (3) |
| C121—C12—C161—C166 | −106.7 (3) | C221—C22—C261—C262 | 104.9 (3) |
| C151—C12—C161—C166 | 131.3 (3) | C251—C22—C261—C262 | −132.6 (3) |
| C166—C161—C162—C163 | −1.6 (4) | C266—C261—C262—C263 | 0.3 (4) |
| C12—C161—C162—C163 | 177.0 (2) | C22—C261—C262—C263 | −180.0 (3) |
| C161—C162—C163—C164 | 0.0 (4) | C261—C262—C263—C264 | 0.8 (5) |
| C162—C163—C164—C165 | 1.7 (4) | C262—C263—C264—C265 | −1.2 (5) |
| C163—C164—C165—C166 | −1.9 (5) | C263—C264—C265—C266 | 0.5 (5) |
| C164—C165—C166—C161 | 0.3 (4) | C264—C265—C266—C261 | 0.6 (4) |
| C162—C161—C166—C165 | 1.5 (4) | C262—C261—C266—C265 | −1.0 (4) |
| C12—C161—C166—C165 | −177.1 (2) | C22—C261—C266—C265 | 179.3 (3) |
| N1—C2—C3—N4 | 61.6 (4) | N52—C62—C72—N82 | 179.5 (4) |
| N51—C61—C71—N81 | 60.5 (13) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O11—H11···N1 | 0.84 | 1.89 | 2.715 (3) | 169 |
| O12—H12···O11 | 0.84 | 1.92 | 2.687 (2) | 152 |
| O21—H21···N51 | 0.84 | 1.90 | 2.743 (3) | 178 |
| O22—H22···O21 | 0.84 | 2.01 | 2.790 (2) | 154 |
| N1—H1A···O11i | 0.91 | 2.31 | 3.103 (3) | 146 |
| N1—H1B···O12i | 0.91 | 2.49 | 3.142 (3) | 129 |
| N4—H4A···O12i | 0.91 | 2.45 | 3.358 (3) | 172 |
| N51—H51A···O21ii | 0.91 | 2.43 | 3.252 (3) | 151 |
| Symmetry codes: (i) −x, −y+1, −z+1; (ii) −x+1, −y, −z. |
Experimental details
| | (I) | (II) |
| Crystal data |
| Chemical formula | [Fe(C18H15O)]·C12H12N2 | [Fe(C18H15O)]·C2H8N2 |
| Mr | 734.69 | 610.55 |
| Crystal system, space group | Triclinic, P1 | Triclinic, P1 |
| Temperature (K) | 150 | 150 |
| a, b, c (Å) | 9.7191 (3), 11.3281 (4), 17.5549 (7) | 9.8551 (2), 11.7183 (2), 27.6810 (5) |
| α, β, γ (°) | 78.6970 (16), 87.8100 (15), 75.772 (2) | 82.3495 (8), 84.4918 (8), 73.4846 (8) |
| V (Å3) | 1837.05 (11) | 3031.88 (10) |
| Z | 2 | 4 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 0.45 | 0.54 |
| Crystal size (mm) | 0.20 × 0.18 × 0.18 | 0.32 × 0.28 × 0.26 |
| |
| Data collection |
| Diffractometer | Kappa-CCD
diffractometer | Kappa-CCD
diffractometer |
| Absorption correction | Multi-scan
(DENZO-SMN; Otwinowski & Minor, 1997) | Multi-scan
(DENZO-SMN; Otwinowski & Minor, 1997) |
| Tmin, Tmax | 0.915, 0.923 | 0.848, 0.873 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 23051, 8061, 4882 | 39122, 13826, 8093 |
| Rint | 0.094 | 0.088 |
| (sin θ/λ)max (Å−1) | 0.643 | 0.650 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.051, 0.125, 0.97 | 0.050, 0.123, 0.98 |
| No. of reflections | 8061 | 13826 |
| No. of parameters | 481 | 795 |
| No. of restraints | 0 | 9 |
| H-atom treatment | H-atom parameters constrained | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.29, −0.40 | 0.38, −0.42 |
Selected torsion angles (º) for (I) top| O1—C1—C11—C12 | −155.0 (2) | O2—C2—C21—C22 | 34.4 (3) |
| C31—C1—C11—C12 | 85.7 (3) | C51—C2—C21—C22 | −78.2 (3) |
| C41—C1—C11—C12 | −37.2 (4) | C61—C2—C21—C22 | 156.6 (2) |
| C73—C74—C77—C87 | 128.6 (3) | C74—C77—C87—C84 | −179.8 (2) |
| C83—C84—C87—C77 | −64.3 (3) | | |
Hydrogen-bond geometry (Å, º) for (I) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1—H1···N71 | 0.84 | 1.92 | 2.751 (3) | 170 |
| O2—H2···O1 | 0.84 | 2.07 | 2.845 (2) | 153 |
Selected torsion angles (º) for (II) top| O11—C11—C111—C112 | −153.6 (2) | O21—C21—C211—C212 | 161.3 (2) |
| C131—C11—C111—C112 | 88.1 (3) | C231—C21—C211—C212 | −79.4 (3) |
| C141—C11—C111—C112 | −35.0 (3) | C241—C21—C211—C212 | 42.5 (3) |
| O12—C12—C121—C122 | 41.0 (3) | O22—C22—C221—C222 | −36.2 (3) |
| C151—C12—C121—C122 | −73.4 (3) | C251—C22—C221—C222 | 78.7 (3) |
| C161—C12—C121—C122 | 165.0 (2) | C261—C22—C221—C222 | −159.7 (2) |
| N1—C2—C3—N4 | 61.6 (4) | N52—C62—C72—N82 | 179.5 (4) |
| N51—C61—C71—N81 | 60.5 (13) | | |
Hydrogen-bond geometry (Å, º) for (II) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O11—H11···N1 | 0.84 | 1.89 | 2.715 (3) | 169 |
| O12—H12···O11 | 0.84 | 1.92 | 2.687 (2) | 152 |
| O21—H21···N51 | 0.84 | 1.90 | 2.743 (3) | 178 |
| O22—H22···O21 | 0.84 | 2.01 | 2.790 (2) | 154 |
| N1—H1A···O11i | 0.91 | 2.31 | 3.103 (3) | 146 |
| N1—H1B···O12i | 0.91 | 2.49 | 3.142 (3) | 129 |
| N4—H4A···O12i | 0.91 | 2.45 | 3.358 (3) | 172 |
| N51—H51A···O21ii | 0.91 | 2.43 | 3.252 (3) | 151 |
| Symmetry codes: (i) −x, −y+1, −z+1; (ii) −x+1, −y, −z. |
 O hydrogen bond in the ferrocenediol component and a single O—H
O hydrogen bond in the ferrocenediol component and a single O—H N hydrogen bond linking the two components into a finite monomeric adduct. Ferrocene-1,1′-diylbis(diphenylmethanol)–ethylenediamine (1/1), [Fe(C18H15O)2]·C2H8N2, crystallizes with Z′ = 2 in space group P
N hydrogen bond linking the two components into a finite monomeric adduct. Ferrocene-1,1′-diylbis(diphenylmethanol)–ethylenediamine (1/1), [Fe(C18H15O)2]·C2H8N2, crystallizes with Z′ = 2 in space group P N and N—H
N and N—H O hydrogen bonds, in which both diamine N atoms participate; in the second type of aggregate, the diamine component is disordered over two sets of sites, but only one N atom is involved in the hydrogen bonding.
O hydrogen bonds, in which both diamine N atoms participate; in the second type of aggregate, the diamine component is disordered over two sets of sites, but only one N atom is involved in the hydrogen bonding.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2001/08/00/gg1062/gg1062fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2001/08/00/gg1062/gg1062fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2001/08/00/gg1062/gg1062fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/c/issues/2001/08/00/gg1062/gg1062fig4thm.gif)




The organometallic diol ferrocene-1,1'-diylbis(diphenylmethanol), [Fe(C5H4CPh2OH)2], forms hydrogen-bonded adducts with a wide range of hydrogen-bond acceptors, particularly cyclic and cage amines (Ferguson et al., 1993, 1995; Glidewell et al., 1994). Thus with piperazine, HN(CH2CH2)2NH, an adduct of 1:1 stoichiometry is formed in which the supramolecular aggregation consists of spiral C22(13) chains of alternating diol and diamine units containing two distinct O—H···N hydrogen bonds (Glidewell et al., 1994); by contrast, in the 1:1 adduct formed with 4,4'-bipyridyl, only half of the bipyridyl molecules are involved in the supramolecular aggregation, which takes the form of the centrosymmetric three-component aggregate (diol)-(diamine)-(diol), while the remainder of the bipyridyl molecules simply occupy isolated sites in the structure (Glidewell et al., 1994). While a wide range of supramolecular structures has been observed in adducts formed by this ferrocenediol (Ferguson et al., 1993, 1995; Glidewell et al., 1994), no one structure type is predictable from knowledge of the remainder: in particular the hydrogen-bond donor and acceptor behaviour of the non-ferrocenediol components is not yet readily predictable. However, a frequently-observed pattern, found in the adducts formed with 4,4'-bipyridyl, 1,4-dioxan, dimethylformamide and hexamethylenetetramine, is that of a single intramolecular O—H···O hydrogen bond in the ferrocenediol component forming an S(8) ring, together with a single O—H···A hydrogen bond to the acceptor atom A, which may be N or O, in the second component.
In the adduct with 1,2-bis(4-pyridyl)ethane, (I) (Fig. 1), there is an intramolecular O—H···O hydrogen bond (Table 2) and, in addition O1 acts as hydrogen-bond donor to N71; N81, on the other hand, does not act as a hydrogen-bond acceptor, even of soft C—H···N hydrogen bonds, so that (I) is a simple finite adduct, whose hydrogen bonding can be characterized by the graph-set descriptor S(8)D, precisely as for the 1:1 adduct formed by the ferrocenediol with dimethylformamide (Glidewell et al., 1994). The constitution of (I) is thus entirely different from that of the analogous 1:1 adduct formed with 4,4'-bipyridyl itself. \sch
The 1,2-diaminoethane adduct (II) also exhibits 1:1 stoichiometry but in this case the diamine component is potentially a fourfold donor of hydrogen bonds as well as potentially a twofold acceptor; adduct (II) crystallizes with Z' = 2 and there are two distinct finite centrosymmetric aggregates in the structure which exhibit different patterns of hydrogen-bonding behaviour. In the aggregate of type 1, which contains ferrocenediol 1, based on Fe1, and diamine 1, containing N1 and N4, the neutral molecular components are all fully ordered. In addition to the intramolecular O—H···O hydrogen bond in the ferrocenediol, which generates the usual S(8) motif, the individual molecular components are linked by both O—H···N and N—H···O hydrogen bonds, but there are no N—H···N hydrogen bonds present. Hydroxyl O11 acts as a hydrogen-bond donor to N1 within the asymmetric unit (Fig. 2); N1 in the diamine at (x, y, z) acts as a hydrogen-bond donor, via H1A and H1B, O11 and O12, respectively, in the ferrocenediol at (-x, 1 - y, 1 - z), and N4 at (x, y, z) also acts as donor, via H4A, to O12 at (-x, 1 - y, 1 - z) but H4B plays no part in the hydrogen bonding. In this manner, a four-component aggregate is formed, centred at (0, 1/2, 1/2) (Fig. 3) and containing a central R44(8) ring and pairs of both R23(6) and R12(7) rings, in which O11 and O12, respectively, are the double acceptors.
The aggregate of type 2 contains the fully ordered ferrocenediol 2, based on Fe2, and the disordered diamine 2 (Fig. 2): in both of the orientations of this diamine, only one N atom participates in the hydrogen bonding, so that the rest of the chain, untethered at the distal end, is free to adopt more than one conformation. By contrast, in the type 1 aggregate both N atoms are engaged in ring formation, so locking the diamine component into a single conformation. The two amine conformers in the type 2 aggregate have refined site-occupation factors of 0.756 (5) and 0.244 (5), respectively. In addition, in the minor conformer, there is rotational disorder of the terminal NH2 group based on N82: the partial H atoms on N82 could not be unambiguously located from difference maps, and hence they were omitted from the refinements. This type 2 aggregate contains the same O—H···O hydrogen bond as in the type 1 aggregate, and O21 in the ferrocenediol acts as hydrogen bond donor to N51 within the asymmetric unit: N51 at (x, y, z) in turn acts as hydrogen-bond donor, via H51A, to O21 in the ferrocenediol at (1 - x, -y, -z), so generating an R44(8) ring centred at (1/2, 0, 0) (Fig. 4). None of the other N—H bonds in this aggregate participates in hydrogen bonding, and there are no interactions between aggregates of the two types. The hydrogen-bonding patterns within these two types of aggregate (Figs. 3 and 4) may be contrasted with the very simple R66(12) ring in the four-component aggregate formed by the ferrocenediol in its adduct with methanol (Ferguson et al., 1993). Equally, the behaviour of the diamine component in (II) may be contrasted with that of piperazine in its adduct with the ferrocenediol (Glidewell et al., 1994), where the diamine acts only as a hydrogen-bond acceptor, and the ferrocenediol only as a hydrogen-bond donor.
In the ferrocenediol components of (I) and (II) the cyclopentadienyl rings are nearly eclipsed. A convenient measure of the relative twist of the rings in (I) is the torsion angle C11—Cg1—Cg2—C21, where Cg1 and Cg2 are the centroids of the two rings: to allow for the fact that the rings do not have local C5 rotational symmetry, because of the variations in the C—C bond distances, the mean value of the torsion angles C1n—Cg1—Cg2—C2n (n = 1 - 5) provides a better measure. For (II), the relevant values are those of C11n—Cg1—Cg2—C12n and C21n—Cg3—Cg4—C22n (n =1 - 5). For perfect eclipsing, this mean value should be (72 x n)° (n = zero or integer): the observed values are 65.4 (4)° in (I) and 53.7 (4)° and -60.0 (4)° in (II), indicating that each independent ferrocenediol adopts a conformation which is close to eclipsed, with the substituents on the two rings offset from one another by one-fifth of a turn.
The orientation of the exocyclic CPh2OH substituents relative to the adjacent rings (Table 1 and 3) is largely determined by the formation of the intramolecular O—H···O hydrogen bonds. In adduct (I), the central aliphatic portion of the diamine has an almost planar skeleton, but the rings are significantly twisted out of this plane (Table 1, Fig. 1). In adduct (II), the synclinal conformation of the diamine component in the type 1 aggregate is most readily ascribed to the hydrogen bonding, but such a simple explanation cannot be valid for the diamine in the type 2 aggregate, where the major and minor components of the amine adopt synclinal and antiperiplanar conformations respectively (Tables 3 and 4).