In 3-
tert-butyl-1-(4-chlorophenyl)-4-phenylindeno[1,2-
b]pyrazolo[4,3-
e]pyridin-5(1
H)-one, C
29H
22ClN
3O, (I), inversion-related pairs of molecules are linked by C—H

O hydrogen bonds to form
R22(18) dimers, which are themselves linked into a chain by a π–π stacking interaction between inversion-related pairs of molecules. In 3-
tert-butyl-1-(4-chlorophenyl)-4-(4-methoxyphenyl)indeno[1,2-
b]pyrazolo[4,3-
e]pyridin-5(1
H)-one, C
30H
24ClN
3O
2, (II), which crystallizes in the space group
P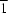
, with
Z′ = 2 and with different orientations for the methoxy groups in the two independent molecules, a combination of C—H

O and C—H

π(arene) hydrogen bonds links the molecules into chains of rings, which are further linked into sheets by a π–π stacking interaction.
Supporting information
CCDC references: 862227; 862228
For the synthesis of compounds (I) and (II), equimolar quantities (1 mmol of
each component) of indan-1,3-dione 4-R-benzaldehyde, where R = H
for compound (I) and R = OMe for compound (II) and
5-amino-3-tert-butyl-1-(4-chlorophenyl)-1H-pyrazole were added
to a mixture of water/triethylamine (3 ml, 15:1 v/v), and then
subjected to microwave irradiation at 353 K with a maximum power of 80 W for
10 min. The reaction mixture was allowed to cool to ambient temperature and it
was then extracted with dichloromethane (3 × 15 ml). The combined
extracts were dried over sodium sulfate, filtered and concentrated to give a
red solid. The products were recrystallized from ethanol, at ambient
temperature and in air, to give light yellow crystals suitable for
single-crystal X-ray diffraction. Compound (I) (R = H): yield 77%. m.p.
510–511 K; MS (70 eV) m/z (%) 465/463 (25/71,
M+2/M), 450/448 (36/100), 435/433 (9/26), 422/420 (7/20); HRMS
m/z found 463.1469, C29H22ClN3O requires m/z
= 463.1451. Compound (II) (R = OMe), yield 75%, m.p. 503–504 K: MS:
(70 eV) m/z (%) 495/493 (28/80, M+2/M), 480/478
(30/100), 465/463 (11/37), 452/450 (10/29); HRMS m/z found 493.1544,
C30H24ClN3O2 requires m/z = 493.1557.
All H atoms were located in difference maps and then treated as riding atoms in
geometrically idealized positions, with C—H distances of 0.95 (aromatic) or
0.98 Å (methyl), and with Uiso(U) = kUeq(C), where
k = 1.5 for the methyl groups, which were permitted to rotate but not
to tilt, and 1.2 otherwise.
For both compounds, data collection: COLLECT (Hooft, 1999); cell refinement: DIRAX/LSQ (Duisenberg et al., 2000); data reduction: EVALCCD (Duisenberg et al., 2003); program(s) used to solve structure: SIR2004 (Burla et al., 2005); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008); molecular graphics: PLATON (Spek, 2009); software used to prepare material for publication: SHELXL97 (Sheldrick, 2008) and PLATON (Spek, 2009).
(I) 3-
tert-butyl-1-(4-chlorophenyl)-4-
phenylindeno[1,2-
b]pyrazolo[4,3-
e]pyridin-5(1
H)-one
top
Crystal data top
| C29H22ClN3O | Z = 2 |
| Mr = 463.95 | F(000) = 484 |
| Triclinic, P1 | Dx = 1.336 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 8.1014 (5) Å | Cell parameters from 5311 reflections |
| b = 11.3554 (10) Å | θ = 3.0–27.5° |
| c = 13.5762 (11) Å | µ = 0.19 mm−1 |
| α = 102.755 (7)° | T = 120 K |
| β = 90.013 (8)° | Block, yellow |
| γ = 108.268 (7)° | 0.43 × 0.22 × 0.16 mm |
| V = 1153.47 (16) Å3 | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 5310 independent reflections |
| Radiation source: Bruker–Nonius FR591 rotating anode | 4065 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.045 |
| Detector resolution: 9.091 pixels mm-1 | θmax = 27.5°, θmin = 3.0° |
| ϕ and ω scans | h = −10→10 |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | k = −14→14 |
| Tmin = 0.921, Tmax = 0.970 | l = −17→17 |
| 29371 measured reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.043 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.098 | H-atom parameters constrained |
| S = 1.06 | w = 1/[σ2(Fo2) + (0.034P)2 + 0.6438P]
where P = (Fo2 + 2Fc2)/3 |
| 5310 reflections | (Δ/σ)max = 0.001 |
| 310 parameters | Δρmax = 0.22 e Å−3 |
| 0 restraints | Δρmin = −0.39 e Å−3 |
Crystal data top
| C29H22ClN3O | γ = 108.268 (7)° |
| Mr = 463.95 | V = 1153.47 (16) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 8.1014 (5) Å | Mo Kα radiation |
| b = 11.3554 (10) Å | µ = 0.19 mm−1 |
| c = 13.5762 (11) Å | T = 120 K |
| α = 102.755 (7)° | 0.43 × 0.22 × 0.16 mm |
| β = 90.013 (8)° | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 5310 independent reflections |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | 4065 reflections with I > 2σ(I) |
| Tmin = 0.921, Tmax = 0.970 | Rint = 0.045 |
| 29371 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.043 | 0 restraints |
| wR(F2) = 0.098 | H-atom parameters constrained |
| S = 1.06 | Δρmax = 0.22 e Å−3 |
| 5310 reflections | Δρmin = −0.39 e Å−3 |
| 310 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| N1 | 1.02625 (16) | 0.70513 (12) | 0.52904 (9) | 0.0174 (3) | |
| N2 | 1.11894 (17) | 0.80399 (12) | 0.60701 (10) | 0.0194 (3) | |
| C3 | 1.0432 (2) | 0.78827 (15) | 0.69158 (12) | 0.0186 (3) | |
| C3A | 0.8913 (2) | 0.67420 (14) | 0.67044 (11) | 0.0169 (3) | |
| C4 | 0.7534 (2) | 0.60694 (15) | 0.72231 (11) | 0.0178 (3) | |
| C4A | 0.6384 (2) | 0.49531 (15) | 0.66349 (11) | 0.0180 (3) | |
| C5 | 0.4793 (2) | 0.40028 (15) | 0.68971 (12) | 0.0202 (3) | |
| C5A | 0.4119 (2) | 0.30062 (15) | 0.59350 (12) | 0.0187 (3) | |
| C6 | 0.2704 (2) | 0.18974 (15) | 0.57584 (13) | 0.0222 (3) | |
| H6 | 0.1999 | 0.1665 | 0.6288 | 0.027* | |
| C7 | 0.2351 (2) | 0.11339 (16) | 0.47773 (13) | 0.0238 (4) | |
| H7 | 0.1386 | 0.0370 | 0.4634 | 0.029* | |
| C8 | 0.3394 (2) | 0.14757 (15) | 0.40026 (13) | 0.0227 (3) | |
| H8 | 0.3126 | 0.0941 | 0.3339 | 0.027* | |
| C9 | 0.4824 (2) | 0.25901 (15) | 0.41851 (12) | 0.0196 (3) | |
| H9 | 0.5536 | 0.2822 | 0.3658 | 0.024* | |
| C9A | 0.51688 (19) | 0.33451 (14) | 0.51606 (12) | 0.0172 (3) | |
| C9B | 0.65699 (19) | 0.45486 (14) | 0.55882 (11) | 0.0166 (3) | |
| N10 | 0.77787 (16) | 0.51619 (12) | 0.50610 (10) | 0.0174 (3) | |
| C10A | 0.88979 (19) | 0.62382 (14) | 0.56436 (11) | 0.0162 (3) | |
| C11 | 1.07391 (19) | 0.70754 (14) | 0.42839 (11) | 0.0172 (3) | |
| C12 | 0.9708 (2) | 0.62101 (16) | 0.34494 (12) | 0.0215 (3) | |
| H12 | 0.8713 | 0.5542 | 0.3545 | 0.026* | |
| C13 | 1.0137 (2) | 0.63248 (16) | 0.24773 (12) | 0.0237 (4) | |
| H13 | 0.9425 | 0.5744 | 0.1907 | 0.028* | |
| C14 | 1.1601 (2) | 0.72867 (16) | 0.23410 (12) | 0.0228 (3) | |
| Cl14 | 1.20917 (6) | 0.74609 (5) | 0.11228 (3) | 0.03335 (13) | |
| C15 | 1.2674 (2) | 0.81194 (16) | 0.31660 (13) | 0.0230 (3) | |
| H15 | 1.3704 | 0.8756 | 0.3065 | 0.028* | |
| C16 | 1.2239 (2) | 0.80201 (15) | 0.41393 (12) | 0.0204 (3) | |
| H16 | 1.2963 | 0.8596 | 0.4707 | 0.024* | |
| C31 | 1.1282 (2) | 0.88690 (16) | 0.78844 (12) | 0.0225 (3) | |
| C32 | 1.3107 (2) | 0.96804 (17) | 0.76897 (13) | 0.0260 (4) | |
| H32A | 1.3009 | 1.0128 | 0.7164 | 0.039* | |
| H32B | 1.3816 | 0.9127 | 0.7465 | 0.039* | |
| H32C | 1.3661 | 1.0304 | 0.8316 | 0.039* | |
| C33 | 1.0198 (2) | 0.97662 (18) | 0.82136 (15) | 0.0350 (4) | |
| H33A | 0.9038 | 0.9274 | 0.8367 | 0.052* | |
| H33B | 1.0082 | 1.0176 | 0.7665 | 0.052* | |
| H33C | 1.0782 | 1.0420 | 0.8819 | 0.052* | |
| C34 | 1.1488 (3) | 0.8224 (2) | 0.87339 (14) | 0.0388 (5) | |
| H34A | 1.2138 | 0.7629 | 0.8504 | 0.058* | |
| H34B | 1.0335 | 0.7757 | 0.8913 | 0.058* | |
| H34C | 1.2125 | 0.8873 | 0.9329 | 0.058* | |
| C41 | 0.7185 (2) | 0.64766 (15) | 0.83053 (11) | 0.0191 (3) | |
| C42 | 0.6362 (2) | 0.73944 (16) | 0.85842 (12) | 0.0231 (3) | |
| H42 | 0.6110 | 0.7813 | 0.8098 | 0.028* | |
| C43 | 0.5908 (2) | 0.77002 (17) | 0.95746 (13) | 0.0274 (4) | |
| H43 | 0.5355 | 0.8332 | 0.9764 | 0.033* | |
| C44 | 0.6260 (2) | 0.70857 (17) | 1.02828 (12) | 0.0267 (4) | |
| H44 | 0.5954 | 0.7299 | 1.0959 | 0.032* | |
| C45 | 0.7058 (2) | 0.61610 (17) | 1.00049 (12) | 0.0256 (4) | |
| H45 | 0.7292 | 0.5734 | 1.0490 | 0.031* | |
| C46 | 0.7516 (2) | 0.58556 (16) | 0.90190 (12) | 0.0237 (4) | |
| H46 | 0.8060 | 0.5218 | 0.8831 | 0.028* | |
| O5 | 0.41358 (15) | 0.40214 (12) | 0.77009 (9) | 0.0282 (3) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| N1 | 0.0163 (6) | 0.0172 (6) | 0.0169 (6) | 0.0031 (5) | 0.0029 (5) | 0.0036 (5) |
| N2 | 0.0187 (7) | 0.0197 (7) | 0.0180 (7) | 0.0049 (5) | 0.0007 (5) | 0.0028 (5) |
| C3 | 0.0172 (7) | 0.0201 (8) | 0.0189 (8) | 0.0065 (6) | 0.0023 (6) | 0.0048 (6) |
| C3A | 0.0191 (7) | 0.0185 (7) | 0.0149 (7) | 0.0081 (6) | 0.0025 (6) | 0.0048 (6) |
| C4 | 0.0190 (8) | 0.0211 (8) | 0.0165 (7) | 0.0094 (6) | 0.0027 (6) | 0.0065 (6) |
| C4A | 0.0176 (7) | 0.0214 (8) | 0.0179 (7) | 0.0083 (6) | 0.0042 (6) | 0.0077 (6) |
| C5 | 0.0180 (8) | 0.0239 (8) | 0.0216 (8) | 0.0076 (7) | 0.0033 (6) | 0.0097 (7) |
| C5A | 0.0178 (7) | 0.0213 (8) | 0.0202 (8) | 0.0087 (6) | 0.0025 (6) | 0.0076 (6) |
| C6 | 0.0189 (8) | 0.0229 (8) | 0.0290 (9) | 0.0080 (7) | 0.0069 (7) | 0.0129 (7) |
| C7 | 0.0196 (8) | 0.0182 (8) | 0.0324 (9) | 0.0046 (7) | 0.0021 (7) | 0.0055 (7) |
| C8 | 0.0224 (8) | 0.0198 (8) | 0.0251 (8) | 0.0074 (7) | 0.0014 (7) | 0.0026 (7) |
| C9 | 0.0199 (8) | 0.0194 (8) | 0.0214 (8) | 0.0074 (6) | 0.0034 (6) | 0.0069 (6) |
| C9A | 0.0159 (7) | 0.0172 (7) | 0.0213 (8) | 0.0075 (6) | 0.0029 (6) | 0.0067 (6) |
| C9B | 0.0172 (7) | 0.0182 (7) | 0.0171 (7) | 0.0083 (6) | 0.0024 (6) | 0.0056 (6) |
| N10 | 0.0174 (6) | 0.0177 (6) | 0.0189 (6) | 0.0069 (5) | 0.0042 (5) | 0.0063 (5) |
| C10A | 0.0154 (7) | 0.0174 (7) | 0.0180 (7) | 0.0067 (6) | 0.0036 (6) | 0.0063 (6) |
| C11 | 0.0168 (7) | 0.0197 (8) | 0.0184 (7) | 0.0085 (6) | 0.0060 (6) | 0.0074 (6) |
| C12 | 0.0175 (8) | 0.0251 (8) | 0.0212 (8) | 0.0050 (7) | 0.0054 (6) | 0.0065 (7) |
| C13 | 0.0221 (8) | 0.0310 (9) | 0.0182 (8) | 0.0092 (7) | 0.0042 (6) | 0.0049 (7) |
| C14 | 0.0249 (8) | 0.0306 (9) | 0.0202 (8) | 0.0145 (7) | 0.0096 (7) | 0.0124 (7) |
| Cl14 | 0.0376 (3) | 0.0441 (3) | 0.0229 (2) | 0.0134 (2) | 0.01290 (18) | 0.01654 (19) |
| C15 | 0.0218 (8) | 0.0217 (8) | 0.0281 (9) | 0.0073 (7) | 0.0094 (7) | 0.0104 (7) |
| C16 | 0.0196 (8) | 0.0203 (8) | 0.0231 (8) | 0.0077 (7) | 0.0040 (6) | 0.0069 (6) |
| C31 | 0.0211 (8) | 0.0239 (8) | 0.0177 (8) | 0.0029 (7) | 0.0018 (6) | 0.0018 (7) |
| C32 | 0.0215 (8) | 0.0276 (9) | 0.0232 (8) | 0.0031 (7) | 0.0012 (7) | 0.0012 (7) |
| C33 | 0.0247 (9) | 0.0320 (10) | 0.0369 (10) | 0.0055 (8) | 0.0043 (8) | −0.0098 (8) |
| C34 | 0.0380 (11) | 0.0435 (12) | 0.0246 (10) | −0.0045 (9) | −0.0080 (8) | 0.0126 (9) |
| C41 | 0.0173 (7) | 0.0228 (8) | 0.0154 (7) | 0.0038 (6) | 0.0036 (6) | 0.0048 (6) |
| C42 | 0.0235 (8) | 0.0264 (9) | 0.0206 (8) | 0.0090 (7) | 0.0035 (6) | 0.0063 (7) |
| C43 | 0.0301 (9) | 0.0302 (9) | 0.0232 (9) | 0.0134 (8) | 0.0068 (7) | 0.0035 (7) |
| C44 | 0.0271 (9) | 0.0306 (9) | 0.0167 (8) | 0.0038 (7) | 0.0072 (7) | 0.0021 (7) |
| C45 | 0.0279 (9) | 0.0298 (9) | 0.0187 (8) | 0.0058 (7) | 0.0022 (7) | 0.0095 (7) |
| C46 | 0.0254 (9) | 0.0264 (9) | 0.0206 (8) | 0.0094 (7) | 0.0042 (7) | 0.0067 (7) |
| O5 | 0.0247 (6) | 0.0369 (7) | 0.0196 (6) | 0.0030 (5) | 0.0079 (5) | 0.0093 (5) |
Geometric parameters (Å, º) top
| N1—C10A | 1.3640 (19) | C13—C14 | 1.382 (2) |
| N1—N2 | 1.3752 (18) | C13—H13 | 0.9500 |
| N1—C11 | 1.4237 (19) | C14—C15 | 1.386 (2) |
| N2—C3 | 1.3223 (19) | C14—Cl14 | 1.7402 (16) |
| C3—C3A | 1.453 (2) | C15—C16 | 1.387 (2) |
| C3—C31 | 1.524 (2) | C15—H15 | 0.9500 |
| C3A—C4 | 1.425 (2) | C16—H16 | 0.9500 |
| C3A—C10A | 1.426 (2) | C31—C34 | 1.531 (2) |
| C4—C4A | 1.388 (2) | C31—C32 | 1.536 (2) |
| C4—C41 | 1.495 (2) | C31—C33 | 1.539 (2) |
| C4A—C9B | 1.417 (2) | C32—H32A | 0.9800 |
| C4A—C5 | 1.500 (2) | C32—H32B | 0.9800 |
| C5—O5 | 1.2133 (18) | C32—H32C | 0.9800 |
| C5—C5A | 1.498 (2) | C33—H33A | 0.9800 |
| C5A—C6 | 1.385 (2) | C33—H33B | 0.9800 |
| C5A—C9A | 1.397 (2) | C33—H33C | 0.9800 |
| C6—C7 | 1.394 (2) | C34—H34A | 0.9800 |
| C6—H6 | 0.9500 | C34—H34B | 0.9800 |
| C7—C8 | 1.395 (2) | C34—H34C | 0.9800 |
| C7—H7 | 0.9500 | C41—C46 | 1.389 (2) |
| C8—C9 | 1.395 (2) | C41—C42 | 1.391 (2) |
| C8—H8 | 0.9500 | C42—C43 | 1.392 (2) |
| C9—C9A | 1.384 (2) | C42—H42 | 0.9500 |
| C9—H9 | 0.9500 | C43—C44 | 1.383 (2) |
| C9A—C9B | 1.473 (2) | C43—H43 | 0.9500 |
| C9B—N10 | 1.3225 (19) | C44—C45 | 1.383 (2) |
| N10—C10A | 1.349 (2) | C44—H44 | 0.9500 |
| C11—C12 | 1.393 (2) | C45—C46 | 1.388 (2) |
| C11—C16 | 1.393 (2) | C45—H45 | 0.9500 |
| C12—C13 | 1.388 (2) | C46—H46 | 0.9500 |
| C12—H12 | 0.9500 | | |
| | | |
| C10A—N1—N2 | 110.46 (12) | C12—C13—H13 | 120.1 |
| C10A—N1—C11 | 130.76 (13) | C13—C14—C15 | 120.63 (15) |
| N2—N1—C11 | 118.55 (12) | C13—C14—Cl14 | 119.68 (13) |
| C3—N2—N1 | 108.25 (12) | C15—C14—Cl14 | 119.68 (13) |
| N2—C3—C3A | 109.96 (13) | C14—C15—C16 | 119.73 (15) |
| N2—C3—C31 | 116.82 (13) | C14—C15—H15 | 120.1 |
| C3A—C3—C31 | 133.21 (14) | C16—C15—H15 | 120.1 |
| C4—C3A—C10A | 116.95 (14) | C15—C16—C11 | 119.97 (15) |
| C4—C3A—C3 | 139.19 (14) | C15—C16—H16 | 120.0 |
| C10A—C3A—C3 | 103.84 (13) | C11—C16—H16 | 120.0 |
| C4A—C4—C3A | 115.22 (14) | C3—C31—C34 | 111.04 (14) |
| C4A—C4—C41 | 118.19 (13) | C3—C31—C32 | 109.51 (13) |
| C3A—C4—C41 | 126.54 (14) | C34—C31—C32 | 107.80 (14) |
| C4—C4A—C9B | 121.36 (14) | C3—C31—C33 | 110.11 (14) |
| C4—C4A—C5 | 131.04 (14) | C34—C31—C33 | 110.35 (15) |
| C9B—C4A—C5 | 107.57 (13) | C32—C31—C33 | 107.95 (14) |
| O5—C5—C5A | 125.87 (15) | C31—C32—H32A | 109.5 |
| O5—C5—C4A | 128.74 (15) | C31—C32—H32B | 109.5 |
| C5A—C5—C4A | 105.37 (13) | H32A—C32—H32B | 109.5 |
| C6—C5A—C9A | 121.31 (15) | C31—C32—H32C | 109.5 |
| C6—C5A—C5 | 129.46 (14) | H32A—C32—H32C | 109.5 |
| C9A—C5A—C5 | 109.23 (13) | H32B—C32—H32C | 109.5 |
| C5A—C6—C7 | 117.73 (15) | C31—C33—H33A | 109.5 |
| C5A—C6—H6 | 121.1 | C31—C33—H33B | 109.5 |
| C7—C6—H6 | 121.1 | H33A—C33—H33B | 109.5 |
| C6—C7—C8 | 120.97 (15) | C31—C33—H33C | 109.5 |
| C6—C7—H7 | 119.5 | H33A—C33—H33C | 109.5 |
| C8—C7—H7 | 119.5 | H33B—C33—H33C | 109.5 |
| C7—C8—C9 | 121.12 (15) | C31—C34—H34A | 109.5 |
| C7—C8—H8 | 119.4 | C31—C34—H34B | 109.5 |
| C9—C8—H8 | 119.4 | H34A—C34—H34B | 109.5 |
| C9A—C9—C8 | 117.70 (15) | C31—C34—H34C | 109.5 |
| C9A—C9—H9 | 121.2 | H34A—C34—H34C | 109.5 |
| C8—C9—H9 | 121.2 | H34B—C34—H34C | 109.5 |
| C9—C9A—C5A | 121.18 (14) | C46—C41—C42 | 119.31 (14) |
| C9—C9A—C9B | 130.47 (14) | C46—C41—C4 | 120.38 (14) |
| C5A—C9A—C9B | 108.35 (13) | C42—C41—C4 | 120.00 (14) |
| N10—C9B—C4A | 125.90 (14) | C41—C42—C43 | 120.10 (16) |
| N10—C9B—C9A | 124.64 (14) | C41—C42—H42 | 120.0 |
| C4A—C9B—C9A | 109.44 (13) | C43—C42—H42 | 120.0 |
| C9B—N10—C10A | 112.11 (13) | C44—C43—C42 | 120.15 (16) |
| N10—C10A—N1 | 124.18 (13) | C44—C43—H43 | 119.9 |
| N10—C10A—C3A | 128.35 (14) | C42—C43—H43 | 119.9 |
| N1—C10A—C3A | 107.47 (13) | C45—C44—C43 | 119.91 (15) |
| C12—C11—C16 | 119.83 (14) | C45—C44—H44 | 120.0 |
| C12—C11—N1 | 121.46 (13) | C43—C44—H44 | 120.0 |
| C16—C11—N1 | 118.67 (14) | C44—C45—C46 | 120.11 (16) |
| C13—C12—C11 | 119.92 (15) | C44—C45—H45 | 119.9 |
| C13—C12—H12 | 120.0 | C46—C45—H45 | 119.9 |
| C11—C12—H12 | 120.0 | C45—C46—C41 | 120.41 (16) |
| C14—C13—C12 | 119.84 (16) | C45—C46—H46 | 119.8 |
| C14—C13—H13 | 120.1 | C41—C46—H46 | 119.8 |
| | | |
| C10A—N1—N2—C3 | −0.66 (17) | C9A—C9B—N10—C10A | 179.97 (13) |
| C11—N1—N2—C3 | 174.40 (13) | C9B—N10—C10A—N1 | −179.73 (14) |
| N1—N2—C3—C3A | −0.45 (17) | C9B—N10—C10A—C3A | 0.5 (2) |
| N1—N2—C3—C31 | 178.79 (13) | N2—N1—C10A—N10 | −178.30 (13) |
| N2—C3—C3A—C4 | −176.85 (18) | C11—N1—C10A—N10 | 7.4 (2) |
| C31—C3—C3A—C4 | 4.1 (3) | N2—N1—C10A—C3A | 1.50 (16) |
| N2—C3—C3A—C10A | 1.31 (17) | C11—N1—C10A—C3A | −172.77 (14) |
| C31—C3—C3A—C10A | −177.76 (16) | C4—C3A—C10A—N10 | −3.2 (2) |
| C10A—C3A—C4—C4A | 3.4 (2) | C3—C3A—C10A—N10 | 178.13 (14) |
| C3—C3A—C4—C4A | −178.58 (17) | C4—C3A—C10A—N1 | 176.99 (13) |
| C10A—C3A—C4—C41 | −173.94 (14) | C3—C3A—C10A—N1 | −1.66 (16) |
| C3—C3A—C4—C41 | 4.1 (3) | C10A—N1—C11—C12 | 3.3 (2) |
| C3A—C4—C4A—C9B | −1.6 (2) | N2—N1—C11—C12 | −170.54 (14) |
| C41—C4—C4A—C9B | 176.05 (14) | C10A—N1—C11—C16 | −179.09 (15) |
| C3A—C4—C4A—C5 | −179.59 (15) | N2—N1—C11—C16 | 7.0 (2) |
| C41—C4—C4A—C5 | −2.0 (2) | C16—C11—C12—C13 | −2.7 (2) |
| C4—C4A—C5—O5 | 1.5 (3) | N1—C11—C12—C13 | 174.81 (14) |
| C9B—C4A—C5—O5 | −176.75 (16) | C11—C12—C13—C14 | 1.0 (2) |
| C4—C4A—C5—C5A | −179.94 (15) | C12—C13—C14—C15 | 1.7 (2) |
| C9B—C4A—C5—C5A | 1.81 (16) | C12—C13—C14—Cl14 | −177.80 (13) |
| O5—C5—C5A—C6 | −3.2 (3) | C13—C14—C15—C16 | −2.6 (2) |
| C4A—C5—C5A—C6 | 178.18 (15) | Cl14—C14—C15—C16 | 176.91 (12) |
| O5—C5—C5A—C9A | 176.85 (16) | C14—C15—C16—C11 | 0.8 (2) |
| C4A—C5—C5A—C9A | −1.76 (17) | C12—C11—C16—C15 | 1.8 (2) |
| C9A—C5A—C6—C7 | −0.6 (2) | N1—C11—C16—C15 | −175.77 (14) |
| C5—C5A—C6—C7 | 179.46 (15) | N2—C3—C31—C34 | −131.81 (16) |
| C5A—C6—C7—C8 | 0.2 (2) | C3A—C3—C31—C34 | 47.2 (2) |
| C6—C7—C8—C9 | 0.1 (2) | N2—C3—C31—C32 | −12.9 (2) |
| C7—C8—C9—C9A | −0.1 (2) | C3A—C3—C31—C32 | 166.15 (16) |
| C8—C9—C9A—C5A | −0.2 (2) | N2—C3—C31—C33 | 105.68 (17) |
| C8—C9—C9A—C9B | 179.16 (15) | C3A—C3—C31—C33 | −75.3 (2) |
| C6—C5A—C9A—C9 | 0.6 (2) | C4A—C4—C41—C46 | 75.0 (2) |
| C5—C5A—C9A—C9 | −179.44 (14) | C3A—C4—C41—C46 | −107.65 (19) |
| C6—C5A—C9A—C9B | −178.90 (14) | C4A—C4—C41—C42 | −98.47 (18) |
| C5—C5A—C9A—C9B | 1.05 (17) | C3A—C4—C41—C42 | 78.8 (2) |
| C4—C4A—C9B—N10 | −1.3 (2) | C46—C41—C42—C43 | 1.2 (2) |
| C5—C4A—C9B—N10 | 177.16 (14) | C4—C41—C42—C43 | 174.77 (15) |
| C4—C4A—C9B—C9A | −179.69 (14) | C41—C42—C43—C44 | −0.5 (3) |
| C5—C4A—C9B—C9A | −1.24 (17) | C42—C43—C44—C45 | −0.3 (3) |
| C9—C9A—C9B—N10 | 2.3 (3) | C43—C44—C45—C46 | 0.5 (3) |
| C5A—C9A—C9B—N10 | −178.29 (14) | C44—C45—C46—C41 | 0.2 (3) |
| C9—C9A—C9B—C4A | −179.32 (15) | C42—C41—C46—C45 | −1.0 (2) |
| C5A—C9A—C9B—C4A | 0.13 (17) | C4—C41—C46—C45 | −174.60 (15) |
| C4A—C9B—N10—C10A | 1.8 (2) | | |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| C44—H44···O5i | 0.95 | 2.59 | 3.232 (2) | 125 |
| Symmetry code: (i) −x+1, −y+1, −z+2. |
(II) 3-
tert-butyl-1-(4-chlorophenyl)-4-
(4-methoxyphenyl)indeno[1,2-
b]pyrazolo[4,3-
e]pyridin-5(1
H)-one
top
Crystal data top
| C30H24ClN3O2 | Z = 4 |
| Mr = 493.97 | F(000) = 1032 |
| Triclinic, P1 | Dx = 1.358 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 11.1911 (9) Å | Cell parameters from 11085 reflections |
| b = 11.4258 (8) Å | θ = 3.0–27.5° |
| c = 20.095 (2) Å | µ = 0.19 mm−1 |
| α = 97.411 (6)° | T = 120 K |
| β = 93.272 (7)° | Block, yellow |
| γ = 107.635 (6)° | 0.42 × 0.30 × 0.29 mm |
| V = 2415.7 (4) Å3 | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 11085 independent reflections |
| Radiation source: Bruker–Nonius FR591 rotating anode | 7328 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.061 |
| Detector resolution: 9.091 pixels mm-1 | θmax = 27.5°, θmin = 3.0° |
| ϕ and ω scans | h = −14→14 |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | k = −14→14 |
| Tmin = 0.924, Tmax = 0.946 | l = −26→26 |
| 63410 measured reflections | |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.048 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.115 | H-atom parameters constrained |
| S = 1.06 | w = 1/[σ2(Fo2) + (0.0383P)2 + 1.5471P]
where P = (Fo2 + 2Fc2)/3 |
| 11085 reflections | (Δ/σ)max = 0.001 |
| 657 parameters | Δρmax = 0.34 e Å−3 |
| 0 restraints | Δρmin = −0.37 e Å−3 |
Crystal data top
| C30H24ClN3O2 | γ = 107.635 (6)° |
| Mr = 493.97 | V = 2415.7 (4) Å3 |
| Triclinic, P1 | Z = 4 |
| a = 11.1911 (9) Å | Mo Kα radiation |
| b = 11.4258 (8) Å | µ = 0.19 mm−1 |
| c = 20.095 (2) Å | T = 120 K |
| α = 97.411 (6)° | 0.42 × 0.30 × 0.29 mm |
| β = 93.272 (7)° | |
Data collection top
Bruker–Nonius KappaCCD
diffractometer | 11085 independent reflections |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2003) | 7328 reflections with I > 2σ(I) |
| Tmin = 0.924, Tmax = 0.946 | Rint = 0.061 |
| 63410 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.048 | 0 restraints |
| wR(F2) = 0.115 | H-atom parameters constrained |
| S = 1.06 | Δρmax = 0.34 e Å−3 |
| 11085 reflections | Δρmin = −0.37 e Å−3 |
| 657 parameters | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| N11 | 0.56735 (15) | 0.29090 (15) | 0.07001 (8) | 0.0218 (4) | |
| N12 | 0.46516 (15) | 0.22309 (15) | 0.02484 (8) | 0.0229 (4) | |
| C13 | 0.39573 (18) | 0.13181 (18) | 0.05363 (10) | 0.0208 (4) | |
| C13A | 0.45258 (18) | 0.13760 (18) | 0.12131 (10) | 0.0202 (4) | |
| C14 | 0.43084 (18) | 0.06972 (18) | 0.17645 (10) | 0.0212 (4) | |
| C14A | 0.51871 (18) | 0.11965 (18) | 0.23286 (10) | 0.0211 (4) | |
| C15 | 0.53088 (18) | 0.07236 (18) | 0.29793 (10) | 0.0219 (4) | |
| C15A | 0.64932 (18) | 0.16126 (18) | 0.33651 (10) | 0.0223 (4) | |
| C16 | 0.7073 (2) | 0.1574 (2) | 0.39844 (11) | 0.0263 (5) | |
| H16 | 0.6707 | 0.0939 | 0.4242 | 0.032* | |
| C17 | 0.8208 (2) | 0.2492 (2) | 0.42176 (11) | 0.0279 (5) | |
| H17 | 0.8630 | 0.2482 | 0.4639 | 0.033* | |
| C18 | 0.8730 (2) | 0.3423 (2) | 0.38429 (11) | 0.0272 (5) | |
| H18 | 0.9504 | 0.4043 | 0.4015 | 0.033* | |
| C19 | 0.81465 (19) | 0.34693 (19) | 0.32199 (10) | 0.0236 (4) | |
| H19 | 0.8505 | 0.4112 | 0.2966 | 0.028* | |
| C19A | 0.70208 (18) | 0.25398 (18) | 0.29840 (10) | 0.0211 (4) | |
| C19B | 0.62271 (18) | 0.22842 (18) | 0.23356 (10) | 0.0206 (4) | |
| N110 | 0.64826 (15) | 0.29262 (15) | 0.18321 (8) | 0.0213 (4) | |
| C110 | 0.56192 (18) | 0.24340 (18) | 0.12891 (10) | 0.0209 (4) | |
| C111 | 0.66203 (19) | 0.38911 (18) | 0.04842 (10) | 0.0211 (4) | |
| C112 | 0.7847 (2) | 0.4315 (2) | 0.07960 (11) | 0.0277 (5) | |
| H112 | 0.8072 | 0.3949 | 0.1162 | 0.033* | |
| C113 | 0.8739 (2) | 0.5274 (2) | 0.05709 (11) | 0.0281 (5) | |
| H113 | 0.9577 | 0.5573 | 0.0785 | 0.034* | |
| C114 | 0.84072 (19) | 0.57909 (19) | 0.00358 (11) | 0.0245 (5) | |
| Cl14 | 0.95510 (5) | 0.69863 (5) | −0.02428 (3) | 0.03492 (14) | |
| C115 | 0.7195 (2) | 0.5371 (2) | −0.02799 (11) | 0.0276 (5) | |
| H115 | 0.6978 | 0.5733 | −0.0650 | 0.033* | |
| C116 | 0.6295 (2) | 0.44192 (19) | −0.00538 (10) | 0.0251 (5) | |
| H116 | 0.5457 | 0.4129 | −0.0267 | 0.030* | |
| C131 | 0.27873 (18) | 0.04187 (19) | 0.01135 (10) | 0.0233 (4) | |
| C132 | 0.2536 (2) | 0.0929 (2) | −0.05300 (11) | 0.0323 (5) | |
| H13A | 0.2451 | 0.1757 | −0.0410 | 0.048* | |
| H13B | 0.1756 | 0.0372 | −0.0786 | 0.048* | |
| H13C | 0.3240 | 0.0986 | −0.0806 | 0.048* | |
| C133 | 0.3003 (2) | −0.0832 (2) | −0.00877 (12) | 0.0327 (5) | |
| H13D | 0.3760 | −0.0708 | −0.0327 | 0.049* | |
| H13E | 0.2273 | −0.1393 | −0.0383 | 0.049* | |
| H13F | 0.3115 | −0.1195 | 0.0318 | 0.049* | |
| C134 | 0.16281 (19) | 0.0254 (2) | 0.05084 (11) | 0.0295 (5) | |
| H13G | 0.1768 | −0.0077 | 0.0922 | 0.044* | |
| H13H | 0.0886 | −0.0326 | 0.0230 | 0.044* | |
| H13I | 0.1491 | 0.1059 | 0.0625 | 0.044* | |
| C141 | 0.32811 (18) | −0.04872 (18) | 0.17933 (10) | 0.0210 (4) | |
| C142 | 0.34209 (19) | −0.16257 (19) | 0.15461 (11) | 0.0256 (5) | |
| H142 | 0.4120 | −0.1648 | 0.1304 | 0.031* | |
| C143 | 0.25517 (19) | −0.27316 (19) | 0.16482 (11) | 0.0272 (5) | |
| H143 | 0.2658 | −0.3505 | 0.1477 | 0.033* | |
| C144 | 0.15299 (18) | −0.27030 (18) | 0.20006 (11) | 0.0232 (4) | |
| C145 | 0.13599 (19) | −0.15711 (19) | 0.22340 (11) | 0.0245 (5) | |
| H145 | 0.0640 | −0.1552 | 0.2458 | 0.029* | |
| C146 | 0.22390 (18) | −0.04713 (19) | 0.21404 (10) | 0.0235 (4) | |
| H146 | 0.2133 | 0.0301 | 0.2314 | 0.028* | |
| O144 | 0.06442 (13) | −0.37368 (13) | 0.21522 (8) | 0.0314 (4) | |
| C147 | 0.0924 (2) | −0.4879 (2) | 0.20332 (13) | 0.0343 (5) | |
| H14A | 0.0931 | −0.5117 | 0.1548 | 0.052* | |
| H14B | 0.0280 | −0.5529 | 0.2205 | 0.052* | |
| H14C | 0.1752 | −0.4775 | 0.2265 | 0.052* | |
| O15 | 0.46228 (13) | −0.01917 (13) | 0.31718 (7) | 0.0289 (3) | |
| N21 | 0.41570 (15) | 0.41471 (15) | 0.29199 (8) | 0.0206 (4) | |
| N22 | 0.45913 (15) | 0.49317 (15) | 0.24625 (8) | 0.0215 (4) | |
| C23 | 0.57028 (18) | 0.57217 (18) | 0.27160 (10) | 0.0192 (4) | |
| C23A | 0.60364 (18) | 0.54681 (18) | 0.33813 (10) | 0.0191 (4) | |
| C24 | 0.70349 (18) | 0.59396 (18) | 0.39119 (10) | 0.0197 (4) | |
| C24A | 0.69021 (18) | 0.52704 (18) | 0.44453 (10) | 0.0199 (4) | |
| C25 | 0.77463 (19) | 0.54600 (19) | 0.50847 (10) | 0.0237 (4) | |
| C25A | 0.71036 (18) | 0.44324 (19) | 0.54602 (10) | 0.0223 (4) | |
| C26 | 0.7481 (2) | 0.4161 (2) | 0.60764 (10) | 0.0258 (5) | |
| H26 | 0.8246 | 0.4667 | 0.6331 | 0.031* | |
| C27 | 0.6708 (2) | 0.3127 (2) | 0.63098 (11) | 0.0282 (5) | |
| H27 | 0.6943 | 0.2926 | 0.6732 | 0.034* | |
| C28 | 0.5592 (2) | 0.2381 (2) | 0.59319 (11) | 0.0270 (5) | |
| H28 | 0.5083 | 0.1670 | 0.6095 | 0.032* | |
| C29 | 0.52149 (19) | 0.26675 (19) | 0.53164 (10) | 0.0247 (5) | |
| H29 | 0.4449 | 0.2165 | 0.5061 | 0.030* | |
| C29A | 0.59773 (18) | 0.36952 (19) | 0.50863 (10) | 0.0213 (4) | |
| C29B | 0.58416 (18) | 0.42161 (18) | 0.44612 (10) | 0.0192 (4) | |
| N210 | 0.48715 (15) | 0.37700 (15) | 0.39927 (8) | 0.0201 (4) | |
| C210 | 0.50089 (18) | 0.44282 (18) | 0.34759 (10) | 0.0198 (4) | |
| C211 | 0.29240 (18) | 0.32774 (18) | 0.27826 (10) | 0.0203 (4) | |
| C212 | 0.22507 (18) | 0.27967 (19) | 0.32998 (10) | 0.0235 (4) | |
| H212 | 0.2638 | 0.2998 | 0.3751 | 0.028* | |
| C213 | 0.10145 (19) | 0.20230 (19) | 0.31581 (11) | 0.0260 (5) | |
| H213 | 0.0553 | 0.1695 | 0.3511 | 0.031* | |
| C214 | 0.04602 (18) | 0.17333 (18) | 0.25011 (11) | 0.0242 (4) | |
| Cl24 | −0.10857 (5) | 0.07462 (6) | 0.23252 (3) | 0.03698 (15) | |
| C215 | 0.11179 (19) | 0.21924 (19) | 0.19823 (11) | 0.0245 (5) | |
| H215 | 0.0725 | 0.1982 | 0.1533 | 0.029* | |
| C216 | 0.23593 (19) | 0.29643 (19) | 0.21189 (10) | 0.0230 (4) | |
| H216 | 0.2820 | 0.3277 | 0.1763 | 0.028* | |
| C231 | 0.63711 (18) | 0.66646 (19) | 0.22811 (10) | 0.0215 (4) | |
| C232 | 0.5705 (2) | 0.6280 (2) | 0.15604 (10) | 0.0257 (5) | |
| H23A | 0.5692 | 0.5434 | 0.1388 | 0.039* | |
| H23B | 0.6160 | 0.6853 | 0.1269 | 0.039* | |
| H23C | 0.4839 | 0.6309 | 0.1564 | 0.039* | |
| C233 | 0.6289 (2) | 0.79539 (19) | 0.25499 (11) | 0.0291 (5) | |
| H23D | 0.5404 | 0.7905 | 0.2573 | 0.044* | |
| H23E | 0.6667 | 0.8540 | 0.2247 | 0.044* | |
| H23F | 0.6745 | 0.8241 | 0.3001 | 0.044* | |
| C234 | 0.77494 (19) | 0.6702 (2) | 0.22475 (11) | 0.0325 (5) | |
| H23G | 0.8222 | 0.7052 | 0.2691 | 0.049* | |
| H23H | 0.8122 | 0.7222 | 0.1914 | 0.049* | |
| H23I | 0.7783 | 0.5858 | 0.2116 | 0.049* | |
| C241 | 0.81715 (18) | 0.70594 (18) | 0.39442 (10) | 0.0197 (4) | |
| C242 | 0.80933 (19) | 0.82528 (19) | 0.41020 (10) | 0.0243 (4) | |
| H242 | 0.7296 | 0.8363 | 0.4160 | 0.029* | |
| C243 | 0.9160 (2) | 0.92818 (19) | 0.41757 (11) | 0.0267 (5) | |
| H243 | 0.9092 | 1.0092 | 0.4282 | 0.032* | |
| C244 | 1.03293 (19) | 0.91287 (18) | 0.40936 (10) | 0.0231 (4) | |
| C245 | 1.04326 (19) | 0.79517 (19) | 0.39425 (10) | 0.0243 (5) | |
| H245 | 1.1231 | 0.7843 | 0.3886 | 0.029* | |
| C246 | 0.93508 (18) | 0.69277 (19) | 0.38738 (10) | 0.0234 (4) | |
| H246 | 0.9422 | 0.6117 | 0.3776 | 0.028* | |
| O244 | 1.13345 (13) | 1.02023 (13) | 0.41752 (8) | 0.0312 (4) | |
| C247 | 1.2541 (2) | 1.0084 (2) | 0.40632 (12) | 0.0349 (5) | |
| H24A | 1.2811 | 0.9672 | 0.4415 | 0.052* | |
| H24B | 1.3152 | 1.0911 | 0.4077 | 0.052* | |
| H24C | 1.2490 | 0.9589 | 0.3620 | 0.052* | |
| O25 | 0.87457 (13) | 0.62675 (14) | 0.52722 (7) | 0.0320 (4) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| N11 | 0.0199 (9) | 0.0224 (9) | 0.0183 (9) | 0.0003 (7) | −0.0036 (7) | 0.0034 (7) |
| N12 | 0.0190 (9) | 0.0236 (9) | 0.0222 (9) | 0.0026 (7) | −0.0034 (7) | 0.0015 (7) |
| C13 | 0.0198 (10) | 0.0216 (10) | 0.0205 (11) | 0.0062 (8) | −0.0003 (8) | 0.0033 (8) |
| C13A | 0.0178 (10) | 0.0186 (10) | 0.0220 (11) | 0.0040 (8) | 0.0004 (8) | 0.0003 (8) |
| C14 | 0.0192 (10) | 0.0208 (10) | 0.0241 (11) | 0.0068 (8) | 0.0032 (8) | 0.0034 (9) |
| C14A | 0.0179 (10) | 0.0218 (10) | 0.0228 (11) | 0.0045 (8) | 0.0013 (8) | 0.0046 (9) |
| C15 | 0.0196 (10) | 0.0229 (11) | 0.0253 (11) | 0.0082 (8) | 0.0038 (8) | 0.0064 (9) |
| C15A | 0.0214 (10) | 0.0226 (10) | 0.0231 (11) | 0.0074 (8) | 0.0021 (8) | 0.0031 (9) |
| C16 | 0.0293 (12) | 0.0284 (11) | 0.0234 (11) | 0.0108 (9) | 0.0027 (9) | 0.0077 (9) |
| C17 | 0.0304 (12) | 0.0322 (12) | 0.0226 (11) | 0.0136 (10) | −0.0042 (9) | 0.0037 (9) |
| C18 | 0.0262 (11) | 0.0270 (11) | 0.0257 (12) | 0.0069 (9) | −0.0045 (9) | 0.0011 (9) |
| C19 | 0.0223 (11) | 0.0228 (11) | 0.0241 (11) | 0.0052 (9) | −0.0004 (8) | 0.0041 (9) |
| C19A | 0.0212 (10) | 0.0229 (10) | 0.0186 (10) | 0.0071 (8) | 0.0003 (8) | 0.0014 (8) |
| C19B | 0.0211 (10) | 0.0194 (10) | 0.0208 (11) | 0.0061 (8) | 0.0003 (8) | 0.0025 (8) |
| N110 | 0.0212 (9) | 0.0224 (9) | 0.0183 (9) | 0.0042 (7) | −0.0008 (7) | 0.0035 (7) |
| C110 | 0.0202 (10) | 0.0217 (10) | 0.0194 (10) | 0.0043 (8) | 0.0003 (8) | 0.0040 (8) |
| C111 | 0.0237 (10) | 0.0183 (10) | 0.0198 (11) | 0.0045 (8) | 0.0016 (8) | 0.0028 (8) |
| C112 | 0.0266 (11) | 0.0304 (12) | 0.0245 (12) | 0.0052 (9) | −0.0030 (9) | 0.0104 (10) |
| C113 | 0.0251 (11) | 0.0284 (12) | 0.0275 (12) | 0.0025 (9) | −0.0006 (9) | 0.0077 (10) |
| C114 | 0.0264 (11) | 0.0200 (10) | 0.0255 (11) | 0.0037 (9) | 0.0065 (9) | 0.0041 (9) |
| Cl14 | 0.0327 (3) | 0.0294 (3) | 0.0399 (3) | 0.0013 (2) | 0.0065 (2) | 0.0147 (3) |
| C115 | 0.0315 (12) | 0.0291 (12) | 0.0216 (11) | 0.0067 (10) | 0.0005 (9) | 0.0096 (9) |
| C116 | 0.0242 (11) | 0.0265 (11) | 0.0221 (11) | 0.0048 (9) | −0.0013 (9) | 0.0039 (9) |
| C131 | 0.0194 (10) | 0.0253 (11) | 0.0220 (11) | 0.0040 (8) | −0.0027 (8) | 0.0016 (9) |
| C132 | 0.0285 (12) | 0.0373 (13) | 0.0224 (12) | −0.0003 (10) | −0.0064 (9) | 0.0027 (10) |
| C133 | 0.0272 (12) | 0.0278 (12) | 0.0363 (13) | 0.0037 (10) | −0.0036 (10) | −0.0042 (10) |
| C134 | 0.0201 (11) | 0.0367 (13) | 0.0274 (12) | 0.0047 (9) | −0.0030 (9) | 0.0020 (10) |
| C141 | 0.0192 (10) | 0.0230 (10) | 0.0198 (10) | 0.0041 (8) | −0.0014 (8) | 0.0067 (8) |
| C142 | 0.0199 (10) | 0.0272 (11) | 0.0296 (12) | 0.0068 (9) | 0.0042 (9) | 0.0042 (9) |
| C143 | 0.0250 (11) | 0.0204 (11) | 0.0344 (13) | 0.0062 (9) | 0.0022 (9) | 0.0007 (9) |
| C144 | 0.0194 (10) | 0.0214 (10) | 0.0259 (11) | 0.0009 (8) | −0.0021 (8) | 0.0081 (9) |
| C145 | 0.0193 (10) | 0.0280 (11) | 0.0268 (12) | 0.0066 (9) | 0.0038 (8) | 0.0071 (9) |
| C146 | 0.0231 (11) | 0.0225 (10) | 0.0244 (11) | 0.0064 (9) | 0.0012 (9) | 0.0037 (9) |
| O144 | 0.0259 (8) | 0.0223 (8) | 0.0430 (10) | 0.0015 (6) | 0.0058 (7) | 0.0074 (7) |
| C147 | 0.0367 (13) | 0.0217 (11) | 0.0416 (14) | 0.0034 (10) | 0.0030 (11) | 0.0087 (10) |
| O15 | 0.0267 (8) | 0.0287 (8) | 0.0304 (9) | 0.0044 (7) | 0.0023 (6) | 0.0122 (7) |
| N21 | 0.0183 (8) | 0.0229 (9) | 0.0181 (9) | 0.0016 (7) | −0.0006 (7) | 0.0073 (7) |
| N22 | 0.0218 (9) | 0.0232 (9) | 0.0188 (9) | 0.0044 (7) | 0.0019 (7) | 0.0066 (7) |
| C23 | 0.0186 (10) | 0.0205 (10) | 0.0181 (10) | 0.0056 (8) | 0.0002 (8) | 0.0035 (8) |
| C23A | 0.0190 (10) | 0.0195 (10) | 0.0191 (10) | 0.0055 (8) | 0.0026 (8) | 0.0046 (8) |
| C24 | 0.0189 (10) | 0.0207 (10) | 0.0195 (10) | 0.0067 (8) | 0.0017 (8) | 0.0026 (8) |
| C24A | 0.0165 (10) | 0.0235 (10) | 0.0185 (10) | 0.0047 (8) | −0.0007 (8) | 0.0032 (8) |
| C25 | 0.0208 (11) | 0.0289 (11) | 0.0215 (11) | 0.0077 (9) | 0.0019 (8) | 0.0053 (9) |
| C25A | 0.0207 (10) | 0.0278 (11) | 0.0204 (11) | 0.0097 (9) | 0.0023 (8) | 0.0052 (9) |
| C26 | 0.0246 (11) | 0.0347 (12) | 0.0206 (11) | 0.0122 (9) | −0.0002 (9) | 0.0075 (9) |
| C27 | 0.0322 (12) | 0.0369 (13) | 0.0213 (11) | 0.0165 (10) | 0.0020 (9) | 0.0114 (10) |
| C28 | 0.0282 (12) | 0.0285 (11) | 0.0274 (12) | 0.0097 (9) | 0.0059 (9) | 0.0121 (10) |
| C29 | 0.0232 (11) | 0.0290 (11) | 0.0225 (11) | 0.0081 (9) | 0.0007 (8) | 0.0071 (9) |
| C29A | 0.0208 (10) | 0.0255 (11) | 0.0195 (10) | 0.0093 (8) | 0.0016 (8) | 0.0054 (9) |
| C29B | 0.0192 (10) | 0.0224 (10) | 0.0181 (10) | 0.0084 (8) | 0.0022 (8) | 0.0054 (8) |
| N210 | 0.0194 (9) | 0.0221 (9) | 0.0181 (9) | 0.0047 (7) | 0.0001 (7) | 0.0055 (7) |
| C210 | 0.0185 (10) | 0.0217 (10) | 0.0188 (10) | 0.0055 (8) | −0.0003 (8) | 0.0040 (8) |
| C211 | 0.0183 (10) | 0.0190 (10) | 0.0220 (11) | 0.0036 (8) | 0.0001 (8) | 0.0039 (8) |
| C212 | 0.0217 (11) | 0.0261 (11) | 0.0197 (11) | 0.0034 (9) | 0.0002 (8) | 0.0038 (9) |
| C213 | 0.0242 (11) | 0.0262 (11) | 0.0266 (12) | 0.0047 (9) | 0.0055 (9) | 0.0062 (9) |
| C214 | 0.0181 (10) | 0.0190 (10) | 0.0313 (12) | 0.0025 (8) | −0.0015 (9) | −0.0009 (9) |
| Cl24 | 0.0207 (3) | 0.0382 (3) | 0.0403 (3) | −0.0038 (2) | −0.0004 (2) | −0.0031 (3) |
| C215 | 0.0228 (11) | 0.0252 (11) | 0.0227 (11) | 0.0050 (9) | −0.0036 (8) | 0.0026 (9) |
| C216 | 0.0235 (11) | 0.0243 (11) | 0.0196 (11) | 0.0044 (9) | 0.0021 (8) | 0.0055 (9) |
| C231 | 0.0192 (10) | 0.0254 (11) | 0.0174 (10) | 0.0025 (8) | −0.0002 (8) | 0.0064 (9) |
| C232 | 0.0254 (11) | 0.0303 (11) | 0.0188 (11) | 0.0039 (9) | 0.0010 (8) | 0.0071 (9) |
| C233 | 0.0362 (13) | 0.0218 (11) | 0.0246 (12) | 0.0011 (9) | −0.0012 (9) | 0.0081 (9) |
| C234 | 0.0203 (11) | 0.0493 (15) | 0.0263 (12) | 0.0055 (10) | 0.0033 (9) | 0.0133 (11) |
| C241 | 0.0184 (10) | 0.0237 (10) | 0.0154 (10) | 0.0037 (8) | −0.0008 (8) | 0.0051 (8) |
| C242 | 0.0218 (10) | 0.0274 (11) | 0.0245 (11) | 0.0082 (9) | 0.0036 (8) | 0.0052 (9) |
| C243 | 0.0299 (12) | 0.0202 (10) | 0.0288 (12) | 0.0061 (9) | 0.0049 (9) | 0.0030 (9) |
| C244 | 0.0235 (11) | 0.0211 (10) | 0.0190 (11) | −0.0014 (8) | 0.0014 (8) | 0.0039 (8) |
| C245 | 0.0181 (10) | 0.0271 (11) | 0.0260 (11) | 0.0045 (9) | 0.0002 (8) | 0.0052 (9) |
| C246 | 0.0212 (10) | 0.0225 (10) | 0.0255 (11) | 0.0061 (8) | −0.0005 (8) | 0.0028 (9) |
| O244 | 0.0246 (8) | 0.0243 (8) | 0.0379 (9) | −0.0018 (6) | 0.0048 (7) | 0.0022 (7) |
| C247 | 0.0215 (11) | 0.0364 (13) | 0.0374 (14) | −0.0035 (10) | −0.0005 (10) | 0.0045 (11) |
| O25 | 0.0230 (8) | 0.0412 (9) | 0.0239 (8) | −0.0017 (7) | −0.0051 (6) | 0.0083 (7) |
Geometric parameters (Å, º) top
| N11—C110 | 1.361 (3) | N21—C210 | 1.364 (2) |
| N11—N12 | 1.377 (2) | N21—N22 | 1.373 (2) |
| N11—C111 | 1.424 (2) | N21—C211 | 1.422 (2) |
| N12—C13 | 1.320 (3) | N22—C23 | 1.323 (2) |
| C13—C13A | 1.453 (3) | C23—C23A | 1.456 (3) |
| C13—C131 | 1.525 (3) | C23—C231 | 1.521 (3) |
| C13A—C110 | 1.420 (3) | C23A—C210 | 1.423 (3) |
| C13A—C14 | 1.424 (3) | C23A—C24 | 1.424 (3) |
| C14—C14A | 1.394 (3) | C24—C24A | 1.384 (3) |
| C14—C141 | 1.497 (3) | C24—C241 | 1.497 (3) |
| C14A—C19B | 1.420 (3) | C24A—C29B | 1.417 (3) |
| C14A—C15 | 1.492 (3) | C24A—C25 | 1.502 (3) |
| C15—O15 | 1.218 (2) | C25—O25 | 1.217 (2) |
| C15—C15A | 1.501 (3) | C25—C25A | 1.498 (3) |
| C15A—C16 | 1.382 (3) | C25A—C26 | 1.388 (3) |
| C15A—C19A | 1.393 (3) | C25A—C29A | 1.396 (3) |
| C16—C17 | 1.390 (3) | C26—C27 | 1.391 (3) |
| C16—H16 | 0.9500 | C26—H26 | 0.9500 |
| C17—C18 | 1.385 (3) | C27—C28 | 1.393 (3) |
| C17—H17 | 0.9500 | C27—H27 | 0.9500 |
| C18—C19 | 1.393 (3) | C28—C29 | 1.394 (3) |
| C18—H18 | 0.9500 | C28—H28 | 0.9500 |
| C19—C19A | 1.390 (3) | C29—C29A | 1.378 (3) |
| C19—H19 | 0.9500 | C29—H29 | 0.9500 |
| C19A—C19B | 1.478 (3) | C29A—C29B | 1.477 (3) |
| C19B—N110 | 1.319 (3) | C29B—N210 | 1.323 (2) |
| N110—C110 | 1.354 (2) | N210—C210 | 1.348 (2) |
| C111—C116 | 1.386 (3) | C211—C212 | 1.391 (3) |
| C111—C112 | 1.390 (3) | C211—C216 | 1.396 (3) |
| C112—C113 | 1.385 (3) | C212—C213 | 1.388 (3) |
| C112—H112 | 0.9500 | C212—H212 | 0.9500 |
| C113—C114 | 1.377 (3) | C213—C214 | 1.381 (3) |
| C113—H113 | 0.9500 | C213—H213 | 0.9500 |
| C114—C115 | 1.379 (3) | C214—C215 | 1.378 (3) |
| C114—Cl14 | 1.743 (2) | C214—Cl24 | 1.742 (2) |
| C115—C116 | 1.386 (3) | C215—C216 | 1.391 (3) |
| C115—H115 | 0.9500 | C215—H215 | 0.9500 |
| C116—H116 | 0.9500 | C216—H216 | 0.9500 |
| C131—C133 | 1.528 (3) | C231—C233 | 1.534 (3) |
| C131—C132 | 1.531 (3) | C231—C234 | 1.536 (3) |
| C131—C134 | 1.535 (3) | C231—C232 | 1.536 (3) |
| C132—H13A | 0.9800 | C232—H23A | 0.9800 |
| C132—H13B | 0.9800 | C232—H23B | 0.9800 |
| C132—H13C | 0.9800 | C232—H23C | 0.9800 |
| C133—H13D | 0.9800 | C233—H23D | 0.9800 |
| C133—H13E | 0.9800 | C233—H23E | 0.9800 |
| C133—H13F | 0.9800 | C233—H23F | 0.9800 |
| C134—H13G | 0.9800 | C234—H23G | 0.9800 |
| C134—H13H | 0.9800 | C234—H23H | 0.9800 |
| C134—H13I | 0.9800 | C234—H23I | 0.9800 |
| C141—C142 | 1.389 (3) | C241—C246 | 1.386 (3) |
| C141—C146 | 1.396 (3) | C241—C242 | 1.390 (3) |
| C142—C143 | 1.389 (3) | C242—C243 | 1.383 (3) |
| C142—H142 | 0.9500 | C242—H242 | 0.9500 |
| C143—C144 | 1.385 (3) | C243—C244 | 1.388 (3) |
| C143—H143 | 0.9500 | C243—H243 | 0.9500 |
| C144—O144 | 1.374 (2) | C244—O244 | 1.374 (2) |
| C144—C145 | 1.390 (3) | C244—C245 | 1.381 (3) |
| C145—C146 | 1.384 (3) | C245—C246 | 1.391 (3) |
| C145—H145 | 0.9500 | C245—H245 | 0.9500 |
| C146—H146 | 0.9500 | C246—H246 | 0.9500 |
| O144—C147 | 1.426 (3) | O244—C247 | 1.424 (3) |
| C147—H14A | 0.9800 | C247—H24A | 0.9800 |
| C147—H14B | 0.9800 | C247—H24B | 0.9800 |
| C147—H14C | 0.9800 | C247—H24C | 0.9800 |
| | | |
| C110—N11—N12 | 110.55 (16) | C210—N21—N22 | 110.44 (15) |
| C110—N11—C111 | 130.81 (17) | C210—N21—C211 | 130.66 (17) |
| N12—N11—C111 | 118.50 (16) | N22—N21—C211 | 118.70 (15) |
| C13—N12—N11 | 107.99 (16) | C23—N22—N21 | 108.42 (15) |
| N12—C13—C13A | 110.04 (17) | N22—C23—C23A | 109.68 (16) |
| N12—C13—C131 | 116.89 (17) | N22—C23—C231 | 117.16 (17) |
| C13A—C13—C131 | 133.04 (18) | C23A—C23—C231 | 133.15 (17) |
| C110—C13A—C14 | 117.14 (18) | C210—C23A—C24 | 117.15 (17) |
| C110—C13A—C13 | 103.88 (17) | C210—C23A—C23 | 103.91 (16) |
| C14—C13A—C13 | 138.97 (18) | C24—C23A—C23 | 138.94 (18) |
| C14A—C14—C13A | 114.81 (18) | C24A—C24—C23A | 114.57 (17) |
| C14A—C14—C141 | 117.70 (18) | C24A—C24—C241 | 118.63 (17) |
| C13A—C14—C141 | 127.46 (18) | C23A—C24—C241 | 126.80 (17) |
| C14—C14A—C19B | 121.66 (18) | C24—C24A—C29B | 122.18 (18) |
| C14—C14A—C15 | 130.32 (18) | C24—C24A—C25 | 130.48 (18) |
| C19B—C14A—C15 | 107.89 (17) | C29B—C24A—C25 | 107.34 (17) |
| O15—C15—C14A | 128.89 (19) | O25—C25—C25A | 125.94 (19) |
| O15—C15—C15A | 125.61 (19) | O25—C25—C24A | 128.48 (19) |
| C14A—C15—C15A | 105.49 (16) | C25A—C25—C24A | 105.58 (17) |
| C16—C15A—C19A | 121.49 (19) | C26—C25A—C29A | 121.10 (19) |
| C16—C15A—C15 | 129.36 (19) | C26—C25A—C25 | 129.67 (19) |
| C19A—C15A—C15 | 109.09 (17) | C29A—C25A—C25 | 109.22 (17) |
| C15A—C16—C17 | 117.9 (2) | C25A—C26—C27 | 118.1 (2) |
| C15A—C16—H16 | 121.1 | C25A—C26—H26 | 121.0 |
| C17—C16—H16 | 121.1 | C27—C26—H26 | 121.0 |
| C18—C17—C16 | 120.8 (2) | C26—C27—C28 | 120.79 (19) |
| C18—C17—H17 | 119.6 | C26—C27—H27 | 119.6 |
| C16—C17—H17 | 119.6 | C28—C27—H27 | 119.6 |
| C17—C18—C19 | 121.5 (2) | C27—C28—C29 | 120.7 (2) |
| C17—C18—H18 | 119.2 | C27—C28—H28 | 119.6 |
| C19—C18—H18 | 119.2 | C29—C28—H28 | 119.6 |
| C19A—C19—C18 | 117.48 (19) | C29A—C29—C28 | 118.5 (2) |
| C19A—C19—H19 | 121.3 | C29A—C29—H29 | 120.7 |
| C18—C19—H19 | 121.3 | C28—C29—H29 | 120.7 |
| C19—C19A—C15A | 120.79 (18) | C29—C29A—C25A | 120.78 (18) |
| C19—C19A—C19B | 130.56 (18) | C29—C29A—C29B | 130.93 (19) |
| C15A—C19A—C19B | 108.58 (17) | C25A—C29A—C29B | 108.29 (17) |
| N110—C19B—C14A | 125.64 (18) | N210—C29B—C24A | 125.44 (18) |
| N110—C19B—C19A | 125.34 (18) | N210—C29B—C29A | 125.00 (18) |
| C14A—C19B—C19A | 108.93 (17) | C24A—C29B—C29A | 109.56 (17) |
| C19B—N110—C110 | 112.05 (17) | C29B—N210—C210 | 111.92 (16) |
| N110—C110—N11 | 123.82 (17) | N210—C210—N21 | 123.80 (17) |
| N110—C110—C13A | 128.65 (18) | N210—C210—C23A | 128.63 (18) |
| N11—C110—C13A | 107.53 (17) | N21—C210—C23A | 107.52 (16) |
| C116—C111—C112 | 120.08 (19) | C212—C211—C216 | 119.84 (18) |
| C116—C111—N11 | 118.33 (18) | C212—C211—N21 | 121.24 (18) |
| C112—C111—N11 | 121.58 (18) | C216—C211—N21 | 118.81 (17) |
| C113—C112—C111 | 119.77 (19) | C213—C212—C211 | 120.10 (19) |
| C113—C112—H112 | 120.1 | C213—C212—H212 | 119.9 |
| C111—C112—H112 | 120.1 | C211—C212—H212 | 119.9 |
| C114—C113—C112 | 119.7 (2) | C214—C213—C212 | 119.54 (19) |
| C114—C113—H113 | 120.1 | C214—C213—H213 | 120.2 |
| C112—C113—H113 | 120.1 | C212—C213—H213 | 120.2 |
| C113—C114—C115 | 120.96 (19) | C215—C214—C213 | 121.07 (19) |
| C113—C114—Cl14 | 118.84 (16) | C215—C214—Cl24 | 119.60 (16) |
| C115—C114—Cl14 | 120.20 (16) | C213—C214—Cl24 | 119.31 (16) |
| C114—C115—C116 | 119.60 (19) | C214—C215—C216 | 119.80 (19) |
| C114—C115—H115 | 120.2 | C214—C215—H215 | 120.1 |
| C116—C115—H115 | 120.2 | C216—C215—H215 | 120.1 |
| C115—C116—C111 | 119.86 (19) | C215—C216—C211 | 119.62 (19) |
| C115—C116—H116 | 120.1 | C215—C216—H216 | 120.2 |
| C111—C116—H116 | 120.1 | C211—C216—H216 | 120.2 |
| C13—C131—C133 | 109.44 (16) | C23—C231—C233 | 110.22 (16) |
| C13—C131—C132 | 109.87 (17) | C23—C231—C234 | 111.08 (16) |
| C133—C131—C132 | 108.44 (18) | C233—C231—C234 | 110.89 (18) |
| C13—C131—C134 | 110.52 (17) | C23—C231—C232 | 108.83 (16) |
| C133—C131—C134 | 110.39 (18) | C233—C231—C232 | 107.78 (17) |
| C132—C131—C134 | 108.15 (17) | C234—C231—C232 | 107.94 (17) |
| C131—C132—H13A | 109.5 | C231—C232—H23A | 109.5 |
| C131—C132—H13B | 109.5 | C231—C232—H23B | 109.5 |
| H13A—C132—H13B | 109.5 | H23A—C232—H23B | 109.5 |
| C131—C132—H13C | 109.5 | C231—C232—H23C | 109.5 |
| H13A—C132—H13C | 109.5 | H23A—C232—H23C | 109.5 |
| H13B—C132—H13C | 109.5 | H23B—C232—H23C | 109.5 |
| C131—C133—H13D | 109.5 | C231—C233—H23D | 109.5 |
| C131—C133—H13E | 109.5 | C231—C233—H23E | 109.5 |
| H13D—C133—H13E | 109.5 | H23D—C233—H23E | 109.5 |
| C131—C133—H13F | 109.5 | C231—C233—H23F | 109.5 |
| H13D—C133—H13F | 109.5 | H23D—C233—H23F | 109.5 |
| H13E—C133—H13F | 109.5 | H23E—C233—H23F | 109.5 |
| C131—C134—H13G | 109.5 | C231—C234—H23G | 109.5 |
| C131—C134—H13H | 109.5 | C231—C234—H23H | 109.5 |
| H13G—C134—H13H | 109.5 | H23G—C234—H23H | 109.5 |
| C131—C134—H13I | 109.5 | C231—C234—H23I | 109.5 |
| H13G—C134—H13I | 109.5 | H23G—C234—H23I | 109.5 |
| H13H—C134—H13I | 109.5 | H23H—C234—H23I | 109.5 |
| C142—C141—C146 | 118.80 (18) | C246—C241—C242 | 118.14 (18) |
| C142—C141—C14 | 120.42 (18) | C246—C241—C24 | 120.51 (18) |
| C146—C141—C14 | 120.35 (18) | C242—C241—C24 | 121.13 (17) |
| C141—C142—C143 | 120.88 (19) | C243—C242—C241 | 120.95 (19) |
| C141—C142—H142 | 119.6 | C243—C242—H242 | 119.5 |
| C143—C142—H142 | 119.6 | C241—C242—H242 | 119.5 |
| C144—C143—C142 | 119.76 (19) | C242—C243—C244 | 119.92 (19) |
| C144—C143—H143 | 120.1 | C242—C243—H243 | 120.0 |
| C142—C143—H143 | 120.1 | C244—C243—H243 | 120.0 |
| O144—C144—C143 | 124.49 (19) | O244—C244—C245 | 123.93 (18) |
| O144—C144—C145 | 115.60 (18) | O244—C244—C243 | 115.86 (18) |
| C143—C144—C145 | 119.91 (18) | C245—C244—C243 | 120.21 (18) |
| C146—C145—C144 | 120.06 (19) | C244—C245—C246 | 119.07 (19) |
| C146—C145—H145 | 120.0 | C244—C245—H245 | 120.5 |
| C144—C145—H145 | 120.0 | C246—C245—H245 | 120.5 |
| C145—C146—C141 | 120.53 (19) | C241—C246—C245 | 121.71 (19) |
| C145—C146—H146 | 119.7 | C241—C246—H246 | 119.1 |
| C141—C146—H146 | 119.7 | C245—C246—H246 | 119.1 |
| C144—O144—C147 | 116.50 (16) | C244—O244—C247 | 117.41 (17) |
| O144—C147—H14A | 109.5 | O244—C247—H24A | 109.5 |
| O144—C147—H14B | 109.5 | O244—C247—H24B | 109.5 |
| H14A—C147—H14B | 109.5 | H24A—C247—H24B | 109.5 |
| O144—C147—H14C | 109.5 | O244—C247—H24C | 109.5 |
| H14A—C147—H14C | 109.5 | H24A—C247—H24C | 109.5 |
| H14B—C147—H14C | 109.5 | H24B—C247—H24C | 109.5 |
| | | |
| C110—N11—N12—C13 | −1.0 (2) | C210—N21—N22—C23 | −0.6 (2) |
| C111—N11—N12—C13 | 175.01 (17) | C211—N21—N22—C23 | 174.83 (17) |
| N11—N12—C13—C13A | 0.1 (2) | N21—N22—C23—C23A | −0.5 (2) |
| N11—N12—C13—C131 | −178.16 (16) | N21—N22—C23—C231 | 178.74 (16) |
| N12—C13—C13A—C110 | 0.7 (2) | N22—C23—C23A—C210 | 1.3 (2) |
| C131—C13—C13A—C110 | 178.7 (2) | C231—C23—C23A—C210 | −177.8 (2) |
| N12—C13—C13A—C14 | −177.7 (2) | N22—C23—C23A—C24 | −179.2 (2) |
| C131—C13—C13A—C14 | 0.2 (4) | C231—C23—C23A—C24 | 1.7 (4) |
| C110—C13A—C14—C14A | 2.4 (3) | C210—C23A—C24—C24A | 3.3 (3) |
| C13—C13A—C14—C14A | −179.3 (2) | C23—C23A—C24—C24A | −176.2 (2) |
| C110—C13A—C14—C141 | −175.69 (19) | C210—C23A—C24—C241 | −176.39 (18) |
| C13—C13A—C14—C141 | 2.6 (4) | C23—C23A—C24—C241 | 4.2 (4) |
| C13A—C14—C14A—C19B | −1.3 (3) | C23A—C24—C24A—C29B | −1.1 (3) |
| C141—C14—C14A—C19B | 177.01 (18) | C241—C24—C24A—C29B | 178.58 (18) |
| C13A—C14—C14A—C15 | −176.64 (19) | C23A—C24—C24A—C25 | 179.28 (19) |
| C141—C14—C14A—C15 | 1.7 (3) | C241—C24—C24A—C25 | −1.0 (3) |
| C14—C14A—C15—O15 | −3.4 (4) | C24—C24A—C25—O25 | 0.4 (4) |
| C19B—C14A—C15—O15 | −179.2 (2) | C29B—C24A—C25—O25 | −179.2 (2) |
| C14—C14A—C15—C15A | 175.8 (2) | C24—C24A—C25—C25A | −179.6 (2) |
| C19B—C14A—C15—C15A | 0.0 (2) | C29B—C24A—C25—C25A | 0.8 (2) |
| O15—C15—C15A—C16 | 2.6 (3) | O25—C25—C25A—C26 | −0.8 (4) |
| C14A—C15—C15A—C16 | −176.7 (2) | C24A—C25—C25A—C26 | 179.2 (2) |
| O15—C15—C15A—C19A | 179.96 (19) | O25—C25—C25A—C29A | 179.8 (2) |
| C14A—C15—C15A—C19A | 0.7 (2) | C24A—C25—C25A—C29A | −0.2 (2) |
| C19A—C15A—C16—C17 | −0.1 (3) | C29A—C25A—C26—C27 | 0.4 (3) |
| C15—C15A—C16—C17 | 177.0 (2) | C25—C25A—C26—C27 | −179.0 (2) |
| C15A—C16—C17—C18 | 0.7 (3) | C25A—C26—C27—C28 | 0.5 (3) |
| C16—C17—C18—C19 | −0.4 (3) | C26—C27—C28—C29 | −1.1 (3) |
| C17—C18—C19—C19A | −0.5 (3) | C27—C28—C29—C29A | 0.8 (3) |
| C18—C19—C19A—C15A | 1.0 (3) | C28—C29—C29A—C25A | 0.1 (3) |
| C18—C19—C19A—C19B | −175.6 (2) | C28—C29—C29A—C29B | 179.1 (2) |
| C16—C15A—C19A—C19 | −0.8 (3) | C26—C25A—C29A—C29 | −0.7 (3) |
| C15—C15A—C19A—C19 | −178.43 (18) | C25—C25A—C29A—C29 | 178.79 (18) |
| C16—C15A—C19A—C19B | 176.52 (18) | C26—C25A—C29A—C29B | −179.91 (18) |
| C15—C15A—C19A—C19B | −1.1 (2) | C25—C25A—C29A—C29B | −0.4 (2) |
| C14—C14A—C19B—N110 | −0.2 (3) | C24—C24A—C29B—N210 | −1.3 (3) |
| C15—C14A—C19B—N110 | 176.03 (18) | C25—C24A—C29B—N210 | 178.38 (18) |
| C14—C14A—C19B—C19A | −176.89 (18) | C24—C24A—C29B—C29A | 179.27 (18) |
| C15—C14A—C19B—C19A | −0.6 (2) | C25—C24A—C29B—C29A | −1.0 (2) |
| C19—C19A—C19B—N110 | 1.4 (3) | C29—C29A—C29B—N210 | 2.4 (3) |
| C15A—C19A—C19B—N110 | −175.57 (19) | C25A—C29A—C29B—N210 | −178.51 (19) |
| C19—C19A—C19B—C14A | 178.1 (2) | C29—C29A—C29B—C24A | −178.2 (2) |
| C15A—C19A—C19B—C14A | 1.1 (2) | C25A—C29A—C29B—C24A | 0.9 (2) |
| C14A—C19B—N110—C110 | 0.5 (3) | C24A—C29B—N210—C210 | 1.1 (3) |
| C19A—C19B—N110—C110 | 176.61 (18) | C29A—C29B—N210—C210 | −179.52 (18) |
| C19B—N110—C110—N11 | −179.07 (18) | C29B—N210—C210—N21 | 178.46 (18) |
| C19B—N110—C110—C13A | 1.0 (3) | C29B—N210—C210—C23A | 1.5 (3) |
| N12—N11—C110—N110 | −178.47 (18) | N22—N21—C210—N210 | −176.10 (17) |
| C111—N11—C110—N110 | 6.1 (3) | C211—N21—C210—N210 | 9.2 (3) |
| N12—N11—C110—C13A | 1.5 (2) | N22—N21—C210—C23A | 1.4 (2) |
| C111—N11—C110—C13A | −173.90 (19) | C211—N21—C210—C23A | −173.28 (19) |
| C14—C13A—C110—N110 | −2.5 (3) | C24—C23A—C210—N210 | −3.9 (3) |
| C13—C13A—C110—N110 | 178.6 (2) | C23—C23A—C210—N210 | 175.75 (19) |
| C14—C13A—C110—N11 | 177.51 (17) | C24—C23A—C210—N21 | 178.80 (17) |
| C13—C13A—C110—N11 | −1.3 (2) | C23—C23A—C210—N21 | −1.6 (2) |
| C110—N11—C111—C116 | −161.9 (2) | C210—N21—C211—C212 | 18.5 (3) |
| N12—N11—C111—C116 | 23.0 (3) | N22—N21—C211—C212 | −155.80 (18) |
| C110—N11—C111—C112 | 18.5 (3) | C210—N21—C211—C216 | −165.4 (2) |
| N12—N11—C111—C112 | −156.56 (19) | N22—N21—C211—C216 | 20.3 (3) |
| C116—C111—C112—C113 | 0.5 (3) | C216—C211—C212—C213 | −1.0 (3) |
| N11—C111—C112—C113 | −179.94 (19) | N21—C211—C212—C213 | 175.11 (18) |
| C111—C112—C113—C114 | −0.6 (3) | C211—C212—C213—C214 | 0.1 (3) |
| C112—C113—C114—C115 | 0.2 (3) | C212—C213—C214—C215 | 0.5 (3) |
| C112—C113—C114—Cl14 | −179.43 (17) | C212—C213—C214—Cl24 | 179.22 (16) |
| C113—C114—C115—C116 | 0.3 (3) | C213—C214—C215—C216 | −0.2 (3) |
| Cl14—C114—C115—C116 | 179.91 (16) | Cl24—C214—C215—C216 | −178.94 (16) |
| C114—C115—C116—C111 | −0.4 (3) | C214—C215—C216—C211 | −0.7 (3) |
| C112—C111—C116—C115 | 0.0 (3) | C212—C211—C216—C215 | 1.3 (3) |
| N11—C111—C116—C115 | −179.59 (19) | N21—C211—C216—C215 | −174.93 (18) |
| N12—C13—C131—C133 | 108.3 (2) | N22—C23—C231—C233 | 105.2 (2) |
| C13A—C13—C131—C133 | −69.6 (3) | C23A—C23—C231—C233 | −75.8 (3) |
| N12—C13—C131—C132 | −10.7 (2) | N22—C23—C231—C234 | −131.46 (19) |
| C13A—C13—C131—C132 | 171.5 (2) | C23A—C23—C231—C234 | 47.5 (3) |
| N12—C13—C131—C134 | −129.98 (19) | N22—C23—C231—C232 | −12.8 (2) |
| C13A—C13—C131—C134 | 52.2 (3) | C23A—C23—C231—C232 | 166.2 (2) |
| C14A—C14—C141—C142 | −93.5 (2) | C24A—C24—C241—C246 | 71.6 (3) |
| C13A—C14—C141—C142 | 84.6 (3) | C23A—C24—C241—C246 | −108.7 (2) |
| C14A—C14—C141—C146 | 78.8 (2) | C24A—C24—C241—C242 | −102.8 (2) |
| C13A—C14—C141—C146 | −103.1 (2) | C23A—C24—C241—C242 | 76.8 (3) |
| C146—C141—C142—C143 | −0.8 (3) | C246—C241—C242—C243 | 1.1 (3) |
| C14—C141—C142—C143 | 171.57 (19) | C24—C241—C242—C243 | 175.63 (19) |
| C141—C142—C143—C144 | 0.0 (3) | C241—C242—C243—C244 | −0.2 (3) |
| C142—C143—C144—O144 | −177.4 (2) | C242—C243—C244—O244 | 179.91 (19) |
| C142—C143—C144—C145 | 1.8 (3) | C242—C243—C244—C245 | −0.4 (3) |
| O144—C144—C145—C146 | 176.40 (18) | O244—C244—C245—C246 | 179.73 (19) |
| C143—C144—C145—C146 | −2.8 (3) | C243—C244—C245—C246 | 0.1 (3) |
| C144—C145—C146—C141 | 2.0 (3) | C242—C241—C246—C245 | −1.4 (3) |
| C142—C141—C146—C145 | −0.2 (3) | C24—C241—C246—C245 | −176.03 (19) |
| C14—C141—C146—C145 | −172.61 (19) | C244—C245—C246—C241 | 0.9 (3) |
| C143—C144—O144—C147 | 10.7 (3) | C245—C244—O244—C247 | 3.2 (3) |
| C145—C144—O144—C147 | −168.46 (19) | C243—C244—O244—C247 | −177.08 (19) |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| C18—H18···O25i | 0.95 | 2.50 | 3.151 (3) | 126 |
| C28—H28···O15ii | 0.95 | 2.46 | 3.226 (3) | 138 |
| C146—H146···Cg | 0.95 | 2.71 | 3.655 (2) | 172 |
| Symmetry codes: (i) −x+2, −y+1, −z+1; (ii) −x+1, −y, −z+1. |
Experimental details
| | (I) | (II) |
| Crystal data |
| Chemical formula | C29H22ClN3O | C30H24ClN3O2 |
| Mr | 463.95 | 493.97 |
| Crystal system, space group | Triclinic, P1 | Triclinic, P1 |
| Temperature (K) | 120 | 120 |
| a, b, c (Å) | 8.1014 (5), 11.3554 (10), 13.5762 (11) | 11.1911 (9), 11.4258 (8), 20.095 (2) |
| α, β, γ (°) | 102.755 (7), 90.013 (8), 108.268 (7) | 97.411 (6), 93.272 (7), 107.635 (6) |
| V (Å3) | 1153.47 (16) | 2415.7 (4) |
| Z | 2 | 4 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 0.19 | 0.19 |
| Crystal size (mm) | 0.43 × 0.22 × 0.16 | 0.42 × 0.30 × 0.29 |
| |
| Data collection |
| Diffractometer | Bruker–Nonius KappaCCD
diffractometer | Bruker–Nonius KappaCCD
diffractometer |
| Absorption correction | Multi-scan
(SADABS; Sheldrick, 2003) | Multi-scan
(SADABS; Sheldrick, 2003) |
| Tmin, Tmax | 0.921, 0.970 | 0.924, 0.946 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 29371, 5310, 4065 | 63410, 11085, 7328 |
| Rint | 0.045 | 0.061 |
| (sin θ/λ)max (Å−1) | 0.650 | 0.650 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.043, 0.098, 1.06 | 0.048, 0.115, 1.06 |
| No. of reflections | 5310 | 11085 |
| No. of parameters | 310 | 657 |
| H-atom treatment | H-atom parameters constrained | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 0.22, −0.39 | 0.34, −0.37 |
Selected torsional angles (°) for compounds (I) and (II) top| Parameter | | (I) | (II) | (II) |
| | x = nul | x = 1 | x = 2 |
| Nx2—Nx1—Cx11—Cx12 | | -170.54 (14) | -156.56 (19) | -155.80 (18) |
| Nx2—Cx3—Cx31—Cx32 | | -12.9 (2) | -10.7 (2) | -12.8 (2) |
| Nx2—Cx3—Cx31—Cx33 | | 105.68 (17) | 108.3 (2) | 106.2 (2) |
| Nx2—Cx3—Cx31—Cx34 | | -131.81 (16) | -129.98 (19) | -131.46 (19) |
| Cx3A—Cx4—Cx41—Cx42 | | 78.8 (2) | 84.6 (3) | 76.8 (3) |
| Cx43—Cx44—Ox44—Cx47 | | | 10.7 (3) | -177.08 (19) |
Hydrogen bond parameters (Å, °) for compounds (I) and (II). top| Compound | D—H···A | | D—H | H···A | D···A | D—H···A |
| (I) | C44—H44···O5i | | 0.95 | 2.59 | 3.232 (2) | 125 |
| | | | | | |
| (II) | C146—H146···Cga | | 0.95 | 2.71 | 3.655 (2) | 172 |
| C18—H18···O25ii | | 0.95 | 2.50 | 3.151 (3) | 126 |
| C28—H28···O15iii | | 0.95 | 2.46 | 3.226 (3) | 138 |
| Note: (a) Cg represents the centroid of the C211–C216 ring.
Symmetry codes: (i) -x+1, -y+1, -z+2; (ii) -x+2, -y+1, -z+1);
(iii) -x+1, -y, -z+1. |
 O hydrogen bonds to form R22(18) dimers, which are themselves linked into a chain by a π–π stacking interaction between inversion-related pairs of molecules. In 3-tert-butyl-1-(4-chlorophenyl)-4-(4-methoxyphenyl)indeno[1,2-b]pyrazolo[4,3-e]pyridin-5(1H)-one, C30H24ClN3O2, (II), which crystallizes in the space group P
O hydrogen bonds to form R22(18) dimers, which are themselves linked into a chain by a π–π stacking interaction between inversion-related pairs of molecules. In 3-tert-butyl-1-(4-chlorophenyl)-4-(4-methoxyphenyl)indeno[1,2-b]pyrazolo[4,3-e]pyridin-5(1H)-one, C30H24ClN3O2, (II), which crystallizes in the space group P O and C—H
O and C—H π(arene) hydrogen bonds links the molecules into chains of rings, which are further linked into sheets by a π–π stacking interaction.
π(arene) hydrogen bonds links the molecules into chains of rings, which are further linked into sheets by a π–π stacking interaction.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2011/12/00/fg3236/fg3236fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2011/12/00/fg3236/fg3236fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2011/12/00/fg3236/fg3236fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/c/issues/2011/12/00/fg3236/fg3236fig4thm.gif)
![[Figure 5]](http://journals.iucr.org/c/issues/2011/12/00/fg3236/fg3236fig5thm.gif)




Pyrazolo[3,4-b]pyridines are fused heterocyclic compounds of considerable interest for drug development because of the wide range of biological activities that they exhibit (Quiroga et al., 2005, 2007). Independently, indenopyridine compounds have shown potential activity as antioxidant, antihistamine and antidepressant agents (de Almeida et al., 1976; Strunz & Findlay, 1985; Earl et al., 1998; Padwa et al., 2000; Peters et al., 2004; Evdokimov et al., 2011). As part of a programme whose aim is to prepare new heterocyclic derivatives combining both these fused ring systems, we report here the structure of 3-tert-butyl-1-(4-chlorophenyl)-4–phenylindeno[1,2-b]pyrazolo[4,3-e]pyridin-5(1H)-one, (I), and the 4-(4-methoxyphenyl)- derivative, (II) (Figs. 1 and 2), which were prepared in good yield by means of a tricomponent reaction between indan-1,3-dione, the corresponding substituted benzaldehyde and 5-amino-3-tert-butyl-1-(4-chlorophenyl)-1H-pyrazole (see Scheme), which was induced by smooth microwave irradiation using water as solvent and triethylamine as catalyst.
Compounds (I) and (II) both crystallize in the space group P1, but with Z' values of 1 and 2, respectively (Figs. 1 and 2); in (II) it will be convenient to refer to the molecules having x = 1 and 2 as type 1 and type 2 molecules, respectively. For each of the independent molecules, the conformations can be defined in terms of the orientations of the tert-butyl group and of the two pendent aryl substituents relative to the fused ring system, along with the orientation of the methoxy groups in (II).
None of the molecules exhibits any internal symmetry and hence they are all conformationally chiral; however, in each compound the centrosymmetric space group accommodates equal numbers of both conformational enantiomers. In each of the independent molecules, the fused ring system is essentially planar. In compound (I), the maximum deviations from the mean plane through the fused ring system are 0.060 (2) Å for atom C3 and 0.045 (2) Å for atom C5A, displaced to opposite sides of the plane; for the type 1 molecule in compound (II), the maximum deviations are 0.118 (2) Å for atom C17 and 0.086 (2) Å for atom C19B, again displaced to opposite sides of the plane; and in the type 2 molecule of compound (II), the maximum deviations are 0.086 (2) Å for atom C23 and 0.057 (2) Å for atom C28, this time with both displaced to the same side of the mean plane. Amongst the three independent molecules, no obvious pattern for these displacements can be recognized, so that the displacements are not regarded as chemically or structurally significant.
The corresponding bond distances within the fused ring systems are very similar, and they are consistent with aromatic delocalization in the fused aryl ring, and with delocalization in the pyridine ring accompanied by strong bond fixation in the pyrazole ring, as typically observed in pyrazolopyridine derivatives (Low et al., 2007; Quiroga et al., 2009, 2010; Insuasty et al., 2010).
The key torsion angles (Table 1) confirm that the two independent molecules within the selected asymmetric unit of compound (II) have the same conformation, which is the same as that for the selected asymmetric unit in compound (I). The corresponding values for each of the torsion angles amongst the three independent molecular species are remarkably similar. In each case, one of the methyl C atoms of the tert-butyl group, designated C32 in compound (I) and C132 and C232 in the two independent molecules of compound (II) (Figs. 1 and 2), lies close to, but not exactly in, the plane of the adjacent pyrazole ring. The deviations of this atom from the pyrazole plane are 0.388 (2) Å for atom C32 in compound (I), and 0.210 (2) Å and 0.386 (2) Å for atoms C132 and C232, respectively, in compound (II). Curiously, however, the two independent molecules in compound (II) exhibit different orientations for their methoxy groups (Table 1 and Fig. 2), and this alone suffices to preclude the possibility of any additional crystallographic symmetry.
Despite their close similarity in both constitution and conformation, the molecules of compounds (I) and (II) show different patterns of supramolecular aggregation, leading to arrays which are one- and two-dimensional, respectively.
In the crystal structure of (I), inversion-related pairs of molecules are linked by rather long and weak C—H···O hydrogen bonds (Table 2) into centrosymmetric R22(18) (Bernstein et al., 1995) dimers, and these hydrogen-bonded dimers are linked into chains by a single π–π stacking interaction. The planes of the pyridine ring in the molecule at (x, y, z) and the fused aryl ring in the molecule at (-x+1, -y+1, -z+1) make a dihedral angle of only 1.2 (2)°; the corresponding ring-centroid separation is 3.573 (2) Å and the interplanar spacing is ca 3.36 Å, giving a ring-centroid offset of ca 1.22 Å. The effect of this π-stacking interaction is to link the hydrogen-bonded dimers into a chain running parallel to the [001] direction, with the hydrogen-bonded rings centred at (1/2, 1/2, n), where n represents an integer, alternating with the π-stacking interactions across (1/2, 1/2, n + 1/2), where n again represents an integer (Fig. 3). It is notable that neither of the pendent aryl rings, viz. C11–C16 and C41–C46, participates in any π–π stacking interactions and that, despite the number of aromatic rings present, there are no C—H···π hydrogen bonds in the structure of (I).
The direction-specific intermolecular interactions in the crystal structure of compound (II) differ from those in the structure of compound (I) in several respects: firstly, Z' = 2 in compound (II); secondly, there is a C—H···π(arene) hydrogen bond present; and thirdly, this hydrogen bond involves both types of pendent aryl ring, with the 4-methoxyphenyl ring in the type 1 molecule providing the donor and the 4-chlorophenyl ring in the type 2 molecule acting as the acceptor.
The single, fairly short C—H···π(arene) hydrogen bond (Table 2) links the two independent molecules in the selected asymmetric unit. Two independent C—H···O hydrogen bonds, each linking inversion-related pairs of molecules into R22(14) rings, thus generate, in combination with the C—H···π(arene) hydrogen bond, a chain of rings running parallel to the [110] direction (Fig. 4). There are two independent π–π stacking interactions present in the structure of compound (II). The first of these occurs within the selected asymmetric unit, where the fused aryl ring of the type 1 molecule and the pyridine ring of the the type 2 molecule make a dihedral angle of 2.5 (2)°; the ring-centroid separation is 3.701 (2) Å and the interplanar separation is ca 3.53 Å, corresponding to a ring-centroid offset of ca 1.11 Å. Hence this interaction provides some modest reinforcement to the action of the C—H···π(arene) hydrogen bond.
Only type 2 molecules of compound (II) are involved in the second type of stacking interaction. The pyridine ring of the type 2 molecule at (x, y, z) and the fused aryl ring of the type 2 molecule at (1 - x, 1 - y, 1 - z) make a dihedral angle of 2.2 (2)°, with a ring-centroid separation off 3.743 (2) Å, an interplanar spacing of ca 3.56 Å, and a ring-centroid offset of ca 1.16 Å (Fig. 5). The effect of this interaction is to link the hydrogen-bonded chains into a sheet parallel to (001). It may be noted here that the overall behaviour of this π-stacking interaction of the type 2 molecules of compound (II) closely corresponds to the π-stacking interaction in compound (I), although the metrics of the two interaction differ somewhat, not surprisingly in view of the presence of an additional molecular type in compound (II).