Buy article online - an online subscription or single-article purchase is required to access this article.

The synthesis of BaC
2O
4·0.5H
2O and its thermal decomposition to α-BaC
2O
4 and β-BaC
2O
4 was investigated. BaC
2O
4·0.5H
2O is precipitated at room temperature from aqueous solutions of barium chloride and ammonium oxalate. The deuterated compound BaC
2O
4·0.5D
2O was made in analogy with D
2O as the solvent. The compounds were characterized by X-ray and neutron diffraction analysis. Single-crystal X-ray diffraction of BaC
2O
4·0.5H
2O measured at 120 K gave the triclinic cell
a = 8.692 (1),
b = 9.216 (1),
c = 6.146 (1) Å, α = 95.094 (3), β = 95.492 (3), γ = 64.500 (3)°, space group
P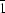
,
Z = 4. Two independent Ba atoms are each coordinated to nine O atoms at distances from 2.73 (1) to 2.99 (1) Å. One of the two oxalate ions deviates significantly from planarity. The water molecule does form weak hydrogen bonds.
In situ X-ray powder diffraction was used to study the thermal decomposition of BaC
2O
4·0.5H
2O and the formation of α-BaC
2O
4. The X-ray powder pattern of α-BaC
2O
4 measured at 473 K was indexed on a triclinic cell with
a = 5.137 (3),
b = 8.764 (6),
c = 9.006 (4) Å, α = 83.57 (4), β = 98.68 (5), γ = 99.53 (5)°, and the space group
P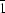
with
Z = 4.
Supporting information
CCDC reference: 195793
Data collection: SMART (Siemens, 1995); cell refinement: SAINT (Siemens, 1995); data reduction: SAINT (Siemens, 1995); program(s) used to solve structure: SIR97 (Altomare et al., 1997 ), KRYSTAL; program(s) used to refine structure: modified ORFLS(1962), KRYSTAL; molecular graphics: ORTEP-III (Burnett & Johnson, 1996), KRYSTAL; software used to prepare material for publication: KRYSTAL.
Crystal data top
| C4H2Ba2O9 | Z = 2 |
| Mr = 468.72 | F(000) = 420 |
| Triclinic, P1 | Dx = 3.524 Mg m−3 |
| Hall symbol: -P 1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 8.692 (1) Å | Cell parameters from 1438 reflections |
| b = 9.216 (1) Å | θ = 2.5–30.5° |
| c = 6.146 (1) Å | µ = 8.89 mm−1 |
| α = 95.094 (3)° | T = 120 K |
| β = 95.492 (3)° | Plate, colourless |
| γ = 64.500 (3)° | 0.15 × 0.06 × 0.01 mm |
| V = 441.7 (1) Å3 | |
Data collection top
Siemens SMART CCD
diffractometer | 2584 independent reflections |
| Radiation source: x-ray tube | 1339 reflections with I > 3σ(I) |
| Graphite monochromator | Rint = 0.075 |
| ω rotation scans with narrow frames | θmax = 30.5°, θmin = 2.5° |
Absorption correction: integration
XPREP,1995 | h = −12→12 |
| Tmin = 0.28, Tmax = 0.84 | k = −13→13 |
| 12950 measured reflections | l = −8→8 |
Refinement top
| Refinement on F | 0 constraints |
| Least-squares matrix: full | H-atom parameters constrained |
| R[F2 > 2σ(F2)] = 0.050 | w = 1/{[σcs(F2) + 1.03F2]1/2- |F|}2 |
| wR(F2) = 0.054 | (Δ/σ)max = 0.00013 |
| S = 1.20 | Δρmax = 4.0 (5) e Å−3 |
| 1339 reflections | Δρmin = −3.8 (5) e Å−3 |
| 72 parameters | Extinction correction: Type 1 Lorentzian isotropic (Becker and Coppens, 1974) |
| 0 restraints | Extinction coefficient: 7 (1) |
Crystal data top
| C4H2Ba2O9 | γ = 64.500 (3)° |
| Mr = 468.72 | V = 441.7 (1) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 8.692 (1) Å | Mo Kα radiation |
| b = 9.216 (1) Å | µ = 8.89 mm−1 |
| c = 6.146 (1) Å | T = 120 K |
| α = 95.094 (3)° | 0.15 × 0.06 × 0.01 mm |
| β = 95.492 (3)° | |
Data collection top
Siemens SMART CCD
diffractometer | 2584 independent reflections |
Absorption correction: integration
XPREP,1995 | 1339 reflections with I > 3σ(I) |
| Tmin = 0.28, Tmax = 0.84 | Rint = 0.075 |
| 12950 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.050 | 0 restraints |
| wR(F2) = 0.054 | H-atom parameters constrained |
| S = 1.20 | Δρmax = 4.0 (5) e Å−3 |
| 1339 reflections | Δρmin = −3.8 (5) e Å−3 |
| 72 parameters | |
Special details top
Refinement. Because of heavy Ba atoms and weak dataset anisotropic refinement of carbon and
oxygen was not possible. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Ba1 | 0.9558 (1) | 0.2093 (1) | 0.2782 (2) | 0.0088 (7) | |
| Ba2 | 0.3635 (1) | 0.4185 (1) | 0.2133 (2) | 0.0080 (7) | |
| C1 | 0.924 (2) | 0.677 (2) | 0.176 (3) | 0.011 (3)* | |
| C2 | 0.820 (2) | 0.627 (2) | 0.325 (3) | 0.012 (3)* | |
| O1 | 0.875 (1) | 0.686 (1) | −0.019 (2) | 0.016 (3)* | |
| O2 | 1.047 (1) | 0.696 (1) | 0.267 (2) | 0.013 (2)* | |
| O3 | 0.747 (1) | 0.540 (1) | 0.238 (2) | 0.009 (2)* | |
| O4 | 0.819 (2) | 0.673 (1) | 0.524 (2) | 0.020 (3)* | |
| C3 | 0.276 (2) | 0.820 (2) | 0.136 (2) | 0.005 (3)* | |
| C4 | 0.287 (2) | 0.817 (2) | 0.385 (3) | 0.013 (3)* | |
| O5 | 0.356 (1) | 0.689 (1) | 0.029 (2) | 0.010 (2)* | |
| O6 | 0.185 (1) | 0.950 (1) | 0.050 (2) | 0.014 (2)* | |
| O7 | 0.370 (1) | 0.685 (1) | 0.475 (2) | 0.010 (2)* | |
| O8 | 0.199 (1) | 0.947 (1) | 0.486 (2) | 0.015 (2)* | |
| O9 | 0.516 (2) | 0.070 (1) | 0.220 (2) | 0.023 (3)* | |
| H1 | 0.648 | 0.024 | 0.302 | 0.03* | |
| H2 | 0.413 | 0.101 | 0.306 | 0.04* | 0.5 |
| H3 | 0.457 | 0.082 | 0.132 | 0.04* | 0.5 |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Ba1 | 0.0104 (6) | 0.0060 (6) | 0.0123 (6) | −0.0053 (5) | 0.0014 (5) | 0.0025 (4) |
| Ba2 | 0.0080 (6) | 0.0070 (6) | 0.0116 (6) | −0.0053 (5) | 0.0011 (4) | 0.0020 (4) |
Geometric parameters (Å, º) top
| Ba1—O6i | 2.735 (11) | Ba2v—O4 | 2.776 (12) |
| Ba1—O8i | 2.752 (11) | C1—O1 | 1.231 (18) |
| Ba1—O1ii | 2.754 (11) | C1—O2 | 1.239 (19) |
| Ba1—O4iii | 2.763 (12) | C1—C2 | 1.57 (2) |
| Ba1—O3 | 2.819 (10) | C2—O3 | 1.267 (18) |
| Ba1—O2iii | 2.855 (10) | C2—O4 | 1.260 (18) |
| Ba1—O6iv | 2.878 (11) | C3—O6 | 1.248 (17) |
| Ba1—O8v | 2.896 (11) | C3—O5 | 1.265 (16) |
| Ba1—O5iv | 2.989 (11) | C3—C4 | 1.53 (2) |
| Ba2—O5iv | 2.736 (10) | C4—O8 | 1.258 (18) |
| Ba2—O7v | 2.744 (11) | C4—O7 | 1.261 (18) |
| Ba2—O1iv | 2.769 (12) | Ba2iv—O5 | 2.736 (10) |
| Ba2—O4v | 2.776 (12) | Ba2—O5 | 2.806 (10) |
| Ba2—O5 | 2.806 (10) | Ba1iv—O5 | 2.989 (11) |
| Ba2—O7 | 2.830 (10) | Ba1viii—O6 | 2.735 (11) |
| Ba2—O3iv | 2.852 (10) | O6—H1iv | 2.796 (11) |
| Ba2—O2vi | 2.859 (11) | Ba1iv—O6 | 2.878 (11) |
| Ba2—O9 | 2.905 (12) | Ba2v—O7 | 2.744 (11) |
| C1—O1 | 1.231 (18) | Ba2—O7 | 2.830 (10) |
| Ba1ii—O1 | 2.754 (11) | O8—H1v | 1.857 (11) |
| Ba2iv—O1 | 2.769 (12) | Ba1viii—O8 | 2.752 (11) |
| C1—O2 | 1.239 (19) | O8—O9v | 2.880 (17) |
| Ba1iii—O2 | 2.855 (10) | Ba1v—O8 | 2.896 (11) |
| Ba2vii—O2 | 2.859 (11) | O9—H1 | 1.123 (13) |
| C2—O3 | 1.267 (18) | O9—H3 | 0.690 (13) |
| Ba1—O3 | 2.819 (10) | O9—H2 | 1.000 (13) |
| Ba2iv—O3 | 2.852 (10) | O9—H3ix | 2.445 (12) |
| C2—O4 | 1.260 (18) | Ba2—O9 | 2.905 (12) |
| O4—H2v | 2.429 (12) | O9—O9ix | 2.93 (2) |
| Ba1iii—O4 | 2.763 (12) | | |
| | | |
| O6i—Ba1—O8i | 58.4 (3) | O5—Ba2—O7 | 58.0 (3) |
| O1ii—Ba1—O6i | 70.9 (3) | O3iv—Ba2—O5 | 62.9 (3) |
| O4iii—Ba1—O6i | 98.0 (3) | O2vi—Ba2—O5 | 67.6 (3) |
| O3—Ba1—O6i | 140.4 (3) | O5—Ba2—O9 | 147.0 (3) |
| O2iii—Ba1—O6i | 131.0 (3) | O3iv—Ba2—O7 | 118.7 (3) |
| O6i—Ba1—O6iv | 63.6 (4) | O2vi—Ba2—O7 | 62.0 (3) |
| O6i—Ba1—O8v | 100.6 (3) | O7—Ba2—O9 | 138.7 (3) |
| O5iv—Ba1—O6i | 101.9 (3) | O2vi—Ba2—O3iv | 82.2 (3) |
| O1ii—Ba1—O8i | 107.1 (3) | O3iv—Ba2—O9 | 100.4 (3) |
| O4iii—Ba1—O8i | 77.4 (3) | O2vi—Ba2—O9 | 141.9 (3) |
| O3—Ba1—O8i | 154.2 (3) | O2vi—Ba2—O9 | 141.9 (3) |
| O2iii—Ba1—O8i | 73.9 (3) | Ba1ii—O1—C1 | 115.4 (10) |
| O6iv—Ba1—O8i | 100.0 (3) | Ba2iv—O1—C1 | 124.9 (10) |
| O8i—Ba1—O8v | 68.4 (4) | Ba1iii—O2—C1 | 119.1 (9) |
| O5iv—Ba1—O8i | 142.5 (3) | Ba2vii—O2—C1 | 112.6 (10) |
| O1ii—Ba1—O4iii | 61.0 (3) | Ba1—O3—C2 | 112.0 (9) |
| O1ii—Ba1—O3 | 74.6 (3) | Ba2iv—O3—C2 | 116.5 (9) |
| O1ii—Ba1—O2iii | 117.4 (3) | Ba1iii—O4—C2 | 123.1 (10) |
| O1ii—Ba1—O6iv | 100.2 (3) | Ba2v—O4—C2 | 116.4 (10) |
| O1ii—Ba1—O8v | 171.4 (3) | O1—C1—O2 | 128.3 (15) |
| O1ii—Ba1—O5iv | 93.3 (3) | C2—C1—O1 | 115.2 (14) |
| O3—Ba1—O4iii | 81.6 (3) | C2—C1—O2 | 116.4 (13) |
| O2iii—Ba1—O4iii | 58.4 (3) | O3—C2—O4 | 125.2 (14) |
| O4iii—Ba1—O6iv | 158.3 (3) | C1—C2—O3 | 118.1 (13) |
| O4iii—Ba1—O8v | 123.2 (3) | C1—C2—O4 | 116.7 (13) |
| O4iii—Ba1—O5iv | 139.9 (3) | O5—C3—O6 | 123.9 (13) |
| O2iii—Ba1—O3 | 82.5 (3) | C4—C3—O6 | 118.5 (13) |
| O3—Ba1—O6iv | 105.0 (3) | C4—C3—O5 | 117.6 (13) |
| O3—Ba1—O8v | 112.8 (3) | O7—C4—O8 | 124.1 (14) |
| O3—Ba1—O5iv | 61.0 (3) | C3—C4—O8 | 116.2 (13) |
| O2iii—Ba1—O6iv | 142.2 (3) | C3—C4—O7 | 119.4 (13) |
| O2iii—Ba1—O8v | 69.1 (3) | Ba2iv—O5—C3 | 132.7 (9) |
| O2iii—Ba1—O5iv | 124.1 (3) | Ba2—O5—C3 | 113.9 (8) |
| O6iv—Ba1—O8v | 74.0 (3) | Ba1iv—O5—C3 | 85.8 (8) |
| O5iv—Ba1—O6iv | 44.4 (3) | Ba1viii—O6—C3 | 122.2 (9) |
| O5iv—Ba1—O8v | 86.8 (3) | Ba1iv—O6—C3 | 91.2 (8) |
| O5iv—Ba2—O7v | 77.1 (3) | Ba2v—O7—C4 | 133.4 (10) |
| O1iv—Ba2—O5iv | 110.8 (3) | Ba2—O7—C4 | 112.9 (9) |
| O4v—Ba2—O5iv | 144.8 (3) | Ba1viii—O8—C4 | 122.6 (10) |
| O5iv—Ba2—O5 | 73.5 (3) | Ba1v—O8—C4 | 91.5 (9) |
| O5iv—Ba2—O7 | 105.3 (3) | H1—O9—H3 | 154.5 (16) |
| O3iv—Ba2—O5iv | 71.0 (3) | H1—O9—H2 | 121.6 (11) |
| O2vi—Ba2—O5iv | 139.7 (3) | H1—O9—H3ix | 105.9 (7) |
| O5iv—Ba2—O9 | 74.1 (3) | O8v—O9—H1 | 19.3 (5) |
| O1iv—Ba2—O7v | 137.2 (3) | Ba2—O9—H1 | 110.5 (8) |
| O4v—Ba2—O7v | 88.1 (3) | O9ix—O9—H1 | 116.2 (9) |
| O5—Ba2—O7v | 108.5 (3) | H2—O9—H3 | 83.0 (12) |
| O7v—Ba2—O7 | 70.1 (3) | H3—O9—H3ix | 50.4 (8) |
| O3iv—Ba2—O7v | 148.2 (3) | O8v—O9—H3 | 164.7 (13) |
| O2vi—Ba2—O7v | 124.7 (3) | Ba2—O9—H3 | 79.2 (10) |
| O7v—Ba2—O9 | 69.7 (3) | O9ix—O9—H3 | 39.9 (7) |
| O1iv—Ba2—O4v | 60.7 (3) | H2—O9—H3ix | 121.1 (9) |
| O1iv—Ba2—O5 | 114.1 (3) | O8v—O9—H2 | 109.4 (8) |
| O1iv—Ba2—O7 | 138.6 (3) | Ba2—O9—H2 | 78.1 (7) |
| O1iv—Ba2—O3iv | 58.4 (3) | O9ix—O9—H2 | 113.7 (11) |
| O1iv—Ba2—O2vi | 77.2 (3) | O8v—O9—H3ix | 124.0 (5) |
| O1iv—Ba2—O9 | 72.4 (3) | Ba2—O9—H3ix | 117.2 (4) |
| O4v—Ba2—O5 | 141.7 (3) | O9ix—O9—H3ix | 10.4 (2) |
| O4v—Ba2—O7 | 99.2 (3) | Ba2—O9—O8v | 94.2 (4) |
| O3iv—Ba2—O4v | 118.0 (3) | O8v—O9—O9ix | 133.9 (7) |
| O2vi—Ba2—O4v | 74.5 (3) | Ba2—O9—O9ix | 109.7 (6) |
| O4v—Ba2—O9 | 70.8 (3) | | |
| Symmetry codes: (i) x+1, y−1, z; (ii) −x+2, −y+1, −z; (iii) −x+2, −y+1, −z+1; (iv) −x+1, −y+1, −z; (v) −x+1, −y+1, −z+1; (vi) x−1, y, z; (vii) x+1, y, z; (viii) x−1, y+1, z; (ix) −x+1, −y, −z. |
Experimental details
| Crystal data |
| Chemical formula | C4H2Ba2O9 |
| Mr | 468.72 |
| Crystal system, space group | Triclinic, P1 |
| Temperature (K) | 120 |
| a, b, c (Å) | 8.692 (1), 9.216 (1), 6.146 (1) |
| α, β, γ (°) | 95.094 (3), 95.492 (3), 64.500 (3) |
| V (Å3) | 441.7 (1) |
| Z | 2 |
| Radiation type | Mo Kα |
| µ (mm−1) | 8.89 |
| Crystal size (mm) | 0.15 × 0.06 × 0.01 |
| |
| Data collection |
| Diffractometer | Siemens SMART CCD
diffractometer |
| Absorption correction | Integration
XPREP,1995 |
| Tmin, Tmax | 0.28, 0.84 |
No. of measured, independent and
observed [I > 3σ(I)] reflections | 12950, 2584, 1339 |
| Rint | 0.075 |
| (sin θ/λ)max (Å−1) | 0.714 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.050, 0.054, 1.20 |
| No. of reflections | 1339 |
| No. of parameters | 72 |
| H-atom treatment | H-atom parameters constrained |
| Δρmax, Δρmin (e Å−3) | 4.0 (5), −3.8 (5) |

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
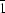 , Z = 4. Two independent Ba atoms are each coordinated to nine O atoms at distances from 2.73 (1) to 2.99 (1) Å. One of the two oxalate ions deviates significantly from planarity. The water molecule does form weak hydrogen bonds. In situ X-ray powder diffraction was used to study the thermal decomposition of BaC2O4·0.5H2O and the formation of α-BaC2O4. The X-ray powder pattern of α-BaC2O4 measured at 473 K was indexed on a triclinic cell with a = 5.137 (3), b = 8.764 (6), c = 9.006 (4) Å, α = 83.57 (4), β = 98.68 (5), γ = 99.53 (5)°, and the space group P
, Z = 4. Two independent Ba atoms are each coordinated to nine O atoms at distances from 2.73 (1) to 2.99 (1) Å. One of the two oxalate ions deviates significantly from planarity. The water molecule does form weak hydrogen bonds. In situ X-ray powder diffraction was used to study the thermal decomposition of BaC2O4·0.5H2O and the formation of α-BaC2O4. The X-ray powder pattern of α-BaC2O4 measured at 473 K was indexed on a triclinic cell with a = 5.137 (3), b = 8.764 (6), c = 9.006 (4) Å, α = 83.57 (4), β = 98.68 (5), γ = 99.53 (5)°, and the space group P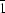 with Z = 4.
with Z = 4.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/b/issues/2002/05/00/os0083/os0083fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/b/issues/2002/05/00/os0083/os0083fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/b/issues/2002/05/00/os0083/os0083fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/b/issues/2002/05/00/os0083/os0083fig4thm.gif)

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


