Buy article online - an online subscription or single-article purchase is required to access this article.

The title mononuclear Co
II complex, [Co(C
5H
7N
6)
2(C
14H
8O
5)
2(H
2O)
2]·2H
2O, has been synthesized and its crystal structure determined by X-ray diffraction. The complex crystallizes in the triclinic space group
P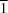
, with one formula unit per cell (
Z = 1 and
Z′ =
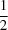
). It consists of a mononuclear unit with the Co
II ion on an inversion centre coordinated by two 2,6-diamino-7
H-purin-1-ium cations, two 4,4′-oxydibenzoate anions (in a nonbridging κ
O-monodentate coordination mode, which is less common for the anion in its Co
II complexes) and two water molecules, defining an octahedral environment around the metal atom. There is a rich assortment of nonbonding interactions, among which a strong N
+—H

O
− bridge, with a short N

O distance of 2.5272 (18) Å, stands out, with the H atom ostensibly displaced away from its expected position at the donor side, towards the acceptor. The complex molecules assemble into a three-dimensional hydrogen-bonded network. A variable-temperature magnetic study between 2 and 300 K reveals an orbital contribution to the magnetic moment and a weak antiferromagnetic interaction between Co
II centres as the temperature decreases. The model leads to the following values:
A (crystal field strength) = 1.81, λ (spin-orbit coupling) = −59.9 cm
−1,
g (Landé factor) = 2.58 and
zJ (exchange coupling) = −0.5 cm
−1.
Supporting information
CCDC reference: 1587307
Data collection: SMART (Bruker, 2001); cell refinement: SAINT (Bruker, 2002); data reduction: SAINT (Bruker, 2002); program(s) used to solve structure: SHELXS97 (Sheldrick, 2008); program(s) used to refine structure: SHELXL2014 (Sheldrick, 2015); molecular graphics: SHELXTL (Sheldrick, 2008); software used to prepare material for publication: SHELXTL (Sheldrick, 2008.
Diaquabis(2,6-diamino-7
H-purin-1-ium-
κN9)bis(4,4'-oxydibenzoato-
κO)cobalt(II) dihydrate
top
Crystal data top
| [Co(C5H7N6)2(C14H8O5)2(H2O)2]·2H2O | Z = 1 |
| Mr = 945.73 | F(000) = 489 |
| Triclinic, P1 | Dx = 1.599 Mg m−3 |
| a = 7.3560 (9) Å | Mo Kα radiation, λ = 0.71073 Å |
| b = 11.5890 (15) Å | Cell parameters from 4255 reflections |
| c = 12.0655 (15) Å | θ = 2.8–27.4° |
| α = 73.757 (2)° | µ = 0.53 mm−1 |
| β = 88.232 (2)° | T = 150 K |
| γ = 83.876 (2)° | Block, pink |
| V = 981.9 (2) Å3 | 0.37 × 0.28 × 0.17 mm |
Data collection top
Bruker SMART CCD area detector
diffractometer | 3861 reflections with I > 2σ(I) |
| CCD rotation images, thin slices scans | Rint = 0.014 |
Absorption correction: multi-scan
(SADABS; Sheldrick, 2008) | θmax = 27.8°, θmin = 1.8° |
| Tmin = 0.85, Tmax = 0.90 | h = −9→9 |
| 8230 measured reflections | k = −14→14 |
| 4207 independent reflections | l = −15→15 |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | Hydrogen site location: mixed |
| R[F2 > 2σ(F2)] = 0.034 | H atoms treated by a mixture of independent and constrained refinement |
| wR(F2) = 0.087 | w = 1/[σ2(Fo2) + (0.0446P)2 + 0.4856P]
where P = (Fo2 + 2Fc2)/3 |
| S = 1.03 | (Δ/σ)max = 0.001 |
| 4207 reflections | Δρmax = 0.37 e Å−3 |
| 326 parameters | Δρmin = −0.23 e Å−3 |
Special details top
Geometry. All esds (except the esd in the dihedral angle between two l.s. planes)
are estimated using the full covariance matrix. The cell esds are taken
into account individually in the estimation of esds in distances, angles
and torsion angles; correlations between esds in cell parameters are only
used when they are defined by crystal symmetry. An approximate (isotropic)
treatment of cell esds is used for estimating esds involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Co1 | 0.500000 | 0.500000 | 0.500000 | 0.01676 (9) | |
| N11 | 0.47146 (18) | 0.62667 (12) | 0.33268 (11) | 0.0207 (3) | |
| N21 | 0.44483 (19) | 0.81452 (12) | 0.21500 (12) | 0.0220 (3) | |
| H21 | 0.452 (3) | 0.8907 (19) | 0.1941 (17) | 0.026* | |
| N31 | 0.33331 (19) | 0.77944 (12) | 0.04178 (12) | 0.0232 (3) | |
| N41 | 0.2233 (2) | 0.70938 (15) | −0.10148 (14) | 0.0326 (4) | |
| H41A | 0.216 (3) | 0.783 (2) | −0.145 (2) | 0.039* | |
| H41B | 0.186 (3) | 0.650 (2) | −0.1236 (19) | 0.039* | |
| N51 | 0.30455 (19) | 0.56951 (12) | 0.06912 (12) | 0.0219 (3) | |
| H51 | 0.254 (4) | 0.503 (3) | 0.020 (3) | 0.086 (10)* | |
| N61 | 0.3814 (2) | 0.41629 (13) | 0.23437 (13) | 0.0238 (3) | |
| H61A | 0.358 (3) | 0.3686 (19) | 0.1978 (18) | 0.029* | |
| H61B | 0.442 (3) | 0.3890 (18) | 0.2970 (18) | 0.029* | |
| C11 | 0.4892 (2) | 0.74191 (14) | 0.32141 (14) | 0.0226 (3) | |
| H11 | 0.529165 | 0.770647 | 0.382028 | 0.027* | |
| C21 | 0.3935 (2) | 0.74306 (14) | 0.15154 (14) | 0.0198 (3) | |
| C31 | 0.2889 (2) | 0.68775 (15) | 0.00598 (14) | 0.0232 (3) | |
| C41 | 0.3682 (2) | 0.53356 (14) | 0.17898 (14) | 0.0200 (3) | |
| C51 | 0.4125 (2) | 0.62581 (14) | 0.22460 (13) | 0.0188 (3) | |
| O12 | 0.37407 (15) | 0.63681 (10) | 0.57105 (9) | 0.0204 (2) | |
| O22 | 0.08856 (16) | 0.58410 (10) | 0.59471 (11) | 0.0248 (3) | |
| O32 | 0.2025 (2) | 1.44220 (11) | 0.94800 (11) | 0.0336 (3) | |
| O42 | 0.26406 (17) | 1.27054 (11) | 1.08592 (10) | 0.0278 (3) | |
| O52 | −0.05963 (17) | 1.10192 (11) | 0.67491 (11) | 0.0285 (3) | |
| C12 | 0.2043 (2) | 0.65636 (14) | 0.59254 (13) | 0.0186 (3) | |
| C22 | 0.1403 (2) | 0.77421 (14) | 0.61705 (13) | 0.0191 (3) | |
| C32 | 0.2609 (2) | 0.85769 (15) | 0.62081 (14) | 0.0232 (3) | |
| H32 | 0.387501 | 0.840318 | 0.607484 | 0.028* | |
| C42 | 0.1987 (2) | 0.96598 (15) | 0.64375 (15) | 0.0257 (4) | |
| H42 | 0.282249 | 1.021996 | 0.647249 | 0.031* | |
| C52 | 0.0134 (2) | 0.99162 (14) | 0.66152 (14) | 0.0224 (3) | |
| C62 | −0.1086 (2) | 0.91020 (16) | 0.65789 (16) | 0.0268 (4) | |
| H62 | −0.235331 | 0.928434 | 0.670017 | 0.032* | |
| C72 | −0.0448 (2) | 0.80172 (15) | 0.63645 (15) | 0.0254 (4) | |
| H72 | −0.128535 | 0.745188 | 0.634942 | 0.030* | |
| C82 | 0.2096 (2) | 1.32678 (14) | 0.98709 (14) | 0.0222 (3) | |
| C92 | 0.1460 (2) | 1.26402 (14) | 0.90480 (14) | 0.0202 (3) | |
| C102 | 0.1499 (2) | 1.13827 (14) | 0.93475 (14) | 0.0224 (3) | |
| H102 | 0.195580 | 1.091393 | 1.008128 | 0.027* | |
| C112 | 0.0878 (2) | 1.08120 (14) | 0.85872 (14) | 0.0233 (3) | |
| H112 | 0.091338 | 0.995647 | 0.879333 | 0.028* | |
| C122 | 0.0204 (2) | 1.15077 (15) | 0.75180 (14) | 0.0212 (3) | |
| C132 | 0.0164 (2) | 1.27536 (15) | 0.71998 (14) | 0.0227 (3) | |
| H132 | −0.029135 | 1.321958 | 0.646447 | 0.027* | |
| C142 | 0.0795 (2) | 1.33135 (14) | 0.79673 (14) | 0.0221 (3) | |
| H142 | 0.077402 | 1.416850 | 0.775266 | 0.026* | |
| O1W | 0.25220 (16) | 0.42894 (11) | 0.49195 (11) | 0.0237 (3) | |
| H1WA | 0.187 (3) | 0.4249 (19) | 0.440 (2) | 0.036* | |
| H1WB | 0.187 (3) | 0.470 (2) | 0.5279 (19) | 0.036* | |
| O2W | 0.48410 (19) | 1.05787 (11) | 1.14845 (12) | 0.0288 (3) | |
| H2WA | 0.396 (3) | 1.118 (2) | 1.135 (2) | 0.043* | |
| H2WB | 0.555 (3) | 1.081 (2) | 1.094 (2) | 0.043* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Co1 | 0.01808 (16) | 0.01436 (15) | 0.01814 (16) | −0.00070 (11) | −0.00183 (11) | −0.00519 (11) |
| N11 | 0.0229 (7) | 0.0182 (6) | 0.0218 (7) | −0.0023 (5) | −0.0016 (5) | −0.0068 (5) |
| N21 | 0.0264 (7) | 0.0155 (6) | 0.0242 (7) | −0.0029 (5) | 0.0015 (6) | −0.0055 (6) |
| N31 | 0.0283 (7) | 0.0188 (7) | 0.0221 (7) | −0.0023 (5) | 0.0012 (6) | −0.0051 (5) |
| N41 | 0.0499 (10) | 0.0235 (8) | 0.0226 (8) | −0.0026 (7) | −0.0074 (7) | −0.0033 (6) |
| N51 | 0.0252 (7) | 0.0195 (6) | 0.0222 (7) | −0.0024 (5) | −0.0003 (5) | −0.0078 (6) |
| N61 | 0.0330 (8) | 0.0175 (7) | 0.0219 (7) | −0.0022 (6) | −0.0075 (6) | −0.0064 (6) |
| C11 | 0.0250 (8) | 0.0198 (8) | 0.0242 (8) | −0.0021 (6) | −0.0013 (6) | −0.0081 (6) |
| C21 | 0.0192 (7) | 0.0179 (7) | 0.0223 (8) | −0.0011 (6) | 0.0029 (6) | −0.0065 (6) |
| C31 | 0.0243 (8) | 0.0228 (8) | 0.0225 (8) | −0.0001 (6) | 0.0015 (6) | −0.0071 (6) |
| C41 | 0.0181 (7) | 0.0198 (7) | 0.0225 (8) | −0.0017 (6) | 0.0013 (6) | −0.0070 (6) |
| C51 | 0.0191 (7) | 0.0181 (7) | 0.0188 (7) | −0.0010 (6) | 0.0008 (6) | −0.0051 (6) |
| O12 | 0.0208 (6) | 0.0188 (5) | 0.0227 (6) | 0.0001 (4) | −0.0017 (4) | −0.0079 (4) |
| O22 | 0.0228 (6) | 0.0210 (6) | 0.0337 (7) | −0.0033 (5) | −0.0003 (5) | −0.0121 (5) |
| O32 | 0.0550 (9) | 0.0198 (6) | 0.0286 (7) | −0.0047 (6) | −0.0121 (6) | −0.0092 (5) |
| O42 | 0.0366 (7) | 0.0231 (6) | 0.0249 (6) | 0.0007 (5) | −0.0068 (5) | −0.0096 (5) |
| O52 | 0.0327 (7) | 0.0233 (6) | 0.0346 (7) | 0.0070 (5) | −0.0122 (5) | −0.0187 (5) |
| C12 | 0.0224 (8) | 0.0171 (7) | 0.0157 (7) | −0.0003 (6) | −0.0031 (6) | −0.0038 (6) |
| C22 | 0.0239 (8) | 0.0170 (7) | 0.0161 (7) | −0.0001 (6) | −0.0011 (6) | −0.0046 (6) |
| C32 | 0.0221 (8) | 0.0219 (8) | 0.0266 (8) | 0.0000 (6) | −0.0011 (6) | −0.0092 (7) |
| C42 | 0.0273 (9) | 0.0209 (8) | 0.0319 (9) | −0.0039 (7) | −0.0042 (7) | −0.0110 (7) |
| C52 | 0.0291 (8) | 0.0191 (7) | 0.0201 (8) | 0.0036 (6) | −0.0050 (6) | −0.0091 (6) |
| C62 | 0.0236 (8) | 0.0268 (9) | 0.0329 (9) | 0.0007 (7) | 0.0007 (7) | −0.0142 (7) |
| C72 | 0.0232 (8) | 0.0240 (8) | 0.0323 (9) | −0.0047 (7) | 0.0012 (7) | −0.0123 (7) |
| C82 | 0.0228 (8) | 0.0204 (8) | 0.0247 (8) | −0.0005 (6) | 0.0002 (6) | −0.0093 (7) |
| C92 | 0.0197 (8) | 0.0201 (8) | 0.0230 (8) | −0.0020 (6) | 0.0020 (6) | −0.0097 (6) |
| C102 | 0.0253 (8) | 0.0198 (8) | 0.0223 (8) | −0.0002 (6) | −0.0016 (6) | −0.0068 (6) |
| C112 | 0.0255 (8) | 0.0173 (7) | 0.0274 (8) | 0.0006 (6) | −0.0010 (7) | −0.0077 (6) |
| C122 | 0.0197 (8) | 0.0234 (8) | 0.0246 (8) | −0.0011 (6) | 0.0002 (6) | −0.0139 (7) |
| C132 | 0.0255 (8) | 0.0224 (8) | 0.0202 (8) | −0.0004 (6) | −0.0013 (6) | −0.0065 (6) |
| C142 | 0.0249 (8) | 0.0176 (7) | 0.0239 (8) | −0.0020 (6) | 0.0019 (6) | −0.0066 (6) |
| O1W | 0.0209 (6) | 0.0260 (6) | 0.0280 (6) | −0.0026 (5) | −0.0023 (5) | −0.0133 (5) |
| O2W | 0.0330 (7) | 0.0189 (6) | 0.0316 (7) | −0.0024 (5) | −0.0017 (5) | −0.0024 (5) |
Geometric parameters (Å, º) top
| Co1—O1Wi | 2.0943 (12) | O52—C52 | 1.3828 (19) |
| Co1—O1W | 2.0943 (12) | O52—C122 | 1.3878 (19) |
| Co1—O12i | 2.1224 (11) | C12—C22 | 1.502 (2) |
| Co1—O12 | 2.1224 (11) | C22—C32 | 1.391 (2) |
| Co1—N11i | 2.1394 (13) | C22—C72 | 1.392 (2) |
| Co1—N11 | 2.1394 (13) | C32—C42 | 1.387 (2) |
| N11—C11 | 1.324 (2) | C32—H32 | 0.9500 |
| N11—C51 | 1.390 (2) | C42—C52 | 1.387 (2) |
| N21—C11 | 1.351 (2) | C42—H42 | 0.9500 |
| N21—C21 | 1.361 (2) | C52—C62 | 1.379 (2) |
| N21—H21 | 0.85 (2) | C62—C72 | 1.384 (2) |
| N31—C31 | 1.327 (2) | C62—H62 | 0.9500 |
| N31—C21 | 1.346 (2) | C72—H72 | 0.9500 |
| N41—C31 | 1.345 (2) | C82—C92 | 1.496 (2) |
| N41—H41A | 0.86 (2) | C92—C142 | 1.392 (2) |
| N41—H41B | 0.88 (2) | C92—C102 | 1.397 (2) |
| N51—C41 | 1.355 (2) | C102—C112 | 1.384 (2) |
| N51—C31 | 1.363 (2) | C102—H102 | 0.9500 |
| N51—H51 | 1.19 (3) | C112—C122 | 1.390 (2) |
| N61—C41 | 1.331 (2) | C112—H112 | 0.9500 |
| N61—H61A | 0.83 (2) | C122—C132 | 1.384 (2) |
| N61—H61B | 0.85 (2) | C132—C142 | 1.386 (2) |
| C11—H11 | 0.9500 | C132—H132 | 0.9500 |
| C21—C51 | 1.393 (2) | C142—H142 | 0.9500 |
| C41—C51 | 1.402 (2) | O1W—H1WA | 0.81 (2) |
| O12—C12 | 1.2773 (19) | O1W—H1WB | 0.84 (2) |
| O22—C12 | 1.2509 (19) | O2W—H2WA | 0.88 (2) |
| O32—C82 | 1.284 (2) | O2W—H2WB | 0.83 (3) |
| O42—C82 | 1.244 (2) | | |
| | | |
| O1W—Co1—N11 | 94.58 (5) | C52—O52—C122 | 119.67 (12) |
| O12—Co1—N11 | 88.49 (5) | O22—C12—O12 | 124.47 (14) |
| O1W—Co1—O12 | 92.75 (5) | O22—C12—C22 | 118.11 (14) |
| O1W—Co1—O12i | 87.25 (5) | O12—C12—C22 | 117.42 (14) |
| O12—Co1—N11i | 91.51 (5) | C32—C22—C72 | 118.60 (14) |
| O1W—Co1—N11i | 85.42 (5) | C32—C22—C12 | 121.86 (14) |
| O1Wi—Co1—O12 | 87.25 (5) | C72—C22—C12 | 119.54 (14) |
| O1Wi—Co1—N11 | 85.42 (5) | C42—C32—C22 | 120.87 (15) |
| O12i—Co1—N11 | 91.51 (5) | C42—C32—H32 | 119.6 |
| N11i—Co1—N11 | 180.0 | C22—C32—H32 | 119.6 |
| O1Wi—Co1—O1W | 180.0 | C32—C42—C52 | 119.31 (15) |
| O12i—Co1—O12 | 180.0 | C32—C42—H42 | 120.3 |
| O1Wi—Co1—O12i | 92.75 (5) | C52—C42—H42 | 120.3 |
| O1Wi—Co1—N11i | 94.58 (5) | C62—C52—O52 | 116.86 (15) |
| O12i—Co1—N11i | 88.49 (5) | C62—C52—C42 | 120.73 (15) |
| C11—N11—C51 | 103.98 (13) | O52—C52—C42 | 122.24 (15) |
| C11—N11—Co1 | 119.17 (11) | C52—C62—C72 | 119.49 (16) |
| C51—N11—Co1 | 136.22 (11) | C52—C62—H62 | 120.3 |
| C11—N21—C21 | 107.21 (13) | C72—C62—H62 | 120.3 |
| C11—N21—H21 | 124.1 (13) | C62—C72—C22 | 120.99 (16) |
| C21—N21—H21 | 128.7 (13) | C62—C72—H72 | 119.5 |
| C31—N31—C21 | 111.90 (14) | C22—C72—H72 | 119.5 |
| C31—N41—H41A | 118.0 (15) | O42—C82—O32 | 123.02 (15) |
| C31—N41—H41B | 119.4 (15) | O42—C82—C92 | 122.04 (15) |
| H41A—N41—H41B | 123 (2) | O32—C82—C92 | 114.94 (14) |
| C41—N51—C31 | 122.03 (14) | C142—C92—C102 | 118.91 (15) |
| C41—N51—H51 | 124.1 (15) | C142—C92—C82 | 119.87 (14) |
| C31—N51—H51 | 113.8 (15) | C102—C92—C82 | 121.21 (14) |
| C41—N61—H61A | 117.9 (14) | C112—C102—C92 | 120.77 (15) |
| C41—N61—H61B | 120.4 (14) | C112—C102—H102 | 119.6 |
| H61A—N61—H61B | 119 (2) | C92—C102—H102 | 119.6 |
| N11—C11—N21 | 113.22 (14) | C102—C112—C122 | 119.11 (15) |
| N11—C11—H11 | 123.4 | C102—C112—H112 | 120.4 |
| N21—C11—H11 | 123.4 | C122—C112—H112 | 120.4 |
| N31—C21—N21 | 126.75 (14) | C132—C122—O52 | 115.90 (14) |
| N31—C21—C51 | 127.53 (15) | C132—C122—C112 | 121.13 (15) |
| N21—C21—C51 | 105.71 (14) | O52—C122—C112 | 122.77 (15) |
| N31—C31—N41 | 119.25 (16) | C122—C132—C142 | 119.18 (15) |
| N31—C31—N51 | 125.61 (15) | C122—C132—H132 | 120.4 |
| N41—C31—N51 | 115.14 (15) | C142—C132—H132 | 120.4 |
| N61—C41—N51 | 118.37 (15) | C132—C142—C92 | 120.90 (15) |
| N61—C41—C51 | 125.76 (15) | C132—C142—H142 | 119.5 |
| N51—C41—C51 | 115.87 (14) | C92—C142—H142 | 119.5 |
| N11—C51—C21 | 109.87 (14) | Co1—O1W—H1WA | 135.0 (16) |
| N11—C51—C41 | 133.10 (15) | Co1—O1W—H1WB | 98.3 (15) |
| C21—C51—C41 | 117.02 (15) | H1WA—O1W—H1WB | 105 (2) |
| C12—O12—Co1 | 127.06 (10) | H2WA—O2W—H2WB | 102 (2) |
| Symmetry code: (i) −x+1, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O1W—H1WB···O22 | 0.84 (2) | 1.81 (2) | 2.6291 (17) | 166 (2) |
| O2W—H2WA···O42 | 0.88 (2) | 1.87 (3) | 2.7290 (18) | 164 (2) |
| N61—H61B···O12i | 0.85 (2) | 2.06 (2) | 2.8885 (19) | 164.6 (19) |
| N41—H41B···O32ii | 0.88 (2) | 2.31 (2) | 3.005 (2) | 136.3 (19) |
| N51—H51···O32ii | 1.19 (3) | 1.35 (3) | 2.5272 (18) | 171 (3) |
| N61—H61A···O42ii | 0.83 (2) | 2.16 (2) | 2.9840 (19) | 169.9 (19) |
| N21—H21···O2Wiii | 0.85 (2) | 1.90 (2) | 2.7522 (19) | 176.3 (19) |
| O1W—H1WA···O22iv | 0.81 (2) | 2.10 (2) | 2.7803 (17) | 141 (2) |
| O2W—H2WB···N31v | 0.83 (3) | 2.16 (3) | 2.9251 (19) | 152 (2) |
| Symmetry codes: (i) −x+1, −y+1, −z+1; (ii) x, y−1, z−1; (iii) x, y, z−1; (iv) −x, −y+1, −z+1; (v) −x+1, −y+2, −z+1. |
Hydrogen-bond geometry (Å, °) for (I). top| Code | D—H···A | D—H | H···A | D···A | D—H···A | 100ρ(r) (a.u) | 100[Nabla]2ρ(r) (a.u) |
| #1 | O1W—H1WB···O22 | 0.84 (2) | 1.81 (2) | 2.6291 (17) | 166 (2) | 3.72 | 1.32 |
| #2 | O2W—H2WA···O42 | 0.88 (2) | 1.87 (3) | 2.7290 (18) | 164 (2) | 2.77 | 1.11 |
| #3 | N61—H61B···O12i | 0.85 (2) | 2.06 (2) | 2.8885 (19) | 164.6 (19) | 1.82 | 0.76 |
| #4 | N41—H41B···O32ii | 0.88 (2) | 2.31 (2) | 3.005 (2) | 136.3 (19) | 0.96 | 0.44 |
| #5 | N51—H51···O32ii | 1.19 (3) | 1.35 (3) | 2.5272 (18) | 171 (3) | 11.1 | 0.64 |
| #6 | N61—H61A···O42ii | 0.83 (2) | 2.16 (2) | 2.9840 (19) | 169.9 (19) | 1.60 | 0.60 |
| #7 | N41—H41A···Cg3iii | 0.83 (2) | 2.72 (2) | 3.3489 (18) | 131.1 (19) | | |
| #8 | N21—H21···O2Wiii | 0.85 (2) | 1.90 (2) | 2.7522 (19) | 176.3 (19) | 2.81 | 1.13 |
| #9 | O1W—H1WA···O22iv | 0.81 (2) | 2.10 (2) | 2.7803 (17) | 141 (2) | 1.76 | 0.70 |
| #10 | O2W—H2WB···N31v | 0.83 (3) | 2.16 (3) | 2.9251 (19) | 152 (2) | 1.60 | 0.59 |
Symmetry codes: (i) -x+1, -y+1, -z+1;
(ii) x, y-1, z-1;
(iii) x, y, z-1;
(iv) -x, -y+1, -z+1;
(v) -x+1, -y+2, -z+1.
Note: Cg3 is the centroid of the C22/C32/C42/C52/C62/C72 ring. |
Analysis of the π–π interactions in (I). top| Code | Group 1/Group 2 | ccd (Å) | da (°) | cpd (Å) |
| #11 | Cg1···Cg4v | 3.7381 (10) | 5.85 (8) | 3.43 (2) |
| #12 | Cg2···Cg4vi | 3.5534 (10) | 4.07 (7) | 3.34 (4) |
| #13 | Cg3···Cg3vi | 3.7946 (11) | 0.00 | 3.5609 (7) |
Symmetry codes: (v) -x+1, -y+2, -z+1;
(vi) -x, -y+2, -z+1.
Notes: ccd is the centre-to-centre distance, da is the dihedral angle and cpd
is the mean centre-to-plane distance. Cg1, Cg2, Cg3 and
Cg4 are the centroids
of the N11/C11/N21/C21/C51, N31/C21/C51/C41/N51/C31, C22/C32/C42/C52/C62/C72
and C92/C102/C112/C122/C132/C14 rings, respectively. |
Analysis of the C—O···π interactions in (I). top| Code | C—O···Cg | O···Cg (Å) | O···Cg/perp (°) |
| #14 | C82—O32···Cg2v | 3.4892 (16) | 12.01 |
| #15 | C82—O42···Cg2v | 3.6653 (14) | 24.42 |
| Symmetry code: (v) -x+1, -y+2, -z+1.
Note: Cg2 is the centroid of the N31/C21/C51/C41/N51/C31 ring. |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
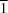 , with one formula unit per cell (Z = 1 and Z′ =
, with one formula unit per cell (Z = 1 and Z′ = 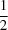 ). It consists of a mononuclear unit with the CoII ion on an inversion centre coordinated by two 2,6-diamino-7H-purin-1-ium cations, two 4,4′-oxydibenzoate anions (in a nonbridging κO-monodentate coordination mode, which is less common for the anion in its CoII complexes) and two water molecules, defining an octahedral environment around the metal atom. There is a rich assortment of nonbonding interactions, among which a strong N+—H
). It consists of a mononuclear unit with the CoII ion on an inversion centre coordinated by two 2,6-diamino-7H-purin-1-ium cations, two 4,4′-oxydibenzoate anions (in a nonbridging κO-monodentate coordination mode, which is less common for the anion in its CoII complexes) and two water molecules, defining an octahedral environment around the metal atom. There is a rich assortment of nonbonding interactions, among which a strong N+—H O− bridge, with a short N
O− bridge, with a short N O distance of 2.5272 (18) Å, stands out, with the H atom ostensibly displaced away from its expected position at the donor side, towards the acceptor. The complex molecules assemble into a three-dimensional hydrogen-bonded network. A variable-temperature magnetic study between 2 and 300 K reveals an orbital contribution to the magnetic moment and a weak antiferromagnetic interaction between CoII centres as the temperature decreases. The model leads to the following values: A (crystal field strength) = 1.81, λ (spin-orbit coupling) = −59.9 cm−1, g (Landé factor) = 2.58 and zJ (exchange coupling) = −0.5 cm−1.
O distance of 2.5272 (18) Å, stands out, with the H atom ostensibly displaced away from its expected position at the donor side, towards the acceptor. The complex molecules assemble into a three-dimensional hydrogen-bonded network. A variable-temperature magnetic study between 2 and 300 K reveals an orbital contribution to the magnetic moment and a weak antiferromagnetic interaction between CoII centres as the temperature decreases. The model leads to the following values: A (crystal field strength) = 1.81, λ (spin-orbit coupling) = −59.9 cm−1, g (Landé factor) = 2.58 and zJ (exchange coupling) = −0.5 cm−1. O− bridge, with a short N
O− bridge, with a short N O distance of 2.5272 (18) Å, stands out, with the H atom ostensibly displaced away from its expected position at the donor side, towards the acceptor. The complex molecules assemble into a three-dimensional hydrogen-bonded network. A variable-temperature magnetic study between 2 and 300 K reveals an orbital contribution to the magnetic moment and a weak antiferromagnetic interaction between CoII centres as the temperature decreases. The model leads to the following values: A (crystal field strength) = 1.81, λ (spin-orbit coupling) = −59.9 cm−1, g (Landé factor) = 2.58 and zJ (exchange coupling) = −0.5 cm−1.
O distance of 2.5272 (18) Å, stands out, with the H atom ostensibly displaced away from its expected position at the donor side, towards the acceptor. The complex molecules assemble into a three-dimensional hydrogen-bonded network. A variable-temperature magnetic study between 2 and 300 K reveals an orbital contribution to the magnetic moment and a weak antiferromagnetic interaction between CoII centres as the temperature decreases. The model leads to the following values: A (crystal field strength) = 1.81, λ (spin-orbit coupling) = −59.9 cm−1, g (Landé factor) = 2.58 and zJ (exchange coupling) = −0.5 cm−1.
 journal menu
journal menu









 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry


