Buy article online - an online subscription or single-article purchase is required to access this article.
The structure of hexafluoroiodium(VII) hexafluoroarsenate, IF
6AsF
6, has been determined by X-ray diffraction using a single crystal grown from a saturated solution in anhydrous HF. IF
6AsF
6 crystallizes in the cubic space group
Pa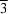
with a simple NaCl-like ionic packing. The I and As atoms occupy the 4
a and 4
b Wyckoff positions, respectively, with
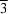
symmetry.
Supporting information
IF6AsF6 was synthesized by the oxidation of iodium pentafluoride by [Ag(HF)n]2+, prepared by reaction of AgF2 with AsF5 in anhydrous HF: 2[Ag(HF)n]2+(AsF6−)2] + IF5 = 2 A g+AsF6− + IF6AsF6 + AsF5. Colourless block-shaped crystals of the title compound were grown from a saturated solution in anhydrous HF. Raman spectra recorded on single crystals were in agreement with literature data for IF6AsF6 (Beaton, 1966; Christe & Sawodny, 1967).
Data collection: CrystalClear (Rigaku Corporation, 1999); cell refinement: CrystalClear; data reduction: CrystalClear; program(s) used to solve structure: PATTY in DIRDIF92 (Beurskens et al., 1992); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); software used to prepare material for publication: WinGX (Version 1.70; Farrugia, 1999), enCIFer (Version 1.2; Allen et
al., 2004) and Diamond (Version 3.1; Bergerhoff et al.,
1996).
Crystal data top
| AsF12I | Dx = 3.428 Mg m−3 |
| Mr = 429.82 | Mo Kα radiation, λ = 0.71069 Å |
| Cubic, Pa3 | Cell parameters from 48 reflections |
| Hall symbol: -P 2ac 2ab 3 | θ = 3.1–29.1° |
| a = 9.409 (2) Å | µ = 7.96 mm−1 |
| V = 832.9 (3) Å3 | T = 200 K |
| Z = 4 | Chunk, colorless |
| F(000) = 776 | 0.3 × 0.3 × 0.2 mm |
Data collection top
Rigaku Mercury CCD
diffractometer | 230 reflections with I > 2σ(I) |
| ω scans | Rint = 0.018 |
Absorption correction: multi-scan
(Blessing, 1995) | θmax = 29.1°, θmin = 3.8° |
| Tmin = 0.115, Tmax = 0.2 | h = −12→11 |
| 3317 measured reflections | k = −5→11 |
| 354 independent reflections | l = −12→12 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.021 | w = 1/[σ2(Fo2) + (0.0225P)2]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.063 | (Δ/σ)max < 0.001 |
| S = 1.10 | Δρmax = 0.72 e Å−3 |
| 354 reflections | Δρmin = −0.60 e Å−3 |
| 24 parameters | Extinction correction: SHELXL97, Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| 0 restraints | Extinction coefficient: 0.0023 (4) |
Crystal data top
| AsF12I | Z = 4 |
| Mr = 429.82 | Mo Kα radiation |
| Cubic, Pa3 | µ = 7.96 mm−1 |
| a = 9.409 (2) Å | T = 200 K |
| V = 832.9 (3) Å3 | 0.3 × 0.3 × 0.2 mm |
Data collection top
Rigaku Mercury CCD
diffractometer | 354 independent reflections |
Absorption correction: multi-scan
(Blessing, 1995) | 230 reflections with I > 2σ(I) |
| Tmin = 0.115, Tmax = 0.2 | Rint = 0.018 |
| 3317 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.021 | 24 parameters |
| wR(F2) = 0.063 | 0 restraints |
| S = 1.10 | Δρmax = 0.72 e Å−3 |
| 354 reflections | Δρmin = −0.60 e Å−3 |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| I | 0.0000 | 0.0000 | 0.0000 | 0.0214 (2) | |
| As | 0.0000 | 0.5000 | 1.0000 | 0.0199 (2) | |
| F1 | −0.13523 (17) | 0.06318 (17) | −0.11527 (15) | 0.0337 (5) | |
| F2 | −0.14038 (17) | 0.55931 (17) | 1.10099 (15) | 0.0351 (5) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| I | 0.0214 (2) | 0.0214 (2) | 0.0214 (2) | −0.00026 (10) | −0.00026 (10) | −0.00026 (10) |
| As | 0.0199 (2) | 0.0199 (2) | 0.0199 (2) | −0.00063 (16) | 0.00063 (16) | 0.00063 (16) |
| F1 | 0.0299 (10) | 0.0397 (11) | 0.0315 (10) | 0.0035 (8) | −0.0043 (8) | 0.0030 (8) |
| F2 | 0.0323 (10) | 0.0381 (11) | 0.0349 (10) | 0.0049 (8) | 0.0066 (8) | −0.0016 (8) |
Geometric parameters (Å, º) top
| I—F1i | 1.7744 (17) | As—F2vi | 1.7200 (17) |
| I—F1ii | 1.7744 (17) | As—F2vii | 1.7200 (17) |
| I—F1 | 1.7744 (17) | As—F2viii | 1.7200 (17) |
| I—F1iii | 1.7744 (17) | As—F2ix | 1.7200 (17) |
| I—F1iv | 1.7744 (17) | As—F2 | 1.7200 (17) |
| I—F1v | 1.7744 (17) | As—F2x | 1.7200 (17) |
| | | |
| F1i—I—F1ii | 89.62 (7) | F2vi—As—F2vii | 90.24 (8) |
| F1i—I—F1 | 90.38 (7) | F2vi—As—F2viii | 180.00 (7) |
| F1ii—I—F1 | 89.62 (7) | F2vii—As—F2viii | 89.76 (8) |
| F1i—I—F1iii | 89.62 (7) | F2vi—As—F2ix | 89.76 (8) |
| F1ii—I—F1iii | 90.38 (7) | F2vii—As—F2ix | 180.00 (7) |
| F1—I—F1iii | 180.00 (12) | F2viii—As—F2ix | 90.24 (8) |
| F1i—I—F1iv | 180.00 (11) | F2vi—As—F2 | 90.24 (8) |
| F1ii—I—F1iv | 90.38 (7) | F2vii—As—F2 | 90.24 (8) |
| F1—I—F1iv | 89.62 (7) | F2viii—As—F2 | 89.76 (8) |
| F1iii—I—F1iv | 90.38 (7) | F2ix—As—F2 | 89.76 (8) |
| F1i—I—F1v | 90.38 (7) | F2vi—As—F2x | 89.76 (8) |
| F1ii—I—F1v | 180.00 (7) | F2vii—As—F2x | 89.76 (8) |
| F1—I—F1v | 90.38 (7) | F2viii—As—F2x | 90.24 (8) |
| F1iii—I—F1v | 89.62 (7) | F2ix—As—F2x | 90.24 (8) |
| F1iv—I—F1v | 89.62 (7) | F2—As—F2x | 180.0 |
| Symmetry codes: (i) z, x, y; (ii) −y, −z, −x; (iii) −x, −y, −z; (iv) −z, −x, −y; (v) y, z, x; (vi) −z+1, x+1/2, −y+3/2; (vii) y−1/2, −z+3/2, −x+1; (viii) z−1, −x+1/2, y+1/2; (ix) −y+1/2, z−1/2, x+1; (x) −x, −y+1, −z+2. |
Experimental details
| Crystal data |
| Chemical formula | AsF12I |
| Mr | 429.82 |
| Crystal system, space group | Cubic, Pa3 |
| Temperature (K) | 200 |
| a (Å) | 9.409 (2) |
| V (Å3) | 832.9 (3) |
| Z | 4 |
| Radiation type | Mo Kα |
| µ (mm−1) | 7.96 |
| Crystal size (mm) | 0.3 × 0.3 × 0.2 |
| |
| Data collection |
| Diffractometer | Rigaku Mercury CCD
diffractometer |
| Absorption correction | Multi-scan
(Blessing, 1995) |
| Tmin, Tmax | 0.115, 0.2 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 3317, 354, 230 |
| Rint | 0.018 |
| (sin θ/λ)max (Å−1) | 0.685 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.021, 0.063, 1.10 |
| No. of reflections | 354 |
| No. of parameters | 24 |
| Δρmax, Δρmin (e Å−3) | 0.72, −0.60 |

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry
The full text of this article is available to subscribers to the journal.
If you have already registered and are using a computer listed in your registration details, please email
support@iucr.org for assistance.
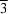 with a simple NaCl-like ionic packing. The I and As atoms occupy the 4a and 4b Wyckoff positions, respectively, with
with a simple NaCl-like ionic packing. The I and As atoms occupy the 4a and 4b Wyckoff positions, respectively, with 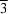 symmetry.
symmetry.
 journal menu
journal menu








![[Figure 1]](https://journals.iucr.org/c/issues/2006/07/00/bc3005/bc3005fig1thm.gif)

 Subscribe to Acta Crystallographica Section C: Structural Chemistry
Subscribe to Acta Crystallographica Section C: Structural Chemistry



Until now, the single-crystal X-ray structure of IF6Sb2F11 represented the only detailed crystallographic study of salts containing the IF6+ cation (Lehmann et al., 2004). IF6AsF6 was first prepared by the reaction of IF7 with AsF5 (Seel & Detmer, 1958, 1959). Powder X-ray diffraction studies of IF6AsF6 were performed later (Beaton, 1966; Christe & Sawodny, 1967). In this work, the structure of IF6AsF6 was determined by single-crystal technique to obtain more precise structural data and to allow a comparison of the cation geometry with that previously found in IF6Sb2F11.
IF6AsF6 crystallizes in the cubic space group Pa3, in agreement with the published data based on X-ray powder diffraction experiments. The structure consists of alternating IF6+ and AsF6− octahedra packed in an NaCl-like arrangement (Fig. 1). The closest As···I distance is 4.705 (1) Å. The I and As atoms are both located at sites with 3 symmetry (the 4a and 4b Wyckoff positions, respectively), resulting in six equal I—F distances and six equal As—F distances. The I—F bond lengths [1.7744 (17) Å] are in the same range as those in IF6Sb2F11 (1.767–1.782 Å at 173 K; Lehmann et al., 2004). The As—F bond lengths 1.7200 (17) Å] are very similar to those in CsAsF6 (1.714 Å; Loss & Röhr, 1998). The neigbouring IF6+ and AsF6− octahedra are mutually tilted, possibly because of packing effects. The geometry of the IF6+ cation in IF6AsF6 is almost identical to that in IF6Sb2F11.
Bond valence analysis of IF6AsF6 gives bond valence sums of 6.972 v.u. (bond valence units) for I and 4.578 v.u. for As (Brese & O'Keeffe, 1991), with contributions of 1.162 v.u. per F1 atom for the former and 0.763 v.u. per F2 atom for the latter.