In the title centrosymmetric dimer, [Pb
2(sbc)
2(phen)
2]·2H
2O [sbc is the 2-sulfonatobenzoate dianion (C
7H
4O
5S) and phen is 1,10-phenanthroline (C
12H
8N
2)], each Pb
II ion is six-coordinated by four O atoms,
viz. carboxylate and sulfonate O atoms from two sbc anions, and two N atoms from a 1,10-phenanthroline ligand. One 1,10-phenanthroline ligand and the carboxylate group of one sbc ligand are chelated to each Pb
II cation, and the sulfonate group of the other sbc unit is monodentate. One O atom of the chelated carboxylate group also bridges to the other Pb
II cation, so that each pair of Pb
II ions is bridged by two sbc anions and has the same coordination environment, forming a dinuclear ring. Each pair of Pb
II ions is thus connected by two different kinds of bridges, namely a carboxylate short bridge and a carboxylate–sulfonate long bridge. There is also a special position of
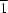
site symmetry at the centre of the two Pb
II cations.
Supporting information
CCDC reference: 251284
The title compound was synthesized by adding a solution of 1,10-phenanthroline (0.04 g, 0.2 mmol), 2-sulfobenzenecarboxylic acid (0.04 g, 0.2 mmol) and 2,2'-dithiosalicylic acid (0.06 g, 0.2 mmol) in dimethylformamide (10 ml) dropwise to a stirred solution of lead nitrate (0.07 g, 0.2 mmol) in water (10 ml) at 298 K. The reaction mixture was filtered and the filtrate allowed to stand for about 6 weeks until colourless prism crystals of (I) were obtained. Crystals suitable for X-ray diffraction were collected by filtration, washed with water and ethanol, and dried in air.
The water H atoms were refined subject to the restraint O—H = 0.82 (2) Å. The remaining H atoms were positioned geometrically and allowed to ride on their parent atoms, with C—H distances of 0.93 Å and with Uiso(H) = 1.2Ueq(C).
Data collection: SMART (Bruker, 2002); cell refinement: SAINT (Bruker, 2002); data reduction: SAINT; program(s) used to solve structure: SHELXS97 (Sheldrick, 1997); program(s) used to refine structure: SHELXL97 (Sheldrick, 1997); molecular graphics: XP in SHELXTL (Bruker, 2002); software used to prepare material for publication: SHELXL97.
Bis(µ-2-sulfobenzenecarboxylato)bis[(1,10-phenanthroline)lead(II)] dihydrate
top
Crystal data top
| [Pb2(C7H4O5S)2(C12H8N2)2]·2H2O | Z = 2 |
| Mr = 605.57 | F(000) = 576 |
| Triclinic, P1 | Dx = 2.151 Mg m−3 |
| Hall symbol: -P1 | Mo Kα radiation, λ = 0.71073 Å |
| a = 7.3232 (7) Å | Cell parameters from 3352 reflections |
| b = 10.8613 (10) Å | θ = 2.3–25.3° |
| c = 12.0558 (11) Å | µ = 9.18 mm−1 |
| α = 97.280 (1)° | T = 298 K |
| β = 100.320 (2)° | Prism, colourless |
| γ = 91.150 (1)° | 0.20 × 0.15 × 0.10 mm |
| V = 934.89 (15) Å3 | |
Data collection top
Bruker APEX CCD area-detector
diffractometer | 3352 independent reflections |
| Radiation source: fine-focus sealed tube | 3162 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.024 |
| ϕ and ω scans | θmax = 25.3°, θmin = 1.9° |
Absorption correction: numerical
(SADABS; Bruker, 2002) | h = −8→8 |
| Tmin = 0.034, Tmax = 0.227 | k = −13→12 |
| 6867 measured reflections | l = −14→14 |
Refinement top
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.025 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.062 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.05 | w = 1/[σ2(Fo2) + (0.0341P)2 + 0.2266P]
where P = (Fo2 + 2Fc2)/3 |
| 3352 reflections | (Δ/σ)max = 0.001 |
| 270 parameters | Δρmax = 1.75 e Å−3 |
| 0 restraints | Δρmin = −0.60 e Å−3 |
Crystal data top
| [Pb2(C7H4O5S)2(C12H8N2)2]·2H2O | γ = 91.150 (1)° |
| Mr = 605.57 | V = 934.89 (15) Å3 |
| Triclinic, P1 | Z = 2 |
| a = 7.3232 (7) Å | Mo Kα radiation |
| b = 10.8613 (10) Å | µ = 9.18 mm−1 |
| c = 12.0558 (11) Å | T = 298 K |
| α = 97.280 (1)° | 0.20 × 0.15 × 0.10 mm |
| β = 100.320 (2)° | |
Data collection top
Bruker APEX CCD area-detector
diffractometer | 3352 independent reflections |
Absorption correction: numerical
(SADABS; Bruker, 2002) | 3162 reflections with I > 2σ(I) |
| Tmin = 0.034, Tmax = 0.227 | Rint = 0.024 |
| 6867 measured reflections | |
Refinement top
| R[F2 > 2σ(F2)] = 0.025 | 0 restraints |
| wR(F2) = 0.062 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.05 | Δρmax = 1.75 e Å−3 |
| 3352 reflections | Δρmin = −0.60 e Å−3 |
| 270 parameters | |
Special details top
Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2) top| | x | y | z | Uiso*/Ueq | |
| O6 | 0.8489 (9) | 0.2030 (7) | 0.0765 (5) | 0.0880 (18) | |
| Pb1 | 0.79333 (2) | 0.605327 (14) | 0.565670 (14) | 0.02970 (8) | |
| S1 | 0.48519 (17) | 0.59828 (12) | 0.31857 (11) | 0.0378 (3) | |
| O1 | 0.9269 (5) | 0.5524 (3) | 0.3827 (3) | 0.0406 (8) | |
| O3 | 0.5714 (5) | 0.7016 (3) | 0.4036 (3) | 0.0421 (8) | |
| O4 | 0.5278 (5) | 0.4785 (3) | 0.3587 (3) | 0.0460 (9) | |
| O5 | 0.2910 (6) | 0.6107 (5) | 0.2809 (4) | 0.0726 (13) | |
| N1 | 1.0069 (5) | 0.7936 (3) | 0.5696 (3) | 0.0329 (9) | |
| N2 | 0.7212 (5) | 0.8020 (3) | 0.6897 (3) | 0.0342 (9) | |
| C1 | 1.1442 (7) | 0.7912 (5) | 0.5113 (5) | 0.0443 (13) | |
| H1 | 1.1553 | 0.7198 | 0.4620 | 0.053* | |
| C2 | 1.2725 (8) | 0.8897 (5) | 0.5200 (5) | 0.0497 (14) | |
| H2 | 1.3679 | 0.8834 | 0.4783 | 0.060* | |
| C3 | 1.2562 (8) | 0.9946 (5) | 0.5900 (6) | 0.0510 (14) | |
| H3 | 1.3391 | 1.0621 | 0.5958 | 0.061* | |
| C4 | 1.1125 (7) | 1.0015 (5) | 0.6545 (5) | 0.0422 (12) | |
| C5 | 1.0891 (9) | 1.1089 (5) | 0.7302 (6) | 0.0633 (18) | |
| H5 | 1.1720 | 1.1770 | 0.7394 | 0.076* | |
| C6 | 0.9492 (10) | 1.1136 (5) | 0.7885 (6) | 0.0615 (17) | |
| H6 | 0.9360 | 1.1855 | 0.8367 | 0.074* | |
| C7 | 0.8204 (8) | 1.0110 (5) | 0.7784 (5) | 0.0462 (13) | |
| C8 | 0.6732 (9) | 1.0109 (5) | 0.8386 (5) | 0.0552 (15) | |
| H8 | 0.6566 | 1.0801 | 0.8890 | 0.066* | |
| C9 | 0.5534 (9) | 0.9084 (5) | 0.8229 (5) | 0.0516 (14) | |
| H9 | 0.4550 | 0.9072 | 0.8620 | 0.062* | |
| C10 | 0.5833 (8) | 0.8068 (5) | 0.7471 (5) | 0.0435 (12) | |
| H10 | 0.5013 | 0.7380 | 0.7360 | 0.052* | |
| C11 | 0.9914 (7) | 0.8976 (4) | 0.6410 (4) | 0.0352 (11) | |
| C12 | 0.8402 (7) | 0.9032 (4) | 0.7041 (4) | 0.0335 (10) | |
| C13 | 0.8885 (6) | 0.4964 (5) | 0.2831 (4) | 0.0339 (11) | |
| C14 | 0.7694 (7) | 0.5571 (4) | 0.1918 (4) | 0.0326 (10) | |
| C15 | 0.5935 (7) | 0.6029 (4) | 0.1977 (4) | 0.0321 (10) | |
| C16 | 0.4948 (7) | 0.6532 (5) | 0.1064 (5) | 0.0425 (12) | |
| H16 | 0.3752 | 0.6791 | 0.1090 | 0.051* | |
| C17 | 0.5708 (8) | 0.6652 (5) | 0.0125 (5) | 0.0487 (14) | |
| H17 | 0.5047 | 0.7019 | −0.0470 | 0.058* | |
| C18 | 0.7449 (9) | 0.6230 (6) | 0.0059 (5) | 0.0501 (14) | |
| H18 | 0.7962 | 0.6312 | −0.0581 | 0.060* | |
| C19 | 0.8436 (8) | 0.5685 (5) | 0.0942 (5) | 0.0418 (12) | |
| H19 | 0.9605 | 0.5391 | 0.0887 | 0.050* | |
| O2 | 0.9549 (5) | 0.3944 (3) | 0.2529 (3) | 0.0468 (9) | |
| H6A | 0.885 (15) | 0.276 (11) | 0.123 (10) | 0.14 (4)* | |
| H6B | 0.89 (2) | 0.136 (14) | 0.107 (13) | 0.24 (8)* | |
Atomic displacement parameters (Å2) top| | U11 | U22 | U33 | U12 | U13 | U23 |
| O6 | 0.099 (4) | 0.079 (4) | 0.066 (3) | 0.006 (3) | −0.015 (3) | −0.022 (3) |
| Pb1 | 0.03257 (11) | 0.02643 (11) | 0.02964 (12) | −0.00129 (7) | 0.00979 (8) | −0.00342 (7) |
| S1 | 0.0344 (6) | 0.0425 (7) | 0.0372 (7) | −0.0036 (5) | 0.0131 (5) | −0.0004 (6) |
| O1 | 0.0406 (19) | 0.051 (2) | 0.0289 (19) | 0.0055 (16) | 0.0103 (15) | −0.0053 (16) |
| O3 | 0.054 (2) | 0.0365 (19) | 0.034 (2) | 0.0015 (17) | 0.0135 (17) | −0.0068 (16) |
| O4 | 0.060 (2) | 0.0359 (19) | 0.045 (2) | −0.0084 (17) | 0.0216 (19) | 0.0020 (17) |
| O5 | 0.042 (2) | 0.111 (4) | 0.067 (3) | 0.004 (2) | 0.017 (2) | 0.009 (3) |
| N1 | 0.038 (2) | 0.029 (2) | 0.032 (2) | −0.0002 (17) | 0.0090 (18) | −0.0005 (17) |
| N2 | 0.039 (2) | 0.030 (2) | 0.034 (2) | 0.0045 (17) | 0.0108 (18) | −0.0012 (17) |
| C1 | 0.044 (3) | 0.038 (3) | 0.053 (4) | 0.002 (2) | 0.019 (3) | 0.001 (3) |
| C2 | 0.043 (3) | 0.050 (3) | 0.059 (4) | −0.003 (3) | 0.020 (3) | 0.004 (3) |
| C3 | 0.040 (3) | 0.043 (3) | 0.068 (4) | −0.009 (2) | 0.007 (3) | 0.008 (3) |
| C4 | 0.044 (3) | 0.031 (3) | 0.049 (3) | −0.005 (2) | 0.007 (2) | 0.000 (2) |
| C5 | 0.063 (4) | 0.035 (3) | 0.086 (5) | −0.017 (3) | 0.014 (4) | −0.013 (3) |
| C6 | 0.072 (4) | 0.035 (3) | 0.073 (5) | −0.003 (3) | 0.016 (4) | −0.017 (3) |
| C7 | 0.059 (3) | 0.035 (3) | 0.042 (3) | 0.001 (2) | 0.009 (3) | −0.005 (2) |
| C8 | 0.075 (4) | 0.042 (3) | 0.050 (4) | 0.006 (3) | 0.026 (3) | −0.014 (3) |
| C9 | 0.064 (4) | 0.051 (3) | 0.046 (3) | 0.012 (3) | 0.031 (3) | 0.001 (3) |
| C10 | 0.048 (3) | 0.041 (3) | 0.043 (3) | 0.000 (2) | 0.017 (3) | 0.002 (2) |
| C11 | 0.040 (3) | 0.031 (2) | 0.033 (3) | −0.002 (2) | 0.007 (2) | −0.001 (2) |
| C12 | 0.044 (3) | 0.026 (2) | 0.029 (3) | 0.001 (2) | 0.006 (2) | −0.0017 (19) |
| C13 | 0.033 (2) | 0.040 (3) | 0.028 (3) | −0.002 (2) | 0.008 (2) | 0.000 (2) |
| C14 | 0.039 (3) | 0.028 (2) | 0.029 (3) | −0.001 (2) | 0.006 (2) | −0.0043 (19) |
| C15 | 0.036 (2) | 0.029 (2) | 0.031 (3) | −0.0027 (19) | 0.009 (2) | −0.002 (2) |
| C16 | 0.034 (3) | 0.046 (3) | 0.046 (3) | 0.001 (2) | 0.004 (2) | 0.006 (3) |
| C17 | 0.061 (4) | 0.049 (3) | 0.037 (3) | 0.006 (3) | 0.003 (3) | 0.017 (3) |
| C18 | 0.064 (4) | 0.055 (3) | 0.036 (3) | 0.005 (3) | 0.018 (3) | 0.010 (3) |
| C19 | 0.045 (3) | 0.046 (3) | 0.036 (3) | 0.004 (2) | 0.012 (2) | 0.003 (2) |
| O2 | 0.061 (2) | 0.0337 (19) | 0.041 (2) | 0.0103 (17) | 0.0004 (18) | −0.0045 (16) |
Geometric parameters (Å, º) top
| O6—H6A | 0.92 (12) | C5—C6 | 1.341 (9) |
| O6—H6B | 0.88 (14) | C5—H5 | 0.9300 |
| Pb1—N1 | 2.540 (4) | C6—C7 | 1.424 (8) |
| Pb1—N2 | 2.569 (4) | C6—H6 | 0.9300 |
| Pb1—O1 | 2.574 (3) | C7—C8 | 1.403 (8) |
| Pb1—O2i | 2.595 (4) | C7—C12 | 1.408 (7) |
| Pb1—O3 | 2.646 (4) | C8—C9 | 1.376 (8) |
| Pb1—O1i | 2.734 (3) | C8—H8 | 0.9300 |
| S1—O5 | 1.428 (4) | C9—C10 | 1.388 (8) |
| S1—O4 | 1.465 (4) | C9—H9 | 0.9300 |
| S1—O3 | 1.469 (4) | C10—H10 | 0.9300 |
| S1—C15 | 1.785 (5) | C11—C12 | 1.450 (7) |
| O1—C13 | 1.256 (6) | C13—O2 | 1.256 (6) |
| O1—Pb1i | 2.734 (3) | C13—C14 | 1.507 (7) |
| N1—C1 | 1.324 (6) | C14—C19 | 1.400 (7) |
| N1—C11 | 1.350 (6) | C14—C15 | 1.400 (7) |
| N2—C10 | 1.321 (6) | C15—C16 | 1.384 (7) |
| N2—C12 | 1.363 (6) | C16—C17 | 1.367 (8) |
| C1—C2 | 1.391 (8) | C16—H16 | 0.9300 |
| C1—H1 | 0.9300 | C17—C18 | 1.376 (8) |
| C2—C3 | 1.351 (8) | C17—H17 | 0.9300 |
| C2—H2 | 0.9300 | C18—C19 | 1.380 (9) |
| C3—C4 | 1.413 (8) | C18—H18 | 0.9300 |
| C3—H3 | 0.9300 | C19—H19 | 0.9300 |
| C4—C11 | 1.396 (7) | O2—Pb1i | 2.595 (4) |
| C4—C5 | 1.421 (8) | | |
| | | |
| H6A—O6—H6B | 114 (10) | C6—C5—H5 | 119.5 |
| N1—Pb1—N2 | 65.03 (12) | C4—C5—H5 | 119.5 |
| N1—Pb1—O1 | 77.52 (12) | C5—C6—C7 | 121.5 (6) |
| N2—Pb1—O1 | 137.24 (12) | C5—C6—H6 | 119.2 |
| N1—Pb1—O2i | 74.86 (12) | C7—C6—H6 | 119.2 |
| N2—Pb1—O2i | 78.37 (12) | C8—C7—C12 | 117.6 (5) |
| O1—Pb1—O2i | 111.94 (12) | C8—C7—C6 | 123.6 (5) |
| N1—Pb1—O3 | 84.87 (12) | C12—C7—C6 | 118.8 (5) |
| N2—Pb1—O3 | 82.95 (11) | C9—C8—C7 | 120.0 (5) |
| O1—Pb1—O3 | 73.54 (11) | C9—C8—H8 | 120.0 |
| O2i—Pb1—O3 | 156.80 (12) | C7—C8—H8 | 120.0 |
| N1—Pb1—O1i | 95.32 (12) | C8—C9—C10 | 118.2 (5) |
| N2—Pb1—O1i | 127.26 (11) | C8—C9—H9 | 120.9 |
| O1—Pb1—O1i | 74.01 (12) | C10—C9—H9 | 120.9 |
| O2i—Pb1—O1i | 48.91 (11) | N2—C10—C9 | 123.9 (5) |
| O3—Pb1—O1i | 146.69 (10) | N2—C10—H10 | 118.0 |
| O5—S1—O4 | 112.8 (3) | C9—C10—H10 | 118.0 |
| O5—S1—O3 | 113.1 (3) | N1—C11—C4 | 122.6 (5) |
| O4—S1—O3 | 110.9 (2) | N1—C11—C12 | 118.9 (4) |
| O5—S1—C15 | 106.9 (3) | C4—C11—C12 | 118.4 (4) |
| O4—S1—C15 | 106.3 (2) | N2—C12—C7 | 121.9 (5) |
| O3—S1—C15 | 106.3 (2) | N2—C12—C11 | 118.2 (4) |
| C13—O1—Pb1 | 142.7 (3) | C7—C12—C11 | 119.9 (4) |
| C13—O1—Pb1i | 90.1 (3) | O1—C13—O2 | 123.2 (5) |
| Pb1—O1—Pb1i | 105.99 (12) | O1—C13—C14 | 119.4 (4) |
| S1—O3—Pb1 | 107.52 (18) | O2—C13—C14 | 117.2 (4) |
| C1—N1—C11 | 117.9 (4) | C19—C14—C15 | 118.3 (5) |
| C1—N1—Pb1 | 122.7 (3) | C19—C14—C13 | 116.4 (4) |
| C11—N1—Pb1 | 119.2 (3) | C15—C14—C13 | 125.3 (4) |
| C10—N2—C12 | 118.4 (4) | C16—C15—C14 | 119.9 (5) |
| C10—N2—Pb1 | 123.3 (3) | C16—C15—S1 | 117.1 (4) |
| C12—N2—Pb1 | 118.1 (3) | C14—C15—S1 | 123.0 (4) |
| N1—C1—C2 | 123.5 (5) | C17—C16—C15 | 120.9 (5) |
| N1—C1—H1 | 118.2 | C17—C16—H16 | 119.6 |
| C2—C1—H1 | 118.2 | C15—C16—H16 | 119.6 |
| C3—C2—C1 | 118.9 (5) | C16—C17—C18 | 120.1 (5) |
| C3—C2—H2 | 120.6 | C16—C17—H17 | 120.0 |
| C1—C2—H2 | 120.6 | C18—C17—H17 | 120.0 |
| C2—C3—C4 | 119.7 (5) | C17—C18—C19 | 120.1 (5) |
| C2—C3—H3 | 120.1 | C17—C18—H18 | 119.9 |
| C4—C3—H3 | 120.1 | C19—C18—H18 | 119.9 |
| C11—C4—C3 | 117.3 (5) | C18—C19—C14 | 120.6 (5) |
| C11—C4—C5 | 120.3 (5) | C18—C19—H19 | 119.7 |
| C3—C4—C5 | 122.3 (5) | C14—C19—H19 | 119.7 |
| C6—C5—C4 | 121.0 (5) | C13—O2—Pb1i | 96.6 (3) |
| | | |
| N1—Pb1—O1—C13 | −148.4 (6) | C7—C8—C9—C10 | 0.2 (9) |
| N2—Pb1—O1—C13 | −119.6 (5) | C12—N2—C10—C9 | −1.0 (8) |
| O2i—Pb1—O1—C13 | 143.8 (5) | Pb1—N2—C10—C9 | 173.5 (4) |
| O3—Pb1—O1—C13 | −60.1 (5) | C8—C9—C10—N2 | 0.7 (9) |
| O1i—Pb1—O1—C13 | 112.3 (6) | C1—N1—C11—C4 | 0.5 (7) |
| N1—Pb1—O1—Pb1i | 99.33 (14) | Pb1—N1—C11—C4 | 175.0 (4) |
| N2—Pb1—O1—Pb1i | 128.15 (15) | C1—N1—C11—C12 | 178.9 (5) |
| O2i—Pb1—O1—Pb1i | 31.51 (16) | Pb1—N1—C11—C12 | −6.6 (6) |
| O3—Pb1—O1—Pb1i | −172.39 (15) | C3—C4—C11—N1 | −0.2 (8) |
| O1i—Pb1—O1—Pb1i | 0.0 | C5—C4—C11—N1 | 179.5 (5) |
| O5—S1—O3—Pb1 | 136.2 (2) | C3—C4—C11—C12 | −178.6 (5) |
| O4—S1—O3—Pb1 | 8.3 (2) | C5—C4—C11—C12 | 1.1 (8) |
| C15—S1—O3—Pb1 | −106.9 (2) | C10—N2—C12—C7 | 0.5 (7) |
| N1—Pb1—O3—S1 | 142.3 (2) | Pb1—N2—C12—C7 | −174.4 (4) |
| N2—Pb1—O3—S1 | −152.2 (2) | C10—N2—C12—C11 | −180.0 (4) |
| O1—Pb1—O3—S1 | 63.87 (19) | Pb1—N2—C12—C11 | 5.2 (6) |
| O2i—Pb1—O3—S1 | 171.3 (2) | C8—C7—C12—N2 | 0.4 (8) |
| O1i—Pb1—O3—S1 | 50.5 (3) | C6—C7—C12—N2 | −179.5 (5) |
| N2—Pb1—N1—C1 | −179.4 (4) | C8—C7—C12—C11 | −179.2 (5) |
| O1—Pb1—N1—C1 | −20.6 (4) | C6—C7—C12—C11 | 0.9 (8) |
| O2i—Pb1—N1—C1 | 96.6 (4) | N1—C11—C12—N2 | 0.9 (7) |
| O3—Pb1—N1—C1 | −94.8 (4) | C4—C11—C12—N2 | 179.4 (5) |
| O1i—Pb1—N1—C1 | 51.8 (4) | N1—C11—C12—C7 | −179.6 (5) |
| N2—Pb1—N1—C11 | 6.4 (3) | C4—C11—C12—C7 | −1.1 (7) |
| O1—Pb1—N1—C11 | 165.2 (4) | Pb1—O1—C13—O2 | −106.1 (6) |
| O2i—Pb1—N1—C11 | −77.6 (3) | Pb1i—O1—C13—O2 | 11.1 (5) |
| O3—Pb1—N1—C11 | 91.0 (3) | Pb1—O1—C13—C14 | 79.0 (7) |
| O1i—Pb1—N1—C11 | −122.4 (3) | Pb1i—O1—C13—C14 | −163.8 (4) |
| N1—Pb1—N2—C10 | 179.6 (4) | O1—C13—C14—C19 | 125.3 (5) |
| O1—Pb1—N2—C10 | 148.3 (4) | O2—C13—C14—C19 | −49.9 (6) |
| O2i—Pb1—N2—C10 | −101.9 (4) | O1—C13—C14—C15 | −54.2 (7) |
| O3—Pb1—N2—C10 | 92.0 (4) | O2—C13—C14—C15 | 130.6 (5) |
| O1i—Pb1—N2—C10 | −103.5 (4) | C19—C14—C15—C16 | 2.6 (7) |
| N1—Pb1—N2—C12 | −5.9 (3) | C13—C14—C15—C16 | −177.8 (5) |
| O1—Pb1—N2—C12 | −37.1 (4) | C19—C14—C15—S1 | −178.3 (4) |
| O2i—Pb1—N2—C12 | 72.7 (3) | C13—C14—C15—S1 | 1.2 (7) |
| O3—Pb1—N2—C12 | −93.5 (3) | O5—S1—C15—C16 | 19.0 (5) |
| O1i—Pb1—N2—C12 | 71.1 (4) | O4—S1—C15—C16 | 139.8 (4) |
| C11—N1—C1—C2 | 0.1 (8) | O3—S1—C15—C16 | −102.0 (4) |
| Pb1—N1—C1—C2 | −174.2 (4) | O5—S1—C15—C14 | −160.1 (4) |
| N1—C1—C2—C3 | −1.0 (9) | O4—S1—C15—C14 | −39.4 (5) |
| C1—C2—C3—C4 | 1.3 (9) | O3—S1—C15—C14 | 78.9 (4) |
| C2—C3—C4—C11 | −0.7 (8) | C14—C15—C16—C17 | −3.7 (8) |
| C2—C3—C4—C5 | 179.5 (6) | S1—C15—C16—C17 | 177.2 (4) |
| C11—C4—C5—C6 | −1.1 (10) | C15—C16—C17—C18 | 2.4 (9) |
| C3—C4—C5—C6 | 178.7 (6) | C16—C17—C18—C19 | −0.1 (9) |
| C4—C5—C6—C7 | 0.9 (11) | C17—C18—C19—C14 | −0.9 (9) |
| C5—C6—C7—C8 | 179.3 (7) | C15—C14—C19—C18 | −0.4 (8) |
| C5—C6—C7—C12 | −0.9 (10) | C13—C14—C19—C18 | −179.9 (5) |
| C12—C7—C8—C9 | −0.7 (9) | O1—C13—O2—Pb1i | −11.8 (5) |
| C6—C7—C8—C9 | 179.2 (6) | C14—C13—O2—Pb1i | 163.3 (4) |
| Symmetry code: (i) −x+2, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O6—H6A···O2i | 0.92 (12) | 1.88 (12) | 2.770 (7) | 163 (10) |
| Symmetry code: (i) −x+2, −y+1, −z+1. |
Experimental details
| Crystal data |
| Chemical formula | [Pb2(C7H4O5S)2(C12H8N2)2]·2H2O |
| Mr | 605.57 |
| Crystal system, space group | Triclinic, P1 |
| Temperature (K) | 298 |
| a, b, c (Å) | 7.3232 (7), 10.8613 (10), 12.0558 (11) |
| α, β, γ (°) | 97.280 (1), 100.320 (2), 91.150 (1) |
| V (Å3) | 934.89 (15) |
| Z | 2 |
| Radiation type | Mo Kα |
| µ (mm−1) | 9.18 |
| Crystal size (mm) | 0.20 × 0.15 × 0.10 |
| |
| Data collection |
| Diffractometer | Bruker APEX CCD area-detector
diffractometer |
| Absorption correction | Numerical
(SADABS; Bruker, 2002) |
| Tmin, Tmax | 0.034, 0.227 |
No. of measured, independent and
observed [I > 2σ(I)] reflections | 6867, 3352, 3162 |
| Rint | 0.024 |
| (sin θ/λ)max (Å−1) | 0.600 |
| |
| Refinement |
| R[F2 > 2σ(F2)], wR(F2), S | 0.025, 0.062, 1.05 |
| No. of reflections | 3352 |
| No. of parameters | 270 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement |
| Δρmax, Δρmin (e Å−3) | 1.75, −0.60 |
Selected geometric parameters (Å, º) top| Pb1—N1 | 2.540 (4) | Pb1—O1i | 2.734 (3) |
| Pb1—N2 | 2.569 (4) | S1—O5 | 1.428 (4) |
| Pb1—O1 | 2.574 (3) | S1—O4 | 1.465 (4) |
| Pb1—O2i | 2.595 (4) | S1—O3 | 1.469 (4) |
| Pb1—O3 | 2.646 (4) | | |
| | | |
| N1—Pb1—N2 | 65.03 (12) | O1—Pb1—O3 | 73.54 (11) |
| N1—Pb1—O1 | 77.52 (12) | O2i—Pb1—O3 | 156.80 (12) |
| N2—Pb1—O1 | 137.24 (12) | N1—Pb1—O1i | 95.32 (12) |
| N1—Pb1—O2i | 74.86 (12) | N2—Pb1—O1i | 127.26 (11) |
| N2—Pb1—O2i | 78.37 (12) | O1—Pb1—O1i | 74.01 (12) |
| O1—Pb1—O2i | 111.94 (12) | O2i—Pb1—O1i | 48.91 (11) |
| N1—Pb1—O3 | 84.87 (12) | O3—Pb1—O1i | 146.69 (10) |
| N2—Pb1—O3 | 82.95 (11) | | |
| | | |
| N1—Pb1—O1—Pb1i | 99.33 (14) | O3—Pb1—O1—Pb1i | −172.39 (15) |
| N2—Pb1—O1—Pb1i | 128.15 (15) | O1i—Pb1—O1—Pb1i | 0.0 |
| O2i—Pb1—O1—Pb1i | 31.51 (16) | | |
| Symmetry code: (i) −x+2, −y+1, −z+1. |
Hydrogen-bond geometry (Å, º) top
| D—H···A | D—H | H···A | D···A | D—H···A |
| O6—H6A···O2i | 0.92 (12) | 1.88 (12) | 2.770 (7) | 163 (10) |
| Symmetry code: (i) −x+2, −y+1, −z+1. |

 journal menu
journal menu








![[Figure 1]](http://journals.iucr.org/c/issues/2004/09/00/av1191/av1191fig1thm.gif)
![[Figure 2]](http://journals.iucr.org/c/issues/2004/09/00/av1191/av1191fig2thm.gif)




Lead(II) is capable of exhibiting variable coordination number and geometry with or without a stereochemically active lone pair of electrons (Wang & Vittal, 2003). Of these, a number of polymeric PbII compounds have been structurally characterized (Cecconi et al., 2003; Bridgewater & Parkin, 2000; Ying et al., 2003). The absence of crystal field-stabilization energy effects also allows PbII cations to adopt a range of different coordination geometries, not restricted to octahedral, tetrahedral or square-planar (Foreman et al., 2000). 2-Sulfobenzenecarboxylate (sbc) is a very interesting ligand, with both carboxylate and sulfonate as potential coordinating groups. It can form not only short bridges between metal ions via one carboxylate end or one sulfonate end, but also a long bridge via the benzene ring. Thus, we have selected the Pb-sbc system to extend this research and we present here the crystal structure of the title compound, [Pb(sbc)(phen)]2·2H2O, (I). \sch
In (I), each PbII ion coordinates to three O atoms from two carboxylate groups of two sbc anions, with a typical range of Pb—O(carboxylate) distances [2.574 (3)–2.595 (4) Å; Foreman et al., 2000], one sulfonate O atom from one sbc anion, with Pb—O distances of 2.646 (4) Å, and two N atoms from one 1,10-phenanthroline ligand, with Pb—N distances of 2.540 (4) and 2.569 (4) Å. The coordination numbers of the two PbII ions are equivalent, but although both PbII cations are six-coordinated, due to the absence of crystal field-stabilization energy effects, the coordination geometries of the PbII cations are not octahedral (Fig. 1). The two N atoms of 1,10-phenanthroline and two O atoms of the carboxylate are chelated to each PbII cation, and the sulfonate group of the other sbc is monodentate. One O atom of the chelated carboxylate group also bridges to the other PbII cation, with Pb—O distances of 2.734 (3) Å. Thus, each pair of PbII ions has the same coordination environment and is bridged by two sbc anions, forming a centrosymmetric dinuclear ring. Each pair of PbII ions is therefore connected by two different kinds of bridges, namely a carboxylate short bridge and a carboxylate-sulfonate long bridge. There is also a special position of 1 site symmetry at the centre of the two PbII cations.
The S1—O3, S1—O4 and S1—O5 distances are 1.469 (4), 1.465 (4) and 1.428 (4) Å, respectively, and all fall within the typical range of S—O bond distances in the sulfonate anion (1.40–1.49 Å; Onoda et al., 2001). The similarity of the three S—O bond distances suggests that strong conjugation of the sulfonate is predominant in (I). In addition, each sbc anion acts as a tetradentate ligand, connecting two PbII ions through its bridging and chelating carboxylate group and its monodentate sulfonate group. This coordination mode is quite different from that in the above-mentioned polymeric PbII compounds, in which the carboxylate groups are either bidentate chelating or monodentate bridging.
It should be noted that hydrogen-bonding interactions and π–π interactions play an important role in the solid-state structure of (I), as shown in Fig. 2. Adjacent units are connected by hydrogen-bond and π–π stacking, resulting in a packing framework stucture.