Buy article online - an online subscription or single-article purchase is required to access this article.

The crystal structure of mayenite (12CaO·7Al
2O
3) has been investigated by single-crystal synchrotron diffraction with high resolution and accuracy, using a four-circle diffractometer equipped with an avalanche photodiode detector (APD) detector installed at PF14A in Tsukuba, Japan. Analysis revealed random displacements of ions by the electrostatic force of the O
2- ion (O3) clathrated in two out of 12 cages. O3 ions are located at general positions close to the
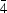
site at the centre of each cage. The difference-density map revealed two large peaks corresponding to displaced Ca ions. The positive ions close to O3 are displaced and one-to-one correspondence was found between one of the four equivalent O3 ions and the displaced ions. When an O3 ion is present in the cage the Al ion at the 16
c position moves 0.946 (3) Å toward the O3 ion. One of the Al-O bonds is broken and a new Al-O3 bond is created. The result is an AlO
4 tetrahedron that is quite deformed. The three O1 ions and the O2 ion of the destroyed AlO
4 tetrahedron are forcibly displaced. O1 and O2 have two and one displaced ions, respectively. The local structure of the cage occupied by one of the four O3 ions was determined by calculating the electrostatic potential and electric field in the deformed cage, although the position of one of the displaced O1 ions was not clearly identified.
Supporting information
Data collection: BL14A diffractometer control software
(Satow. Y. and Iitaka. Y., (1989). Sci. Instrum. 60, 2390-2393;
Vaalsta, T. P. & Hester, J. R. (1997). DIFF14A Software. Photon
Factory, Tsukuba, Japan. ); cell refinement: RSLC-3 UNICS system (Sakurai, T. & Kobayashi, K. (1979), Rep. Inst. Phys.
Chem. Res. 55, 69-77); data reduction: sortref (Sakakura, T. (2008): private communication.); program(s) used to refine structure: MOLLY (Hansen, N. K. and Coppens. P. (1978) Acta Cryst. A34, 909-921.); molecular graphics: gluplot (Sakakura, T. (2008): private communication.).
Crystal data top
| Al14Ca12O33 | Dx = 2.672 Mg m−3 |
| Mr = 1386.66 | Synchrotron radiation, λ = 0.67954 Å |
| Cubic, I43d | Cell parameters from 24 reflections |
| Hall symbol: I -4bd 2c 3 | θ = 34.7–36.1° |
| a = 11.989 (3) Å | µ = 1.99 mm−1 |
| V = 1723.2 (7) Å3 | T = 298 K |
| Z = 2 | Sphere, orange |
| F(000) = 1372 | 0.07 × 0.07 × 0.07 × 0.03 (radius) mm |
Data collection top
Tsukuba BL14A four-circle
diffractometer | Rint = 0.012 |
| integrated intensities data from ω/2θ scans | θmax = 68.4°, θmin = 4.0° |
Absorption correction: for a sphere
Transmission coefficient for spheres tabulated in International Table
II(1972) Table 5.3.6B was interpolated with Lagrangian method | h = −9→32 |
| Tmin = 0.908, Tmax = 0.909 | k = −9→24 |
| 10790 measured reflections | l = −31→31 |
| 3077 independent reflections | 9 standard reflections every 200 reflections |
| 3020 reflections with F > 3.0σ(F) | intensity decay: none |
Refinement top
| Refinement on F | 0 constraints |
| Least-squares matrix: full | w = 1/[σ2(Fo) + (0.016P)2]
where P = (Fo2 + 2Fc2)/3 |
| R[F2 > 2σ(F2)] = 0.016 | (Δ/σ)max = 0.050 |
| wR(F2) = 0.024 | Δρmax = 0.34 e Å−3 |
| S = 1.09 | Δρmin = −0.39 e Å−3 |
| 3020 reflections | Extinction correction: B-C type 2 isotropic |
| 66 parameters | Extinction coefficient: 0.00006 (4) |
| 0 restraints | |
Crystal data top
| Al14Ca12O33 | Z = 2 |
| Mr = 1386.66 | Synchrotron radiation, λ = 0.67954 Å |
| Cubic, I43d | µ = 1.99 mm−1 |
| a = 11.989 (3) Å | T = 298 K |
| V = 1723.2 (7) Å3 | 0.07 × 0.07 × 0.07 × 0.03 (radius) mm |
Data collection top
Tsukuba BL14A four-circle
diffractometer | 3020 reflections with F > 3.0σ(F) |
Absorption correction: for a sphere
Transmission coefficient for spheres tabulated in International Table
II(1972) Table 5.3.6B was interpolated with Lagrangian method | Rint = 0.012 |
| Tmin = 0.908, Tmax = 0.909 | 9 standard reflections every 200 reflections |
| 10790 measured reflections | intensity decay: none |
| 3077 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.016 | 66 parameters |
| wR(F2) = 0.024 | 0 restraints |
| S = 1.09 | Δρmax = 0.34 e Å−3 |
| 3020 reflections | Δρmin = −0.39 e Å−3 |
Special details top
Experimental. Multiple diffraction was avoided using ψ-scan. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | Occ. (<1) |
| Ca1 | 0.139339 (15) | 0 | 0.25 | 0.00869 (3) | 0.75 |
| Al1 | 0.268614 (9) | 0.268614 (9) | 0.268614 (9) | 0.00633 (3) | 0.875 |
| Al2 | 0.25 | 0.125 | 0.5 | 0.00679 (3) | |
| O1 | 0.15052 (4) | 0.03605 (3) | 0.44234 (3) | 0.01155 (7) | 0.916666 |
| O2 | 0.18510 (4) | 0.18510 (4) | 0.18510 (4) | 0.01011 (7) | 0.875 |
| O3 | 0.3559 (6) | 0.0614 (7) | 0.2506 (7) | 0.0163 (8)* | 0.041667 |
| Ca1a | 0.16936 (16) | 0.00673 (17) | 0.2504 (2) | 0.00987 (16)* | 0.041667 |
| Ca1b | 0.20692 (12) | 0 | 0.25 | 0.0128 (2) | 0.083334 |
| Ca1c | 0.13537 (17) | 0.0043 (3) | 0.2354 (2) | 0.00930 (3) | 0.041667 |
| Al1a | 0.3041 (2) | 0.1984 (2) | 0.2619 (2) | 0.0096 (3)* | 0.041667 |
| O1a | 0.1302 (3) | 0.0603 (3) | 0.4329 (3) | 0.0039 (3)* | 0.041667 |
| O1b | 0.1718 (6) | 0.0312 (5) | 0.4352 (5) | 0.0079 (6)* | 0.041667 |
| O2a | 0.1996 (6) | 0.1750 (8) | 0.1893 (7) | 0.00663 (7) | 0.041667 |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Ca1 | 0.00936 (4) | 0.00981 (4) | 0.00691 (4) | 0 | 0 | 0.00090 (4) |
| Al1 | 0.00633 (4) | 0.00633 (4) | 0.00633 (4) | −0.000145 (19) | −0.000145 (19) | −0.000145 (19) |
| Al2 | 0.00774 (5) | 0.00488 (5) | 0.00774 (5) | 0 | 0 | 0 |
| O1 | 0.01081 (8) | 0.01258 (9) | 0.01128 (7) | −0.00569 (8) | 0.00251 (7) | −0.00328 (6) |
| O2 | 0.01011 (8) | 0.01011 (8) | 0.01011 (8) | −0.00174 (6) | −0.00174 (6) | −0.00174 (6) |
| Ca1b | 0.0176 (4) | 0.0113 (3) | 0.0094 (2) | 0 | 0 | 0.0000 (2) |
| Ca1c | 0.00936 (4) | 0.00981 (4) | 0.00691 (4) | 0 | 0 | 0.00090 (4) |
| Al2a | 0.00774 (5) | 0.00488 (5) | 0.00774 (5) | 0 | 0 | 0 |
Geometric parameters (Å, º) top
| Al1i—O1ii | 1.7746 (4) | Ca1xii—O1aii | 2.311 (3) |
| Al1i—O1iii | 1.7746 (4) | Ca1i—O1xiii | 2.3500 (3) |
| Al1i—O1iv | 1.7746 (4) | Ca1i—O1xiv | 2.5143 (4) |
| Al1i—O1aii | 1.739 (4) | Ca1i—O1i | 2.3500 (3) |
| Al1i—O1biii | 2.009 (7) | Ca1i—O1xv | 2.5143 (4) |
| Al1i—O2i | 1.7342 (4) | Ca1i—O2xvi | 2.4148 (4) |
| Al1i—O2ai | 1.688 (9) | Ca1i—O2i | 2.4148 (4) |
| Al1i—O3i | 2.704 (8) | Ca1i—O2ai | 2.335 (9) |
| Al1ai—O1ii | 1.673 (3) | Ca1i—O3i | 2.699 (7) |
| Al1ai—O1iii | 2.709 (3) | Ca1xvii—O2i | 2.4148 (4) |
| Al1ai—O1iv | 1.839 (3) | Ca1xvii—O2ai | 2.387 (8) |
| Al1ai—O1aii | 1.431 (5) | Ca1xi—O1iii | 2.5143 (4) |
| Al1ai—O1biii | 2.936 (7) | Ca1xi—O1iv | 2.3500 (3) |
| Al1ai—O2i | 1.705 (3) | Ca1xi—O1biii | 2.380 (6) |
| Al1ai—O2ai | 1.551 (8) | Ca1xv—O2i | 2.4148 (4) |
| Al1ai—O3i | 1.762 (9) | Ca1xv—O2ai | 2.626 (8) |
| Al2—O1iv | 1.7428 (4) | Ca1ai—O1xiii | 2.378 (3) |
| Al2ii—O1ii | 1.7428 (4) | Ca1ai—O1xiv | 2.841 (2) |
| Al2ii—O1v | 1.7428 (4) | Ca1ai—O1i | 2.339 (3) |
| Al2ii—O1vi | 1.7428 (4) | Ca1ai—O1xv | 2.805 (2) |
| Al2ii—O1i | 1.7428 (4) | Ca1ai—O1aii | 3.221 (4) |
| Al2ii—O1aii | 1.820 (4) | Ca1ai—O2xvi | 2.434 (2) |
| Al2iii—O1iii | 1.7428 (4) | Ca1ai—O2i | 2.285 (2) |
| Al2iii—O1biii | 1.657 (6) | Ca1ai—O2ai | 2.177 (9) |
| Ca1—O1 | 2.3500 (3) | Ca1ai—O3i | 2.331 (8) |
| Ca1—O1vii | 2.3500 (3) | Ca1b—O1 | 2.4416 (5) |
| Ca1—O1viii | 2.5143 (4) | Ca1b—O1vii | 2.4416 (5) |
| Ca1—O1ix | 2.5143 (4) | Ca1b—O1viii | 3.2227 (14) |
| Ca1—O2 | 2.4148 (4) | Ca1b—O1ix | 3.2227 (14) |
| Ca1—O2ii | 2.4148 (4) | Ca1b—O2 | 2.3661 (5) |
| Ca1—O3i | 3.141 (7) | Ca1b—O2ii | 2.3661 (5) |
| Ca1x—O1ii | 2.5143 (4) | Ca1b—O3i | 2.361 (8) |
| Ca1x—O1x | 2.3500 (3) | Ca1cx—O1ii | 2.381 (2) |
| Ca1x—O1vi | 2.5143 (4) | Ca1cx—O1x | 2.517 (3) |
| Ca1x—O1iii | 2.3500 (3) | Ca1cx—O1vi | 2.578 (2) |
| Ca1x—O1aii | 2.905 (4) | Ca1cx—O1iii | 2.192 (3) |
| Ca1x—O1biii | 2.285 (6) | Ca1cx—O1aii | 2.768 (4) |
| Ca1x—O2iii | 2.4148 (4) | Ca1cx—O1biii | 2.134 (7) |
| Ca1x—O2xi | 2.4148 (4) | Ca1cx—O2iii | 2.533 (3) |
| Ca1xii—O1ii | 2.3500 (3) | Ca1cx—O2xi | 2.328 (3) |
| Ca1xii—O1iv | 2.5143 (4) | | |
| | | |
| O1ii—Al1i—O1iii | 101.039 (19) | O1i—Ca1ai—O3i | 92.8 (2) |
| O1ii—Al1i—O1iv | 101.039 (19) | O1xv—Ca1ai—O1xiii | 63.18 (6) |
| O1ii—Al1i—O2i | 116.97 (2) | O1xv—Ca1ai—O1xiv | 58.45 (4) |
| O1ii—Al1i—O3i | 68.26 (18) | O1xv—Ca1ai—O2xvi | 114.17 (8) |
| O1iii—Al1i—O1iv | 101.039 (19) | O1xv—Ca1ai—O2ai | 78.5 (3) |
| O1iii—Al1i—O2i | 116.97 (2) | O1xv—Ca1ai—O3i | 141.4 (2) |
| O1iii—Al1i—O3i | 169.27 (18) | O1xiii—Ca1ai—O1xiv | 105.39 (8) |
| O1iv—Al1i—O2i | 116.97 (2) | O1xiii—Ca1ai—O2xvi | 96.45 (8) |
| O1iv—Al1i—O3i | 82.41 (18) | O1xiii—Ca1ai—O2ai | 83.6 (3) |
| O2i—Al1i—O3i | 69.34 (18) | O1xiii—Ca1ai—O3i | 98.8 (2) |
| O1iv—Al1ai—O3i | 114.7 (3) | O1xiv—Ca1ai—O2xvi | 71.19 (6) |
| O1iv—Al1ai—O1aii | 107.0 (2) | O1xiv—Ca1ai—O2ai | 122.5 (3) |
| O1iv—Al1ai—O1biii | 68.72 (16) | O1xiv—Ca1ai—O3i | 155.2 (2) |
| O1iv—Al1ai—O2ai | 117.5 (3) | O2xvi—Ca1ai—O2ai | 165.9 (3) |
| O3i—Al1ai—O1aii | 88.2 (3) | O2xvi—Ca1ai—O3i | 101.0 (2) |
| O3i—Al1ai—O1biii | 171.9 (3) | O2ai—Ca1ai—O3i | 65.2 (3) |
| O3i—Al1ai—O2ai | 94.2 (4) | O1—Ca1b—O1vii | 147.84 (7) |
| O1aii—Al1ai—O1biii | 83.7 (2) | O1—Ca1b—O1viii | 55.81 (2) |
| O1aii—Al1ai—O2ai | 129.2 (4) | O1—Ca1b—O1ix | 93.51 (4) |
| O1biii—Al1ai—O2ai | 90.3 (3) | O1—Ca1b—O2 | 96.54 (2) |
| O1i—Al2ii—O1v | 104.553 (17) | O1—Ca1b—O2ii | 79.913 (17) |
| O1i—Al2ii—O1vi | 111.985 (17) | O1—Ca1b—O3i | 88.24 (18) |
| O1i—Al2ii—O1aii | 104.51 (12) | O1vii—Ca1b—O1viii | 93.51 (4) |
| O1v—Al2ii—O1vi | 111.985 (17) | O1vii—Ca1b—O1ix | 55.81 (2) |
| O1v—Al2ii—O1aii | 105.73 (12) | O1vii—Ca1b—O2 | 79.913 (17) |
| O1vi—Al2ii—O1aii | 117.04 (12) | O1vii—Ca1b—O2ii | 96.54 (2) |
| O1ii—Al2ii—O1i | 111.985 (17) | O1vii—Ca1b—O3i | 123.85 (19) |
| O1ii—Al2ii—O1v | 111.985 (17) | O1viii—Ca1b—O1ix | 50.65 (2) |
| O1ii—Al2ii—O1vi | 104.553 (17) | O1viii—Ca1b—O2 | 102.88 (5) |
| O1i—Al2ii—O1v | 104.553 (17) | O1viii—Ca1b—O2ii | 64.99 (3) |
| O1i—Al2ii—O1vi | 111.985 (17) | O1viii—Ca1b—O3i | 137.97 (18) |
| O1v—Al2ii—O1vi | 111.985 (17) | O1ix—Ca1b—O2 | 64.99 (3) |
| O1—Ca1—O1vii | 173.462 (14) | O1ix—Ca1b—O2ii | 102.88 (5) |
| O1—Ca1—O1viii | 68.456 (11) | O1ix—Ca1b—O3i | 167.14 (19) |
| O1—Ca1—O1ix | 117.555 (12) | O2—Ca1b—O2ii | 167.31 (7) |
| O1—Ca1—O2 | 97.711 (12) | O2—Ca1b—O3i | 102.16 (19) |
| O1—Ca1—O2ii | 80.788 (12) | O2ii—Ca1b—O3i | 89.97 (18) |
| O1—Ca1—O3i | 73.44 (14) | O1x—Ca1cx—O1vi | 65.04 (6) |
| O1vii—Ca1—O1viii | 117.555 (12) | O1x—Ca1cx—O2iii | 75.39 (8) |
| O1vii—Ca1—O1ix | 68.456 (11) | O1x—Ca1cx—O2xi | 95.49 (11) |
| O1vii—Ca1—O2 | 80.788 (12) | O1x—Ca1cx—O1aii | 117.97 (12) |
| O1vii—Ca1—O2ii | 97.711 (12) | O1x—Ca1cx—O1biii | 163.8 (2) |
| O1vii—Ca1—O3i | 100.03 (14) | O1vi—Ca1cx—O2iii | 74.33 (9) |
| O1viii—Ca1—O1ix | 66.496 (12) | O1vi—Ca1cx—O2xi | 127.57 (12) |
| O1viii—Ca1—O2 | 126.549 (14) | O1vi—Ca1cx—O1aii | 69.18 (9) |
| O1viii—Ca1—O2ii | 77.556 (13) | O1vi—Ca1cx—O1biii | 127.9 (2) |
| O1viii—Ca1—O3i | 134.52 (14) | O2iii—Ca1cx—O2xi | 150.66 (10) |
| O1ix—Ca1—O2 | 77.556 (13) | O2iii—Ca1cx—O1aii | 127.95 (14) |
| O1ix—Ca1—O2ii | 126.549 (14) | O2iii—Ca1cx—O1biii | 97.7 (2) |
| O1ix—Ca1—O3i | 157.65 (14) | O2xi—Ca1cx—O1aii | 81.11 (12) |
| O2—Ca1—O2ii | 153.735 (16) | O2xi—Ca1cx—O1biii | 83.41 (19) |
| O2—Ca1—O3i | 81.77 (14) | O1aii—Ca1cx—O1biii | 78.0 (2) |
| O2ii—Ca1—O3i | 72.62 (14) | Ca1x—O1ii—Ca1xii | 98.221 (13) |
| O1ii—Ca1x—O1iii | 68.456 (11) | Ca1x—O1ii—Al1i | 92.368 (14) |
| O1ii—Ca1x—O1x | 117.555 (12) | Ca1x—O1ii—Al2ii | 94.475 (16) |
| O1ii—Ca1x—O1vi | 66.496 (12) | Ca1xii—O1ii—Al1i | 98.052 (16) |
| O1ii—Ca1x—O2iii | 126.548 (14) | Ca1xii—O1ii—Al2ii | 122.736 (17) |
| O1ii—Ca1x—O2xi | 77.556 (13) | Al1i—O1ii—Al2ii | 136.984 (19) |
| O1iii—Ca1x—O1x | 173.462 (14) | Ca1xi—O1iii—Ca1x | 98.221 (13) |
| O1iii—Ca1x—O1vi | 117.555 (12) | Ca1xi—O1iii—Al1i | 92.368 (14) |
| O1iii—Ca1x—O2iii | 97.711 (12) | Ca1xi—O1iii—Al2iii | 94.475 (16) |
| O1iii—Ca1x—O2xi | 80.788 (12) | Ca1x—O1iii—Al1i | 98.052 (16) |
| O1x—Ca1x—O1vi | 68.456 (11) | Ca1x—O1iii—Al2iii | 122.736 (17) |
| O1x—Ca1x—O2iii | 80.788 (12) | Al1i—O1iii—Al2iii | 136.985 (19) |
| O1x—Ca1x—O2xi | 97.711 (12) | Ca1xii—O1aii—Al2ii | 121.03 (18) |
| O1vi—Ca1x—O2iii | 77.556 (13) | Ca1xii—O1aii—Ca1cx | 88.38 (13) |
| O1vi—Ca1x—O2xi | 126.549 (14) | Ca1xii—O1aii—Al1ai | 97.4 (2) |
| O2iii—Ca1x—O2xi | 153.735 (16) | Al2ii—O1aii—Ca1cx | 83.19 (15) |
| O1i—Ca1i—O1xv | 117.555 (12) | Al2ii—O1aii—Al1ai | 139.9 (3) |
| O1i—Ca1i—O1xiii | 173.462 (14) | Ca1cx—O1aii—Al1ai | 111.1 (2) |
| O1i—Ca1i—O1xiv | 68.456 (11) | Ca1xi—O1biii—Al2iii | 101.9 (3) |
| O1i—Ca1i—O2i | 97.711 (12) | Ca1xi—O1biii—Ca1cx | 102.8 (2) |
| O1i—Ca1i—O2xvi | 80.788 (12) | Ca1xi—O1biii—Al1ai | 89.4 (2) |
| O1i—Ca1i—O3i | 83.81 (16) | Al2iii—O1biii—Ca1cx | 134.5 (4) |
| O1xv—Ca1i—O1xiii | 68.456 (11) | Al2iii—O1biii—Al1ai | 130.6 (3) |
| O1xv—Ca1i—O1xiv | 66.496 (12) | Ca1cx—O1biii—Al1ai | 87.2 (2) |
| O1xv—Ca1i—O2i | 77.556 (13) | Ca1i—O2i—Ca1xvii | 99.237 (15) |
| O1xv—Ca1i—O2xvi | 126.548 (14) | Ca1i—O2i—Ca1xv | 99.237 (15) |
| O1xv—Ca1i—O3i | 136.96 (16) | Ca1i—O2i—Al1i | 118.41 (2) |
| O1xiii—Ca1i—O1xiv | 117.555 (12) | Ca1xvii—O2i—Ca1xv | 99.237 (15) |
| O1xiii—Ca1i—O2i | 80.788 (12) | Ca1xvii—O2i—Al1i | 118.41 (2) |
| O1xiii—Ca1i—O2xvi | 97.711 (12) | Ca1xv—O2i—Al1i | 118.41 (2) |
| O1xiii—Ca1i—O3i | 89.89 (16) | Ca1xvii—O2ai—Ca1xv | 94.3 (3) |
| O1xiv—Ca1i—O2i | 126.549 (14) | Ca1xvii—O2ai—Ca1ai | 101.3 (4) |
| O1xiv—Ca1i—O2xvi | 77.556 (13) | Ca1xvii—O2ai—Al1ai | 111.2 (4) |
| O1xiv—Ca1i—O3i | 151.43 (16) | Ca1xv—O2ai—Ca1ai | 104.0 (3) |
| O2i—Ca1i—O2xvi | 153.735 (16) | Ca1xv—O2ai—Al1ai | 143.3 (5) |
| O2i—Ca1i—O3i | 62.07 (16) | Ca1ai—O2ai—Al1ai | 96.5 (4) |
| O2xvi—Ca1i—O3i | 91.79 (16) | Ca1ai—O3i—Ca1b | 145.5 (4) |
| O1i—Ca1ai—O1xv | 107.60 (8) | Ca1ai—O3i—Al1ai | 85.6 (3) |
| O1i—Ca1ai—O1xiii | 168.36 (10) | Ca1b—O3i—Al1ai | 128.8 (4) |
| O1i—Ca1ai—O1xiv | 62.98 (6) | Ca1—O3i—Ca1i | 150.6 (3) |
| O1i—Ca1ai—O2xvi | 80.61 (8) | Ca1—O3i—Al1i | 126.3 (3) |
| O1i—Ca1ai—O2ai | 102.0 (3) | Ca1i—O3i—Al1i | 83.0 (2) |
| Symmetry codes: (i) −x+3/4, −z+1/4, y+1/4; (ii) z, −x, −y+1/2; (iii) y+1/2, z−1/2, x+1/2; (iv) −x+1/2, y, −z+1; (v) x+1/4, z−3/4, y+1/4; (vi) −z+1, x−1/2, −y+1/2; (vii) x, −y, −z+1/2; (viii) −y, z−1/2, −x+1/2; (ix) −y, −z+1/2, x; (x) −y+1/2, −z, x+1/2; (xi) −x+1/2, −y, z+1/2; (xii) −z+1/2, −x, y+1/2; (xiii) −x+3/4, z−1/4, −y+1/4; (xiv) y+3/4, x−1/4, z−1/4; (xv) y+3/4, −x+1/4, −z+3/4; (xvi) −z+3/4, y−1/4, −x+1/4; (xvii) z+1/4, y+1/4, x+1/4. |
Experimental details
| Crystal data |
| Chemical formula | Al14Ca12O33 |
| Mr | 1386.66 |
| Crystal system, space group | Cubic, I43d |
| Temperature (K) | 298 |
| a (Å) | 11.989 (3) |
| V (Å3) | 1723.2 (7) |
| Z | 2 |
| Radiation type | Synchrotron, λ = 0.67954 Å |
| µ (mm−1) | 1.99 |
| Crystal size (mm) | 0.07 × 0.07 × 0.07 × 0.03 (radius) |
| |
| Data collection |
| Diffractometer | Tsukuba BL14A four-circle
diffractometer |
| Absorption correction | For a sphere
Transmission coefficient for spheres tabulated in International Table
II(1972) Table 5.3.6B was interpolated with Lagrangian method |
| Tmin, Tmax | 0.908, 0.909 |
No. of measured, independent and
observed [F > 3.0σ(F)] reflections | 10790, 3077, 3020 |
| Rint | 0.012 |
| (sin θ/λ)max (Å−1) | 1.368 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.016, 0.024, 1.09 |
| No. of reflections | 3020 |
| No. of parameters | 66 |
| Δρmax, Δρmin (e Å−3) | 0.34, −0.39 |
Selected geometric parameters (Å, º) top| Al1i—O1ii | 1.7746 (4) | Al1ai—O2ai | 1.551 (8) |
| Al1i—O1iii | 1.7746 (4) | Al1ai—O3i | 1.762 (9) |
| Al1i—O1iv | 1.7746 (4) | Ca1—O3i | 3.141 (7) |
| Al1i—O2i | 1.7342 (4) | Ca1i—O3i | 2.699 (7) |
| Al1ai—O1iv | 1.839 (3) | Ca1ai—O3i | 2.331 (8) |
| Al1ai—O1aii | 1.431 (5) | Ca1b—O3i | 2.361 (8) |
| | | |
| O1ii—Al1i—O1iii | 101.039 (19) | O1iv—Al1ai—O2ai | 117.5 (3) |
| O1ii—Al1i—O1iv | 101.039 (19) | O3i—Al1ai—O1aii | 88.2 (3) |
| O1ii—Al1i—O2i | 116.97 (2) | O3i—Al1ai—O1biii | 171.9 (3) |
| O1ii—Al1i—O3i | 68.26 (18) | O3i—Al1ai—O2ai | 94.2 (4) |
| O1iii—Al1i—O1iv | 101.039 (19) | O1aii—Al1ai—O1biii | 83.7 (2) |
| O1iii—Al1i—O2i | 116.97 (2) | O1aii—Al1ai—O2ai | 129.2 (4) |
| O1iii—Al1i—O3i | 169.27 (18) | O1biii—Al1ai—O2ai | 90.3 (3) |
| O1iv—Al1i—O2i | 116.97 (2) | Ca1ai—O3i—Ca1b | 145.5 (4) |
| O1iv—Al1i—O3i | 82.41 (18) | Ca1ai—O3i—Al1ai | 85.6 (3) |
| O2i—Al1i—O3i | 69.34 (18) | Ca1b—O3i—Al1ai | 128.8 (4) |
| O1iv—Al1ai—O3i | 114.7 (3) | Ca1—O3i—Ca1i | 150.6 (3) |
| O1iv—Al1ai—O1aii | 107.0 (2) | Ca1—O3i—Al1i | 126.3 (3) |
| O1iv—Al1ai—O1biii | 68.72 (16) | Ca1i—O3i—Al1i | 83.0 (2) |
| Symmetry codes: (i) −x+3/4, −z+1/4, y+1/4; (ii) z, −x, −y+1/2; (iii) y+1/2, z−1/2, x+1/2; (iv) −x+1/2, y, −z+1. |

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
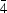 site at the centre of each cage. The difference-density map revealed two large peaks corresponding to displaced Ca ions. The positive ions close to O3 are displaced and one-to-one correspondence was found between one of the four equivalent O3 ions and the displaced ions. When an O3 ion is present in the cage the Al ion at the 16c position moves 0.946 (3) Å toward the O3 ion. One of the Al-O bonds is broken and a new Al-O3 bond is created. The result is an AlO4 tetrahedron that is quite deformed. The three O1 ions and the O2 ion of the destroyed AlO4 tetrahedron are forcibly displaced. O1 and O2 have two and one displaced ions, respectively. The local structure of the cage occupied by one of the four O3 ions was determined by calculating the electrostatic potential and electric field in the deformed cage, although the position of one of the displaced O1 ions was not clearly identified.
site at the centre of each cage. The difference-density map revealed two large peaks corresponding to displaced Ca ions. The positive ions close to O3 are displaced and one-to-one correspondence was found between one of the four equivalent O3 ions and the displaced ions. When an O3 ion is present in the cage the Al ion at the 16c position moves 0.946 (3) Å toward the O3 ion. One of the Al-O bonds is broken and a new Al-O3 bond is created. The result is an AlO4 tetrahedron that is quite deformed. The three O1 ions and the O2 ion of the destroyed AlO4 tetrahedron are forcibly displaced. O1 and O2 have two and one displaced ions, respectively. The local structure of the cage occupied by one of the four O3 ions was determined by calculating the electrostatic potential and electric field in the deformed cage, although the position of one of the displaced O1 ions was not clearly identified.
 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig4thm.gif)
![[Figure 5]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig5thm.gif)
![[Figure 6]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig6thm.gif)
![[Figure 7]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig7thm.gif)
![[Figure 8]](http://journals.iucr.org/b/issues/2011/03/00/og5045/og5045fig8thm.gif)

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


