Buy article online - an online subscription or single-article purchase is required to access this article.
The crystal structure of 2,4,6-triisopropylbenzenesulfonamide, C
15H
25NO
2S, has been solved from X-ray powder diffraction data collected at 120 (1) K using synchrotron radiation and refined by Rietveld methods. The structure was solved by the application of a Monte Carlo method in which trial structures were generated by random movement of the molecule in the unit cell and assessed using a full-profile-fitting technique. Intramolecular flexibility was introduced into the structure solution in the form of four independent asymmetric rotors, allowing the isopropyl and sulfonamide groups to rotate freely within the molecule. The structure is monoclinic
P2
1/
c,
a = 16.9600 (6),
b = 8.1382 (2),
c = 11.7810 (2) Å, β = 104.777 (2)° with
Z = 4. The molecules are linked by N—H

O hydrogen bonds, with N

O distances of 2.77 (1) and 2.92 (1) Å, into two-dimensional sheets built from
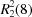
and
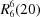
rings.
Supporting information
 | Crystallographic Information File (CIF)
Contains datablocks global, TIBS |
 | Structure factor file (CIF format)
Supplementary material |
CCDC reference: 138582
Data collection: PINCER (CCLRC, 1994); program(s) used to solve structure: OCTOPUS (Tremayne et al. 1997); program(s) used to refine structure: GSAS (Larson et al. 1994).
2,4,6-Tri-isopropylbenzenesulfonamide
top
Crystal data top
| C15H25NO2S | V = 1572.3 (1) Å3 |
| Mr = 283.42 | Z = 4 |
| Monoclinic, P21/c | Dx = 1.200 Mg m−3 |
| a = 16.9600 (6) Å | Synchrotron radiation |
| b = 8.1382 (2) Å | T = 120 K |
| c = 11.7810 (2) Å | white |
| β = 104.777 (2)° | cylinder, 30 × 0.5 mm |
Data collection top
Bede EDR detector
diffractometer | Specimen mounting: quartz capillary |
| Radiation source: synchrotron | Data collection mode: transmission |
| Silicon 111 monochromator | 2θmin = 3.00°, 2θmax = 49.99°, 2θstep = 0.01° |
Refinement top
| Rp = 0.050 | 143 parameters |
| Rwp = 0.071 | 120 restraints |
| R(F2) = 0.0997 | Restr |
| Excluded region(s): 7 small regions excluded due to ice peaks | Preferred orientation correction: none |
| Profile function: pseudovoigt | |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| S1 | 0.0849 (5) | 0.3869 (9) | 0.6677 (6) | 0.072 (4)* | |
| C1 | 0.186 (1) | 0.481 (3) | 0.709 (2) | 0.039 (4)* | |
| C2 | 0.199 (1) | 0.627 (3) | 0.647 (2) | 0.039 (4)* | |
| C3 | 0.274 (1) | 0.705 (2) | 0.686 (2) | 0.039 (4)* | |
| C4 | 0.337 (1) | 0.634 (2) | 0.773 (2) | 0.039 (4)* | |
| C5 | 0.323 (1) | 0.497 (3) | 0.834 (1) | 0.039 (4)* | |
| C6 | 0.247 (1) | 0.414 (3) | 0.798 (2) | 0.039 (4)* | |
| C7 | 0.1306 (7) | 0.715 (1) | 0.551 (1) | 0.064 (6)* | |
| C8 | 0.4210 (9) | 0.723 (1) | 0.811 (1) | 0.064 (6)* | |
| C9 | 0.2456 (6) | 0.241 (2) | 0.857 (1) | 0.064 (6)* | |
| O1 | 0.0724 (8) | 0.349 (2) | 0.550 (1) | 0.046 (5)* | |
| O2 | 0.0771 (8) | 0.265 (2) | 0.741 (1) | 0.046 (5)* | |
| N1 | 0.0199 (9) | 0.540 (2) | 0.681 (1) | 0.017 (7)* | |
| C71 | 0.1659 (9) | 0.715 (2) | 0.447 (1) | 0.063 (4)* | |
| C72 | 0.1057 (8) | 0.876 (2) | 0.593 (1) | 0.063 (4)* | |
| C81 | 0.4853 (9) | 0.625 (2) | 0.775 (1) | 0.063 (4)* | |
| C82 | 0.4501 (9) | 0.759 (2) | 0.940 (1) | 0.063 (4)* | |
| C91 | 0.2575 (9) | 0.265 (2) | 0.985 (1) | 0.063 (4)* | |
| C92 | 0.3061 (9) | 0.123 (2) | 0.830 (1) | 0.063 (4)* | |
| H3 | 0.285 (4) | 0.824 (9) | 0.647 (9) | 0.05* | |
| H5 | 0.364 (4) | 0.465 (9) | 0.920 (6) | 0.05* | |
| H7 | 0.079 (1) | 0.630 (2) | 0.535 (2) | 0.05* | |
| H8 | 0.412 (1) | 0.840 (2) | 0.762 (2) | 0.05* | |
| H9 | 0.183 (1) | 0.195 (3) | 0.818 (2) | 0.05* | |
| H711 | 0.166 (7) | 0.589 (3) | 0.414 (7) | 0.07* | |
| H712 | 0.128 (5) | 0.794 (9) | 0.378 (5) | 0.07* | |
| H713 | 0.228 (3) | 0.763 (9) | 0.472 (4) | 0.07* | |
| H721 | 0.159 (3) | 0.960 (6) | 0.613 (9) | 0.07* | |
| H722 | 0.056 (6) | 0.930 (8) | 0.524 (5) | 0.07* | |
| H723 | 0.084 (7) | 0.856 (4) | 0.672 (6) | 0.07* | |
| H811 | 0.467 (4) | 0.495 (3) | 0.768 (9) | 0.07* | |
| H812 | 0.492 (6) | 0.670 (9) | 0.690 (6) | 0.07* | |
| H813 | 0.543 (2) | 0.638 (9) | 0.842 (6) | 0.07* | |
| H821 | 0.477 (6) | 0.648 (4) | 0.987 (2) | 0.07* | |
| H822 | 0.496 (4) | 0.858 (8) | 0.954 (2) | 0.07* | |
| H823 | 0.398 (2) | 0.799 (9) | 0.974 (3) | 0.07* | |
| H911 | 0.198 (1) | 0.277 (9) | 1.006 (2) | 0.07* | |
| H912 | 0.290 (6) | 0.159 (7) | 1.033 (2) | 0.07* | |
| H913 | 0.293 (6) | 0.377 (8) | 1.014 (3) | 0.07* | |
| H921 | 0.294 (6) | −0.001 (3) | 0.859 (9) | 0.07* | |
| H922 | 0.301 (6) | 0.123 (9) | 0.735 (2) | 0.07* | |
| H923 | 0.368 (1) | 0.160 (9) | 0.877 (9) | 0.07* | |
Geometric parameters (Å, º) top
| S1—O1 | 1.38 (1) | C3—H3 | 1.11 (1) |
| S1—O2 | 1.34 (1) | C5—H5 | 1.10 (1) |
| S1—N1 | 1.70 (1) | C7—H7 | 1.09 (1) |
| S1—C1 | 1.83 (1) | C8—H8 | 1.10 (1) |
| C2—C7 | 1.57 (1) | C9—H9 | 1.11 (3) |
| C4—C8 | 1.56 (1) | C71—H711 | 1.10 (1) |
| C6—C9 | 1.57 (1) | C71—H712 | 1.10 (1) |
| C7—C71 | 1.494 (7) | C71—H713 | 1.10 (1) |
| C7—C72 | 1.499 (7) | C72—H721 | 1.11 (1) |
| C8—C81 | 1.497 (7) | C72—H722 | 1.10 (1) |
| C8—C82 | 1.502 (7) | C72—H723 | 1.10 (1) |
| C9—C91 | 1.484 (7) | C81—H811 | 1.10 (1) |
| C9—C92 | 1.497 (7) | C81—H812 | 1.10 (1) |
| N1—H1 | 1.003 (13) | C81—H813 | 1.09 (1) |
| N1—H2 | 1.001 (13) | C82—H821 | 1.10 (1) |
| N1—O1i | 2.92 (1) | C82—H822 | 1.10 (1) |
| N1—O2ii | 2.77 (1) | C82—H823 | 1.11 (1) |
| C1—C2 | 1.44 (1) | C91—H911 | 1.10 (1) |
| C1—C6 | 1.38 (1) | C91—H912 | 1.10 (1) |
| C2—C3 | 1.39 (1) | C91—H913 | 1.10 (1) |
| C3—C4 | 1.40 (1) | C92—H921 | 1.10 (1) |
| C4—C5 | 1.38 (1) | C92—H922 | 1.10 (1) |
| C5—C6 | 1.42 (1) | C92—H923 | 1.10 (1) |
| | | |
| O1—S1—O2 | 117 (1) | C6—C9—H9 | 104 (1) |
| O1—S1—N1 | 109 (1) | C91—C9—H9 | 109 (1) |
| C1—S1—O1 | 105 (1) | C92—C9—H9 | 109 (1) |
| O2—S1—N1 | 108 (1) | C7—C71—H711 | 109 (1) |
| C1—S1—O2 | 112 (1) | C7—C71—H712 | 109 (1) |
| C1—S1—N1 | 105 (1) | C7—C71—H713 | 110 (1) |
| S1—C1—C2 | 118 (1) | H711—C71—H712 | 110 (1) |
| S1—C1—C6 | 120 (1) | H711—C71—H713 | 110 (1) |
| C1—C2—C7 | 124 (1) | H712—C71—H713 | 109 (1) |
| C3—C2—C7 | 118 (1) | C7—C72—H721 | 109 (1) |
| C1—C6—C9 | 126 (1) | C7—C72—H722 | 109 (1) |
| C5—C6—C9 | 114 (1) | C7—C72—H723 | 109 (1) |
| C3—C4—C8 | 119 (1) | H721—C72—H722 | 110 (1) |
| C5—C4—C8 | 119 (1) | H721—C72—H723 | 110 (1) |
| C2—C7—C71 | 102 (1) | H722—C72—H723 | 109 (1) |
| C2—C7—C72 | 112 (1) | C8—C81—H811 | 109 (1) |
| C71—C7—C72 | 118 (1) | C8—C81—H812 | 109 (1) |
| C4—C8—C81 | 110 (1) | C8—C81—H813 | 109 (1) |
| C4—C8—C82 | 115 (1) | H811—C81—H812 | 110 (1) |
| C81—C8—C82 | 108 (1) | H811—C81—H813 | 109 (1) |
| C6—C9—C91 | 109 (1) | H812—C81—H813 | 110 (1) |
| C6—C9—C92 | 113 (1) | C8—C82—H821 | 110 (1) |
| C91—C9—C92 | 112 (1) | C8—C82—H822 | 109 (1) |
| C2—C1—C6 | 122 (1) | C8—C82—H823 | 110 (1) |
| C1—C2—C3 | 117 (1) | H821—C82—H822 | 110 (1) |
| C2—C3—C4 | 121 (1) | H821—C82—H823 | 109 (1) |
| C3—C4—C5 | 121 (1) | H822—C82—H823 | 109 (1) |
| C4—C5—C6 | 119 (1) | C9—C91—H911 | 110 (1) |
| C1—C6—C5 | 119 (1) | C9—C91—H912 | 110 (1) |
| C2—C3—H3 | 120 (1) | C9—C91—H913 | 110 (1) |
| C4—C3—H3 | 119 (1) | H911—C91—H912 | 109 (1) |
| C4—C5—H5 | 121 (1) | H911—C91—H913 | 109 (1) |
| C6—C5—H5 | 118 (1) | H912—C91—H913 | 109 (1) |
| C2—C7—H7 | 104 (1) | C9—C92—H921 | 109 (1) |
| C71—C7—H7 | 109 (1) | C9—C92—H922 | 110 (1) |
| C72—C7—H7 | 109 (1) | C9—C92—H923 | 110 (1) |
| C4—C8—H8 | 105 (1) | H921—C92—H922 | 110 (1) |
| C81—C8—H8 | 109 (1) | H921—C92—H923 | 110 (1) |
| C82—C8—H8 | 109 (1) | H922—C92—H923 | 111 (1) |
| | | |
| C1—C2—C7—C71 | −123 (1) | C1—C2—C7—C72 | 109 (1) |
| C3—C4—C8—C81 | −113 (1) | C3—C4—C8—C82 | 124 (1) |
| C1—C6—C9—C91 | −122 (1) | C1—C6—C9—C92 | 113 (1) |
| C2—C1—S1—O1 | 59 (1) | C2—C1—S1—O2 | −172 (1) |
| C2—C1—S1—N1 | −55 (1) | | |
| Symmetry codes: (i) −x, −y+1, −z+1; (ii) −x, y+1/2, −z+3/2. |

 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
 O hydrogen bonds, with N
O hydrogen bonds, with N O distances of 2.77 (1) and 2.92 (1) Å, into two-dimensional sheets built from
O distances of 2.77 (1) and 2.92 (1) Å, into two-dimensional sheets built from 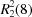 and
and 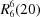 rings.
rings. O hydrogen bonds, with N
O hydrogen bonds, with N O distances of 2.77 (1) and 2.92 (1) Å, into two-dimensional sheets built from
O distances of 2.77 (1) and 2.92 (1) Å, into two-dimensional sheets built from 
 journal menu
journal menu









 Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials
Subscribe to Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials


