The structures of tetragonal (
P4
2/
ncm) lithium chlorite, LiClO
2, and orthorhombic (
Cmcm) potassium chlorite, KClO
2, have been determined by single-crystal X-ray analyses. In LiClO
2, the Li atom is at a site of
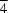
symmetry, while in KClO
2, the K atom is at a site with 2/
m symmetry. In both compounds, the unique Cl and O atoms are at sites with
mm and
m symmetry, respectively. The structure of LiClO
2 consists of layers of Li
+ cations coordinated by ClO
2- anions. In contrast, the structure of KClO
2 contains pseudo-layers of K
+ and ClO
2- ions containing four short K-O distances. The Li
+ and K
+ cations are surrounded by four and eight chlorite O atoms in tetrahedral and distorted cubic coordination environments, respectively.
Supporting information
Lithium and potassium chlorites were synthesized by mixing aqueous solutions of barium chlorite, Ba(ClO2)2, and the corresponding alkali metal sulfates in an equimolar ratio. The reaction mixtures were filtered and the crystals were grown by slow evaporation. In the case of potassium chlorite, the compound decomposes at room temperature in a few hours and its crystal growth was carried out at 273–278 K over approximately 24 h. These conditions yielded crystals in the form of thin plates or needles with a maximum size of 0.5 mm, suitable for X-ray diffraction. The decomposition of lithium chlorite seems to occur more slowly, which allowed crystals to be grown at room temperature. The maximum crystal size was 0.3 mm with a plate morphology. The X-ray powder patterns show good agreement between the bulk products and the single crystals. However, in the case of potassium chlorite, additional peaks in the powder pattern indicate the presence of KCl and KClO3 phases. The precursor used for the preparation of lithium and potassium chlorites [Ba(ClO2)2] was obtained by reacting an aqueous suspension of BaO2 with chlorine dioxide and precipitation from solution by adding a 3:1 mixture of ethanol and diethyl ether. It was found that Ba(ClO2)2 is one of the most stable salts of chlorous acid and is a convenient starting material for preparing other chlorites.
For both compounds, data collection: CD4CA0 (Enraf-Nonius, 1989); cell refinement: CD4CA0; data reduction: CADDAT (Enraf-Nonius, 1989); program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: BS (Ozawa & Kang, 2004); software used to prepare material for publication: SHELXL97.
(I) lithium chlorate(III)
top
Crystal data top
| LiClO2 | Dx = 2.152 Mg m−3 |
| Mr = 74.39 | Mo Kα radiation, λ = 0.71073 Å |
| Tetragonal, P42/ncm | Cell parameters from 20 reflections |
| Hall symbol: -P 4ac 2ac | θ = 12.6–14.8° |
| a = 4.7223 (11) Å | µ = 1.30 mm−1 |
| c = 10.298 (3) Å | T = 293 K |
| V = 229.65 (10) Å3 | Plate, colourless |
| Z = 4 | 0.24 × 0.12 × 0.04 mm |
| F(000) = 144 | |
Data collection top
Enraf-Nonius CAD-4
diffractometer | 87 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.017 |
| Graphite monochromator | θmax = 25.9°, θmin = 4.0° |
| 2θ/θ scans | h = 0→5 |
Absorption correction: empirical (using intensity measurements)
(CADDAT; Enraf-Nonius, 1989) | k = 0→5 |
| Tmin = 0.746, Tmax = 0.950 | l = 0→12 |
| 134 measured reflections | 3 standard reflections every 60 min |
| 133 independent reflections | intensity decay: none |
Refinement top
| Refinement on F2 | 0 restraints |
| Least-squares matrix: full | Primary atom site location: structure-invariant direct methods |
| R[F2 > 2σ(F2)] = 0.047 | Secondary atom site location: difference Fourier map |
| wR(F2) = 0.097 | w = 1/[σ2(Fo2) + (0.0497P)2]
where P = (Fo2 + 2Fc2)/3 |
| S = 0.91 | (Δ/σ)max < 0.001 |
| 133 reflections | Δρmax = 0.35 e Å−3 |
| 13 parameters | Δρmin = −0.33 e Å−3 |
Crystal data top
| LiClO2 | Z = 4 |
| Mr = 74.39 | Mo Kα radiation |
| Tetragonal, P42/ncm | µ = 1.30 mm−1 |
| a = 4.7223 (11) Å | T = 293 K |
| c = 10.298 (3) Å | 0.24 × 0.12 × 0.04 mm |
| V = 229.65 (10) Å3 | |
Data collection top
Enraf-Nonius CAD-4
diffractometer | 87 reflections with I > 2σ(I) |
Absorption correction: empirical (using intensity measurements)
(CADDAT; Enraf-Nonius, 1989) | Rint = 0.017 |
| Tmin = 0.746, Tmax = 0.950 | 3 standard reflections every 60 min |
| 134 measured reflections | intensity decay: none |
| 133 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.047 | 13 parameters |
| wR(F2) = 0.097 | 0 restraints |
| S = 0.91 | Δρmax = 0.35 e Å−3 |
| 133 reflections | Δρmin = −0.33 e Å−3 |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| Li | 0.2500 | 0.7500 | 0.2500 | 0.028 (3) | |
| Cl | 0.7500 | 0.7500 | 0.06960 (18) | 0.0256 (6) | |
| O | 0.5565 (6) | 0.5565 (6) | 0.1575 (4) | 0.0248 (11) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| Li | 0.028 (4) | 0.028 (4) | 0.028 (6) | 0.000 | 0.000 | 0.000 |
| Cl | 0.0256 (8) | 0.0256 (8) | 0.0256 (10) | 0.0001 (18) | 0.000 | 0.000 |
| O | 0.0196 (15) | 0.0196 (15) | 0.035 (2) | 0.0022 (18) | 0.0049 (15) | 0.0049 (15) |
Geometric parameters (Å, º) top
| Li—Oi | 1.959 (2) | Cl—Oiv | 1.578 (4) |
| Li—Oii | 1.959 (2) | Cl—O | 1.578 (4) |
| Li—O | 1.959 (2) | O—Liv | 1.959 (2) |
| Li—Oiii | 1.959 (2) | | |
| | | |
| Oi—Li—Oii | 103.67 (8) | Oii—Li—Oiii | 103.67 (8) |
| Oi—Li—O | 103.67 (8) | O—Li—Oiii | 103.67 (8) |
| O—Li—Oii | 121.82 (19) | O—Cl—Oiv | 110.0 (3) |
| Oi—Li—Oiii | 121.82 (19) | | |
| Symmetry codes: (i) −y+1, x+1/2, −z+1/2; (ii) −x+1/2, −y+3/2, z; (iii) y−1/2, −x+1, −z+1/2; (iv) −x+3/2, −y+3/2, z; (v) x+1/2, −y+1, −z+1/2. |
(II) potassium chlorate(III)
top
Crystal data top
| KClO2 | F(000) = 208 |
| Mr = 106.55 | Dx = 2.448 Mg m−3 |
| Orthorhombic, Cmcm | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -C 2c 2 | Cell parameters from 22 reflections |
| a = 6.1446 (9) Å | θ = 11.1–11.9° |
| b = 6.3798 (12) Å | µ = 2.48 mm−1 |
| c = 7.3755 (19) Å | T = 293 K |
| V = 289.13 (10) Å3 | Plate, colourless |
| Z = 4 | 0.40 × 0.24 × 0.16 mm |
Data collection top
Enraf-Nonius CAD-4
diffractometer | 168 reflections with I > 2σ(I) |
| Radiation source: fine-focus sealed tube | Rint = 0.059 |
| Graphite monochromator | θmax = 27.5°, θmin = 5.4° |
| 2θ/θ scans | h = 0→7 |
Absorption correction: empirical (using intensity measurements)
(CADDAT; Enraf-Nonius, 1989) | k = −1→8 |
| Tmin = 0.494, Tmax = 0.672 | l = −1→9 |
| 243 measured reflections | 3 standard reflections every 60 min |
| 193 independent reflections | intensity decay: none |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.030 | w = 1/[σ2(Fo2) + (0.0464P)2]
where P = (Fo2 + 2Fc2)/3 |
| wR(F2) = 0.074 | (Δ/σ)max < 0.001 |
| S = 1.01 | Δρmax = 0.32 e Å−3 |
| 193 reflections | Δρmin = −1.03 e Å−3 |
| 16 parameters | Extinction correction: SHELXL97 (Sheldrick, 1997), Fc*=kFc[1+0.001xFc2λ3/sin(2θ)]-1/4 |
| 0 restraints | Extinction coefficient: 0.025 (7) |
Crystal data top
| KClO2 | V = 289.13 (10) Å3 |
| Mr = 106.55 | Z = 4 |
| Orthorhombic, Cmcm | Mo Kα radiation |
| a = 6.1446 (9) Å | µ = 2.48 mm−1 |
| b = 6.3798 (12) Å | T = 293 K |
| c = 7.3755 (19) Å | 0.40 × 0.24 × 0.16 mm |
Data collection top
Enraf-Nonius CAD-4
diffractometer | 168 reflections with I > 2σ(I) |
Absorption correction: empirical (using intensity measurements)
(CADDAT; Enraf-Nonius, 1989) | Rint = 0.059 |
| Tmin = 0.494, Tmax = 0.672 | 3 standard reflections every 60 min |
| 243 measured reflections | intensity decay: none |
| 193 independent reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.030 | 16 parameters |
| wR(F2) = 0.074 | 0 restraints |
| S = 1.01 | Δρmax = 0.32 e Å−3 |
| 193 reflections | Δρmin = −1.03 e Å−3 |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| K | 0.0000 | 0.0000 | 0.0000 | 0.0329 (4) | |
| Cl | 0.5000 | −0.03139 (16) | 0.2500 | 0.0261 (4) | |
| O | 0.2937 (4) | −0.1753 (4) | 0.2500 | 0.0401 (8) | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| K | 0.0328 (6) | 0.0358 (6) | 0.0303 (6) | 0.000 | 0.000 | −0.0003 (4) |
| Cl | 0.0226 (6) | 0.0205 (6) | 0.0352 (7) | 0.000 | 0.000 | 0.000 |
| O | 0.0263 (11) | 0.0290 (12) | 0.0651 (17) | −0.0063 (10) | 0.000 | 0.000 |
Geometric parameters (Å, º) top
| K—Oi | 2.8120 (18) | K—Ovii | 3.0493 (19) |
| K—O | 2.8120 (18) | Cl—O | 1.565 (2) |
| K—Oii | 2.8120 (18) | Cl—Oviii | 1.565 (2) |
| K—Oiii | 2.8120 (18) | O—Kix | 2.8120 (18) |
| K—Oiv | 3.0493 (19) | O—Kx | 3.0493 (19) |
| K—Ov | 3.0493 (19) | O—Kxi | 3.0493 (19) |
| K—Ovi | 3.0493 (19) | | |
| | | |
| O—K—Oi | 180.00 (9) | Oi—K—Ovi | 81.93 (2) |
| Oi—K—Oii | 79.86 (7) | O—K—Ovi | 98.07 (2) |
| O—K—Oii | 100.14 (7) | Oii—K—Ovi | 113.15 (2) |
| Oi—K—Oiii | 100.14 (7) | Oiii—K—Ovi | 66.85 (2) |
| O—K—Oiii | 79.86 (7) | Oiv—K—Ovi | 49.12 (8) |
| Oii—K—Oiii | 180.0 | Ov—K—Ovi | 130.88 (8) |
| Oi—K—Oiv | 113.15 (2) | Oi—K—Ovii | 98.07 (2) |
| O—K—Oiv | 66.85 (2) | O—K—Ovii | 81.93 (2) |
| Oii—K—Oiv | 81.93 (2) | Oii—K—Ovii | 66.85 (2) |
| Oiii—K—Oiv | 98.07 (2) | Oiii—K—Ovii | 113.15 (2) |
| Oi—K—Ov | 66.85 (2) | Oiv—K—Ovii | 130.88 (8) |
| O—K—Ov | 113.15 (2) | Ov—K—Ovii | 49.12 (8) |
| Oii—K—Ov | 98.07 (2) | Ovi—K—Ovii | 180.00 (10) |
| Oiii—K—Ov | 81.93 (2) | O—Cl—Oviii | 108.18 (18) |
| Oiv—K—Ov | 180.0 | | |
| Symmetry codes: (i) −x, −y, −z; (ii) x, −y, −z; (iii) −x, y, z; (iv) −x+1/2, y+1/2, z; (v) x−1/2, −y−1/2, −z; (vi) x−1/2, y+1/2, z; (vii) −x+1/2, −y−1/2, −z; (viii) −x+1, y, z; (ix) −x, −y, z+1/2; (x) x+1/2, y−1/2, z; (xi) −x+1/2, −y−1/2, z+1/2. |
Experimental details
| | (I) | (II) |
| Crystal data |
| Chemical formula | LiClO2 | KClO2 |
| Mr | 74.39 | 106.55 |
| Crystal system, space group | Tetragonal, P42/ncm | Orthorhombic, Cmcm |
| Temperature (K) | 293 | 293 |
| a, b, c (Å) | 4.7223 (11), 4.7223 (11), 10.298 (3) | 6.1446 (9), 6.3798 (12), 7.3755 (19) |
| α, β, γ (°) | 90, 90, 90 | 90, 90, 90 |
| V (Å3) | 229.65 (10) | 289.13 (10) |
| Z | 4 | 4 |
| Radiation type | Mo Kα | Mo Kα |
| µ (mm−1) | 1.30 | 2.48 |
| Crystal size (mm) | 0.24 × 0.12 × 0.04 | 0.40 × 0.24 × 0.16 |
| |
| Data collection |
| Diffractometer | Enraf-Nonius CAD-4
diffractometer | Enraf-Nonius CAD-4
diffractometer |
| Absorption correction | Empirical (using intensity measurements)
(CADDAT; Enraf-Nonius, 1989) | Empirical (using intensity measurements)
(CADDAT; Enraf-Nonius, 1989) |
| Tmin, Tmax | 0.746, 0.950 | 0.494, 0.672 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 134, 133, 87 | 243, 193, 168 |
| Rint | 0.017 | 0.059 |
| (sin θ/λ)max (Å−1) | 0.613 | 0.649 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.047, 0.097, 0.91 | 0.030, 0.074, 1.01 |
| No. of reflections | 133 | 193 |
| No. of parameters | 13 | 16 |
| Δρmax, Δρmin (e Å−3) | 0.35, −0.33 | 0.32, −1.03 |
Selected geometric parameters (Å, º) for (I) top| Li—O | 1.959 (2) | Cl—O | 1.578 (4) |
| | | |
| O—Li—Oi | 121.82 (19) | O—Cl—Oiii | 110.0 (3) |
| O—Li—Oii | 103.67 (8) | | |
| Symmetry codes: (i) −x+1/2, −y+3/2, z; (ii) y−1/2, −x+1, −z+1/2; (iii) −x+3/2, −y+3/2, z. |
Selected geometric parameters (Å, º) for (II) top| K—O | 2.8120 (18) | Cl—O | 1.565 (2) |
| K—Oi | 3.0493 (19) | | |
| | | |
| O—K—Oii | 180.00 (9) | O—K—Oi | 98.07 (2) |
| O—K—Oiii | 100.14 (7) | Ov—K—Oi | 49.12 (8) |
| O—K—Oiv | 79.86 (7) | O—K—Ovii | 81.93 (2) |
| O—K—Ov | 66.85 (2) | O—Cl—Oviii | 108.18 (18) |
| O—K—Ovi | 113.15 (2) | | |
| Symmetry codes: (i) x−1/2, y+1/2, z; (ii) −x, −y, −z; (iii) x, −y, −z; (iv) −x, y, z; (v) −x+1/2, y+1/2, z; (vi) x−1/2, −y−1/2, −z; (vii) −x+1/2, −y−1/2, −z; (viii) −x+1, y, z. |
![[Figure 1]](http://journals.iucr.org/c/issues/2005/02/00/bc1064/bc1064fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2005/02/00/bc1064/bc1064fig2thm.gif)
![[Figure 3]](http://journals.iucr.org/c/issues/2005/02/00/bc1064/bc1064fig3thm.gif)
![[Figure 4]](http://journals.iucr.org/c/issues/2005/02/00/bc1064/bc1064fig4thm.gif)

 journal menu
journal menu












Previous crystal structure investigations of chlorites include NH4ClO2 (Levi & Scherillo, 1931; Gillespie et al., 1959), NaClO2·3H2O (Tarimci & Schempp, 1975), NaClO2 (Tarimci et al., 1976), Zn(ClO2)2·2H2O (Pakkanen, 1979), Mg(ClO2)2·6H2O (Ferrari & Colla, 1937; Okuda et al., 1990; Marsh, 1991), AgClO2 (Curti et al., 1957; Cooper & Marsh, 1961; Okuda et al., 1990), Pb(ClO2)2 (Okuda et al., 1990) and La(ClO2)3·3H2O (Coda et al, 1965; Castellani Bisi, 1984). It is apparent that alkali metal chlorites have not been investigated systematically. The limited number of studies is probably due to the difficulty of preparation and investigation of these compounds, resulting from their low stability. A majority of chlorites quickly decompose to the corresponding chlorates and chlorides under the effect of temperature, sunlight or X-radiation. Besides this, the crystallization of chlorites is often difficult and additional research is required to determine appropriate conditions for single-crystal growth. This paper reports the results of our study of two alkali chlorites, namely LiClO2 and KClO2.
The structure of lithium chlorite contains separate layers, within which the Li+ cation is surrounded by four chlorite O atoms forming a tetrahedron which is squashed along a twofold axis (Fig. 1). These layers are parallel to the ab plane and are linked by van der Waals interactions, with the shortest interlayer Cl···Cl distance being 3.6339 (9) Å (Fig. 2). The layers are stacked in such way that adjacent layers are rotated by 90°. The ClO2- anions serve as tetradentate bridging ligands between the Li+ cations.
The structure of potassium chlorite contains pseudo-layers parallel to the ac plane and consisting of K+ and ClO2− ions with short K—O distances (Fig. 3). The K+ cations and Cl atoms are nearly coplanar (Fig. 4). The full coordination environment of the K+ cation involves eight O atoms forming a distorted cube. Four of these O atoms belong to one layer and the other four to the layers above and below.
The structures of LiClO2 and KClO2 show some similarities to those of the corresponding hypophosphites. In the case of LiClO2, the role of ClO2− as a tetradentate ligand is identical to that of H2PO2− in LiH2PO2 (Naumov et al., 2004). However, the structures of the layers are different in these compounds: by sharing edges, the Li+-centred tetrahedra form chains linked in different ways within a layer. In KClO2, the layers and the immediate environment of the K+ cations are very similar to those in the K, Rb and Cs hypophospites (Naumova et al., 2004). The differences arise from the way the layers are joined together and from the K+ environment, which includes two H atoms in the hypophospites instead of two O atoms in KClO2.