Buy article online - an online subscription or single-article purchase is required to access this article.

Two samples of pure NiCl(OH) were produced by hydrothermal synthesis and characterized by chemical analysis, IR spectroscopy, high-resolution laboratory X-ray powder diffraction and scanning electron microscopy. Layers composed of edge-sharing distorted NiCl
6x(OH)
6−6x octahedra were identified as the main building blocks of the crystal structure. NiCl(OH) is isostructural to CoOOH and crystallizes in space group
R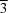 m
m [
a = 3.2606 (1),
c = 17.0062 (9) Å]. Each sample exhibits faults in the stacking pattern of the layers. Crystal intergrowth of (
Aγ
B)(
Bα
C)(
Cβ
A) and (
Aγ
B)(
Aγ
B) [C6 like, β-Ni(OH)
2 related] stacked layers was identified as the main feature of the microstructure of NiCl(OH) by
DIFFaX simulations. A recursion routine for creating distinct stacking patterns of rigid-body-like layers in real space with distinct faults (global optimization) and a Rietveld-compatible approach (local optimization) was realized and implemented in a macro for the program
TOPAS for the first time. This routine enables a recursive creation of supercells containing (
Aγ
B)(
Bα
C)(
Cβ
A), (
Aγ
B)(
Aγ
B) and (
Cβ
A)(
Bα
C)(
Aγ
B) stacking patterns, according to user-defined transition probabilities. Hence it is an enhancement of the few previously published Rietveld-compatible approaches. This routine was applied successfully to create and adapt a detailed microstructure model to the measured data of two stacking-faulted NiCl(OH) samples. The obtained microstructure models were supported by high-resolution scanning electron microscopy images.
Supporting information
CCDC reference: 1426206
Crystal data top
| ClHNiO | V = 156.58 Å3 |
| Mr = 111.15 | Z = 3 |
| Trigonal, R3m | Dx = 3.66 Mg m−3 |
| a = 3.26061 (8) Å | Mo Kα radiation, λ = 0.70930 Å |
| c = 17.0062 (9) Å | T = 295 K |
Data collection top
'Bruker D8-Advance, Debye-Scherrer mode, MPI-FKF, Stuttgart, Germany'
diffractometer | Data collection mode: transmission |
| Johannson Ge(220) primary beam monochromator | Scan method: step |
| Specimen mounting: '0.5 mm glass capillary' | 2θmin = 2.0°, 2θmax = 60.0°, 2θstep = 0.005° |
Refinement top
| Rp = 0.050 | χ2 = 3.568 |
| Rwp = 0.067 | Profile function: Fundamental parameters |
| Rexp = 0.036 | 42 parameters |
| R(F2) = 0.0200 | Background function: 'Chebyshev polynomial' |
Crystal data top
| ClHNiO | V = 156.58 Å3 |
| Mr = 111.15 | Z = 3 |
| Trigonal, R3m | Mo Kα radiation, λ = 0.70930 Å |
| a = 3.26061 (8) Å | T = 295 K |
| c = 17.0062 (9) Å | |
Data collection top
'Bruker D8-Advance, Debye-Scherrer mode, MPI-FKF, Stuttgart, Germany'
diffractometer | Scan method: step |
| Specimen mounting: '0.5 mm glass capillary' | 2θmin = 2.0°, 2θmax = 60.0°, 2θstep = 0.005° |
| Data collection mode: transmission | |
Refinement top
| Rp = 0.050 | R(F2) = 0.0200 |
| Rwp = 0.067 | χ2 = 3.568 |
| Rexp = 0.036 | 42 parameters |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Biso*/Beq | Occ. (<1) |
| Ni1 | 0 | 0 | 0 | 1.21 (2) | |
| O1 | 0.33333 | 0.66667 | 0.0350 (2) | 0.44 (3) | 0.5 |
| Cl1 | 0.33333 | 0.66667 | 0.08790 (8) | 0.44 (3) | 0.5 |
Geometric parameters (Å, º) top
| Ni1—O1 | 1.9744 (11) | Ni1—Cl1 | 2.4038 (9) |
| | | |
| O1i—Ni1i—Cl1i | 20.9 (1) | | |
| Symmetry code: (i) −x, −x+y, −z. |
Experimental details
| Crystal data |
| Chemical formula | ClHNiO |
| Mr | 111.15 |
| Crystal system, space group | Trigonal, R3m |
| Temperature (K) | 295 |
| a, c (Å) | 3.26061 (8), 17.0062 (9) |
| V (Å3) | 156.58 |
| Z | 3 |
| Radiation type | Mo Kα, λ = 0.70930 Å |
| µ (mm−1) | ? |
| Specimen shape, size (mm) | Cylinder, ? × ? × ? |
| |
| Data collection |
| Data collection method | 'Bruker D8-Advance, Debye-Scherrer mode, MPI-FKF, Stuttgart, Germany' |
| Specimen mounting | '0.5 mm glass capillary' |
| Data collection mode | Transmission |
| Scan method | Step |
| 2θ values (°) | 2θmin = 2.0 2θmax = 60.0 2θstep = 0.005 |
| |
| Refinement |
| R factors and goodness of fit | Rp = 0.050, Rwp = 0.067, Rexp = 0.036, R(F2) = 0.0200, χ2 = 3.568 |
| No. of parameters | 42 |
| No. of restraints | ? |

 Subscribe to Journal of Applied Crystallography
Subscribe to Journal of Applied Crystallography
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
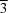 m [a = 3.2606 (1), c = 17.0062 (9) Å]. Each sample exhibits faults in the stacking pattern of the layers. Crystal intergrowth of (AγB)(BαC)(CβA) and (AγB)(AγB) [C6 like, β-Ni(OH)2 related] stacked layers was identified as the main feature of the microstructure of NiCl(OH) by DIFFaX simulations. A recursion routine for creating distinct stacking patterns of rigid-body-like layers in real space with distinct faults (global optimization) and a Rietveld-compatible approach (local optimization) was realized and implemented in a macro for the program TOPAS for the first time. This routine enables a recursive creation of supercells containing (AγB)(BαC)(CβA), (AγB)(AγB) and (CβA)(BαC)(AγB) stacking patterns, according to user-defined transition probabilities. Hence it is an enhancement of the few previously published Rietveld-compatible approaches. This routine was applied successfully to create and adapt a detailed microstructure model to the measured data of two stacking-faulted NiCl(OH) samples. The obtained microstructure models were supported by high-resolution scanning electron microscopy images.
m [a = 3.2606 (1), c = 17.0062 (9) Å]. Each sample exhibits faults in the stacking pattern of the layers. Crystal intergrowth of (AγB)(BαC)(CβA) and (AγB)(AγB) [C6 like, β-Ni(OH)2 related] stacked layers was identified as the main feature of the microstructure of NiCl(OH) by DIFFaX simulations. A recursion routine for creating distinct stacking patterns of rigid-body-like layers in real space with distinct faults (global optimization) and a Rietveld-compatible approach (local optimization) was realized and implemented in a macro for the program TOPAS for the first time. This routine enables a recursive creation of supercells containing (AγB)(BαC)(CβA), (AγB)(AγB) and (CβA)(BαC)(AγB) stacking patterns, according to user-defined transition probabilities. Hence it is an enhancement of the few previously published Rietveld-compatible approaches. This routine was applied successfully to create and adapt a detailed microstructure model to the measured data of two stacking-faulted NiCl(OH) samples. The obtained microstructure models were supported by high-resolution scanning electron microscopy images.
 journal menu
journal menu









 Subscribe to Journal of Applied Crystallography
Subscribe to Journal of Applied Crystallography


