Buy article online - an online subscription or single-article purchase is required to access this article.
The three-dimensional structure of diferric buffalo lactoferrin has been determined at 3.3 Å resolution. The structure was solved by molecular replacement using the coordinates of diferric human lactoferrin as a search model and was refined by simulated annealing (X-PLOR). The final model comprises 5316 protein atoms for all 689 residues, two Fe3+ and two CO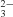 ions. The final R factor was 21.8% for 11 711 reflections in the resolution range 17.0-3.3 Å. The folding of buffalo lactoferrin is essentially similar to that of the other members of the transferrin family. The significant differences are found in the dimensions of the binding cleft and the interlobe orientation. The interlobe interactions are predominantly hydrophobic in nature, thus facilitating the sliding of two lobes owing to external forces. The interdomain interactions are comparable in the N and C lobes.
ions. The final R factor was 21.8% for 11 711 reflections in the resolution range 17.0-3.3 Å. The folding of buffalo lactoferrin is essentially similar to that of the other members of the transferrin family. The significant differences are found in the dimensions of the binding cleft and the interlobe orientation. The interlobe interactions are predominantly hydrophobic in nature, thus facilitating the sliding of two lobes owing to external forces. The interdomain interactions are comparable in the N and C lobes.

 journal menu
journal menu









 Subscribe to Acta Crystallographica Section D: Biological Crystallography
Subscribe to Acta Crystallographica Section D: Biological Crystallography


