Buy article online - an online subscription or single-article purchase is required to access this article.
This work illustrates the structural relationship between three types of metal squarates as well as the ligand in its acid form and in its monoanion salt. Squaric acid, H2C4O4, is known to have a polymeric layer structure with planar molecules connected through intermolecular hydrogen bonds. The interlayer distance is only 2.649 Å. The crystal of H2NMe2[H3(C4O4)2] is found to contain columns of [H3(C4O4)2−] repeating units, again connected by intermolecular hydrogen bonds. Within the repeated unit, there is a symmetric hydrogen bond connected to two HC4O4 moieties. A new type of metal squarate with M(HC4O4)2(H2O)4 M = MnII, FeII both belong to space group P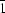 , Z = 1, a = 5.194 (3), b = 7.454 (2), c = 8.901 (2) Å, α = 67.07 (2), β = 77.26 (3), γ = 74.46 (4)°, for MnII is shown to have a layer-type structure, where all [HC4O4] units are bonded into infinite chains via symmetric hydrogen bonds, each (HC4O4)22− ligand bridging two metal ions (μ-2) in a trans fashion. The structurally most well understood metal squarate M(C4O4)(H2O)4 (M = MnII, FeII, CoII, NiII and ZnII, space group C2/c, Z = 4) is again a polymeric chain with C4O42− serving as a bridging ligand between two metal ions (μ-2) in trans positions. A three-dimensional polymeric structure is found to have the formula M(C404)(H2O)2, where C4O42− is a bridging ligand between four metal ions (μ-4). Due to the slight difference in packing, there are two structure types in this category: one is in space group R
, Z = 1, a = 5.194 (3), b = 7.454 (2), c = 8.901 (2) Å, α = 67.07 (2), β = 77.26 (3), γ = 74.46 (4)°, for MnII is shown to have a layer-type structure, where all [HC4O4] units are bonded into infinite chains via symmetric hydrogen bonds, each (HC4O4)22− ligand bridging two metal ions (μ-2) in a trans fashion. The structurally most well understood metal squarate M(C4O4)(H2O)4 (M = MnII, FeII, CoII, NiII and ZnII, space group C2/c, Z = 4) is again a polymeric chain with C4O42− serving as a bridging ligand between two metal ions (μ-2) in trans positions. A three-dimensional polymeric structure is found to have the formula M(C404)(H2O)2, where C4O42− is a bridging ligand between four metal ions (μ-4). Due to the slight difference in packing, there are two structure types in this category: one is in space group R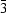 [M = FeII, a = 11.440 (2), c = 14.504 (3) Å, Z = 9], the other is in Pn
[M = FeII, a = 11.440 (2), c = 14.504 (3) Å, Z = 9], the other is in Pn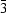 n [M = CoII, a = 16.255 (3) Å, Z = 24]. The structural relationship between all these structures relies heavily on the understanding of intra- and intermolecular hydrogen bonds. The interesting building blocks of each compound will be illustrated. There are tunnels of various sizes in all these structures.
n [M = CoII, a = 16.255 (3) Å, Z = 24]. The structural relationship between all these structures relies heavily on the understanding of intra- and intermolecular hydrogen bonds. The interesting building blocks of each compound will be illustrated. There are tunnels of various sizes in all these structures.

 journal menu
journal menu









 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


