Open  access
access
 access
accessIn the title compound, C13H10Cl2N2O, the ring N atom and its three attached atoms are essentially coplanar with angles adding to 359.8°, indicating conjugation with the 2-formylacrylonitrile subunit. The aldehyde group is oriented to place the carbonyl O atom 2.02 (3) Å from the N—H hydrogen atom. Intramolecular N—H O and C—H
O and C—H Cl interactions occur. The geometry of the exocyclic double bond is Z. In the crystal, weak C—H
Cl interactions occur. The geometry of the exocyclic double bond is Z. In the crystal, weak C—H N hydrogen bonds link the molecules into chains along [1
N hydrogen bonds link the molecules into chains along [1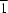 0].
0].
 O and C—H
O and C—H Cl interactions occur. The geometry of the exocyclic double bond is Z. In the crystal, weak C—H
Cl interactions occur. The geometry of the exocyclic double bond is Z. In the crystal, weak C—H N hydrogen bonds link the molecules into chains along [1
N hydrogen bonds link the molecules into chains along [1Open  access
access
 access
accessReaction of 2-(4-chloro-3,3,7-trimethyl-2,3-dihydro-1H-indol-2-ylidene)propanedial with hydroxylamine gives the title compound, C14H14ClN3O, in which the ring N atom is essentially planar [sum of angles around the ring N atom = 361°], indicating conjugation with the 2-cyanoacrylamide unit. The orientation of the acetamide group arises from intramolecular hydrogen bonding between the indole N—H and carbonyl groups. In the crystal, inversion-related acetamide groups form N—H O hydrogen-bonded dimers in graph-set R22(8) motifs, whilst dimers are also formed by pairs of amine–nitrile N—H
O hydrogen-bonded dimers in graph-set R22(8) motifs, whilst dimers are also formed by pairs of amine–nitrile N—H N hydrogen bonds in R22(12) motifs. These interactions together generate ribbons that propagate along the b-axis direction.
N hydrogen bonds in R22(12) motifs. These interactions together generate ribbons that propagate along the b-axis direction.
 O hydrogen-bonded dimers in graph-set R22(8) motifs, whilst dimers are also formed by pairs of amine–nitrile N—H
O hydrogen-bonded dimers in graph-set R22(8) motifs, whilst dimers are also formed by pairs of amine–nitrile N—H N hydrogen bonds in R22(12) motifs. These interactions together generate ribbons that propagate along the b-axis direction.
N hydrogen bonds in R22(12) motifs. These interactions together generate ribbons that propagate along the b-axis direction.
 journal menu
journal menu











