The molecules of the title compound, C17H21N3OS, are characterized by a wide C-C-C angle at the methine C atom linking the aryl and thiazolidine rings, associated with a short repulsive intramolecular S H contact between atoms in these two rings. A single piperidine-arene C-H
H contact between atoms in these two rings. A single piperidine-arene C-H
![[pi]](/logos/entities/pi_rmgif.gif) hydrogen bond links pairs of molecules into centrosymmetric dimers.
hydrogen bond links pairs of molecules into centrosymmetric dimers.
 H contact between atoms in these two rings. A single piperidine-arene C-H
H contact between atoms in these two rings. A single piperidine-arene C-H
![[pi]](/logos/entities/pi_rmgif.gif) hydrogen bond links pairs of molecules into centrosymmetric dimers.
hydrogen bond links pairs of molecules into centrosymmetric dimers.In methyl 4-(4-chloroanilino)-3-nitrobenzoate, C14H11ClN2O4, (I), there is an intramolecular N-H O hydrogen bond and the intramolecular distances provide evidence for electronic polarization of the o-quinonoid type. The molecules are linked into sheets built from N-H
O hydrogen bond and the intramolecular distances provide evidence for electronic polarization of the o-quinonoid type. The molecules are linked into sheets built from N-H O, C-H
O, C-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds, together with an aromatic
(arene) hydrogen bonds, together with an aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction. The molecules of methyl 1-benzyl-2-(4-chlorophenyl)-1H-benzimidazole-5-carboxylate, C22H17ClN2O2, (II), are also linked into sheets, this time by a combination of C-H
stacking interaction. The molecules of methyl 1-benzyl-2-(4-chlorophenyl)-1H-benzimidazole-5-carboxylate, C22H17ClN2O2, (II), are also linked into sheets, this time by a combination of C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds and aromatic
(arene) hydrogen bonds and aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interactions.
stacking interactions.
 O hydrogen bond and the intramolecular distances provide evidence for electronic polarization of the o-quinonoid type. The molecules are linked into sheets built from N-H
O hydrogen bond and the intramolecular distances provide evidence for electronic polarization of the o-quinonoid type. The molecules are linked into sheets built from N-H O, C-H
O, C-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds, together with an aromatic
(arene) hydrogen bonds, together with an aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction. The molecules of methyl 1-benzyl-2-(4-chlorophenyl)-1H-benzimidazole-5-carboxylate, C22H17ClN2O2, (II), are also linked into sheets, this time by a combination of C-H
stacking interaction. The molecules of methyl 1-benzyl-2-(4-chlorophenyl)-1H-benzimidazole-5-carboxylate, C22H17ClN2O2, (II), are also linked into sheets, this time by a combination of C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds and aromatic
(arene) hydrogen bonds and aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interactions.
stacking interactions.In each of ethyl N-{2-amino-5-formyl-6-[methyl(phenyl)amino]pyrimidin-4-yl}glycinate, C16H19N5O3, (I), N-{2-amino-5-formyl-6-[methyl(phenyl)amino]pyrimidin-4-yl}glycinamide, C14H16N6O2, (II), and ethyl 3-amino-N-{2-amino-5-formyl-6-[methyl(phenyl)amino]pyrimidin-4-yl}propionate, C17H21N5O3, (III), the pyrimidine ring is effectively planar, but in each of methyl N-{2-amino-6-[benzyl(methyl)amino]-5-formylpyrimidin-4-yl}glycinate, C16H19N5O3, (IV), ethyl 3-amino-N-{2-amino-6-[benzyl(methyl)amino]-5-formylpyrimidin-4-yl}propionate, C18H23N5O3, (V), and ethyl 3-amino-N-[2-amino-5-formyl-6-(piperidin-4-yl)pyrimidin-4-yl]propionate, C15H23N5O3, (VI), the pyrimidine ring is folded into a boat conformation. The bond lengths in each of (I)-(VI) provide evidence for significant polarization of the electronic structure. The molecules of (I) are linked by paired N-H N hydrogen bonds to form isolated dimeric aggregates, and those of (III) are linked by a combination of N-H
N hydrogen bonds to form isolated dimeric aggregates, and those of (III) are linked by a combination of N-H N and N-H
N and N-H O hydrogen bonds into a chain of edge-fused rings. In the structure of (IV), molecules are linked into sheets by means of two hydrogen bonds, both of N-H
O hydrogen bonds into a chain of edge-fused rings. In the structure of (IV), molecules are linked into sheets by means of two hydrogen bonds, both of N-H O type, in the structure of (V) by three hydrogen bonds, two of N-H
O type, in the structure of (V) by three hydrogen bonds, two of N-H N type and one of C-H
N type and one of C-H O type, and in the structure of (VI) by four hydrogen bonds, all of N-H
O type, and in the structure of (VI) by four hydrogen bonds, all of N-H O type. Molecules of (II) are linked into a three-dimensional framework structure by a combination of three N-H
O type. Molecules of (II) are linked into a three-dimensional framework structure by a combination of three N-H O hydrogen bonds and one C-H
O hydrogen bonds and one C-H O hydrogen bond.
O hydrogen bond.
 N hydrogen bonds to form isolated dimeric aggregates, and those of (III) are linked by a combination of N-H
N hydrogen bonds to form isolated dimeric aggregates, and those of (III) are linked by a combination of N-H N and N-H
N and N-H O hydrogen bonds into a chain of edge-fused rings. In the structure of (IV), molecules are linked into sheets by means of two hydrogen bonds, both of N-H
O hydrogen bonds into a chain of edge-fused rings. In the structure of (IV), molecules are linked into sheets by means of two hydrogen bonds, both of N-H O type, in the structure of (V) by three hydrogen bonds, two of N-H
O type, in the structure of (V) by three hydrogen bonds, two of N-H N type and one of C-H
N type and one of C-H O type, and in the structure of (VI) by four hydrogen bonds, all of N-H
O type, and in the structure of (VI) by four hydrogen bonds, all of N-H O type. Molecules of (II) are linked into a three-dimensional framework structure by a combination of three N-H
O type. Molecules of (II) are linked into a three-dimensional framework structure by a combination of three N-H O hydrogen bonds and one C-H
O hydrogen bonds and one C-H O hydrogen bond.
O hydrogen bond.The molecular skeleton of the title compound, C11H9F3N4O2, is almost planar and exhibits a polarized (charge-separated) electronic structure in the nitroaniline portion. Molecules are linked by N-H N and C-H
N and C-H O hydrogen bonds to form a chain in which centrosymmetric R22(6) and R22(16) rings alternate.
O hydrogen bonds to form a chain in which centrosymmetric R22(6) and R22(16) rings alternate.
 N and C-H
N and C-H O hydrogen bonds to form a chain in which centrosymmetric R22(6) and R22(16) rings alternate.
O hydrogen bonds to form a chain in which centrosymmetric R22(6) and R22(16) rings alternate.(2R,4S)-2-(3-Methylthiophen-2-yl)-2,3,4,5-tetrahydro-1,4-epoxynaphtho[1,2-b]azepine, C19H17NOS, (I), crystallizes with a single enantiomer in each crystal, whereas its geometrical isomer (2RS,4SR)-2-(5-methylthiophen-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-naphtho[1,2-b]azepine, (II), and (2RS,4SR)-2-(5-bromothiophen-2-yl)-2,3,4,5-tetrahydro-1,4-epoxynaphtho[1,2-b]azepine, C18H14BrNOS, (III), both crystallize as racemic mixtures. A combination of one C-H O hydrogen bond and two C-H
O hydrogen bond and two C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds links the molecules of (I) into a three-dimensional framework; the molecules of (II) are linked into a C(4)C(4)[R22(7)] chain of rings by a combination of C-H
(arene) hydrogen bonds links the molecules of (I) into a three-dimensional framework; the molecules of (II) are linked into a C(4)C(4)[R22(7)] chain of rings by a combination of C-H N and C-H
N and C-H O hydrogen bonds; and in (III), where Z' = 2, a combination of four C-H
O hydrogen bonds; and in (III), where Z' = 2, a combination of four C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds and two C-H
(arene) hydrogen bonds and two C-H
![[pi]](/logos/entities/pi_rmgif.gif) (thienyl) hydrogen bonds links the molecules into complex sheets. Comparisons are made with the assembly patterns in some aryl-substituted 1,4-epoxynaphtho[1,2-b]azepines.
(thienyl) hydrogen bonds links the molecules into complex sheets. Comparisons are made with the assembly patterns in some aryl-substituted 1,4-epoxynaphtho[1,2-b]azepines.
 O hydrogen bond and two C-H
O hydrogen bond and two C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds links the molecules of (I) into a three-dimensional framework; the molecules of (II) are linked into a C(4)C(4)[R22(7)] chain of rings by a combination of C-H
(arene) hydrogen bonds links the molecules of (I) into a three-dimensional framework; the molecules of (II) are linked into a C(4)C(4)[R22(7)] chain of rings by a combination of C-H N and C-H
N and C-H O hydrogen bonds; and in (III), where Z' = 2, a combination of four C-H
O hydrogen bonds; and in (III), where Z' = 2, a combination of four C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds and two C-H
(arene) hydrogen bonds and two C-H
![[pi]](/logos/entities/pi_rmgif.gif) (thienyl) hydrogen bonds links the molecules into complex sheets. Comparisons are made with the assembly patterns in some aryl-substituted 1,4-epoxynaphtho[1,2-b]azepines.
(thienyl) hydrogen bonds links the molecules into complex sheets. Comparisons are made with the assembly patterns in some aryl-substituted 1,4-epoxynaphtho[1,2-b]azepines.(2RS,4SR)-7-Bromo-2-(2-methylphenyl)-2,3,4,5-tetrahydro-1H-naphtho[1,2-b]azepin-4-ol, C21H20BrNO, (I), and (2RS,4SR)-2-(3-methylthiophen-2-yl)-2,3,4,5-tetrahydro-1H-naphtho[1,2-b]azepin-4-ol, C19H19NOS, (II), both crystallize with Z' = 2 in the space groups P21/c and Cc, respectively; compound (II) crystallizes as a nonmerohedral twin, with twin fractions 0.183 (2) and 0.817 (2). The molecules of (I) are linked by O-H O and O-H
O and O-H N hydrogen bonds to form a cyclic centrosymmetric R44(16) tetramer. The molecules of (II) are linked by O-H
N hydrogen bonds to form a cyclic centrosymmetric R44(16) tetramer. The molecules of (II) are linked by O-H O hydrogen bonds to form a C22(4) chain and these chains are weakly linked by a single C-H
O hydrogen bonds to form a C22(4) chain and these chains are weakly linked by a single C-H
![[pi]](/logos/entities/pi_rmgif.gif) (thienyl) interaction to form a three-dimensional array. Comparisons are made with some related compounds.
(thienyl) interaction to form a three-dimensional array. Comparisons are made with some related compounds.
 O and O-H
O and O-H N hydrogen bonds to form a cyclic centrosymmetric R44(16) tetramer. The molecules of (II) are linked by O-H
N hydrogen bonds to form a cyclic centrosymmetric R44(16) tetramer. The molecules of (II) are linked by O-H O hydrogen bonds to form a C22(4) chain and these chains are weakly linked by a single C-H
O hydrogen bonds to form a C22(4) chain and these chains are weakly linked by a single C-H
![[pi]](/logos/entities/pi_rmgif.gif) (thienyl) interaction to form a three-dimensional array. Comparisons are made with some related compounds.
(thienyl) interaction to form a three-dimensional array. Comparisons are made with some related compounds.In the molecule of 4-(2-chlorophenyl)pyrrolo[1,2-a]quinoxaline, C17H11ClN2, (I), the bond lengths are consistent with electron delocalization in the two outer rings of the fused tricyclic system, with a localized double bond in the central ring. The molecules of (I) are linked into chains by a ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction. In (4RS)-4-(1,3-benzodioxol-6-yl)-4,5-dihydropyrrolo[1,2-a]quinoxaline, C18H14N2O2, (II), the central ring of the fused tricyclic system adopts a conformation intermediate between screw-boat and half-chair forms. A combination of N-H
stacking interaction. In (4RS)-4-(1,3-benzodioxol-6-yl)-4,5-dihydropyrrolo[1,2-a]quinoxaline, C18H14N2O2, (II), the central ring of the fused tricyclic system adopts a conformation intermediate between screw-boat and half-chair forms. A combination of N-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds links the molecules of (II) into a sheet. Comparisons are made with related compounds.
(arene) hydrogen bonds links the molecules of (II) into a sheet. Comparisons are made with related compounds.
![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction. In (4RS)-4-(1,3-benzodioxol-6-yl)-4,5-dihydropyrrolo[1,2-a]quinoxaline, C18H14N2O2, (II), the central ring of the fused tricyclic system adopts a conformation intermediate between screw-boat and half-chair forms. A combination of N-H
stacking interaction. In (4RS)-4-(1,3-benzodioxol-6-yl)-4,5-dihydropyrrolo[1,2-a]quinoxaline, C18H14N2O2, (II), the central ring of the fused tricyclic system adopts a conformation intermediate between screw-boat and half-chair forms. A combination of N-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds links the molecules of (II) into a sheet. Comparisons are made with related compounds.
(arene) hydrogen bonds links the molecules of (II) into a sheet. Comparisons are made with related compounds.The title compound, C16H9FN2S, crystallizes as a nonmerohedral twin with twin rotation about the reciprocal-lattice vector [10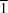 ]*. The molecules are nearly planar and the dihedral angle between the planes of the two aryl rings is only 4.4 (2)°. The molecules are linked by pairs of C-H
]*. The molecules are nearly planar and the dihedral angle between the planes of the two aryl rings is only 4.4 (2)°. The molecules are linked by pairs of C-H N hydrogen bonds to form cyclic centrosymmetric R22(18) dimers, which are linked into chains by an aromatic
N hydrogen bonds to form cyclic centrosymmetric R22(18) dimers, which are linked into chains by an aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction. Comparisons are made with some related 3-aryl-2-thienylacrylonitriles.
stacking interaction. Comparisons are made with some related 3-aryl-2-thienylacrylonitriles.
 N hydrogen bonds to form cyclic centrosymmetric R22(18) dimers, which are linked into chains by an aromatic
N hydrogen bonds to form cyclic centrosymmetric R22(18) dimers, which are linked into chains by an aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction. Comparisons are made with some related 3-aryl-2-thienylacrylonitriles.
stacking interaction. Comparisons are made with some related 3-aryl-2-thienylacrylonitriles.The molecules of both methyl 4-[2-(4-chlorobenzoyl)hydrazinyl]-3-nitrobenzoate, C15H12ClN3O5, (I), and methyl 4-[2-(2-fluorobenzoyl)hydrazinyl]-3-nitrobenzoate, C15H12FN3O5, (II), contain an intramolecular N-H O hydrogen bond, and both show electronic polarization in the nitrated aryl ring. In both compounds, molecules are linked by a combination of N-H
O hydrogen bond, and both show electronic polarization in the nitrated aryl ring. In both compounds, molecules are linked by a combination of N-H O and C-H
O and C-H O hydrogen bonds to form sheets, which are built from R43(18) rings in (I) and from R44(28) rings in (II). In each of methyl 3-phenyl-1,2,4-benzotriazine-6-carboxylate, C15H11N3O2, (III), and methyl 3-(4-methylphenyl)-1,2,4-benzotriazine-6-carboxylate, C16H13N3O2, (IV), the benzotriazine unit shows naphthalene-type delocalization. There are no hydrogen bonds in the structures of compounds (III) and (IV), but in both compounds, the molecules are linked into chains by
O hydrogen bonds to form sheets, which are built from R43(18) rings in (I) and from R44(28) rings in (II). In each of methyl 3-phenyl-1,2,4-benzotriazine-6-carboxylate, C15H11N3O2, (III), and methyl 3-(4-methylphenyl)-1,2,4-benzotriazine-6-carboxylate, C16H13N3O2, (IV), the benzotriazine unit shows naphthalene-type delocalization. There are no hydrogen bonds in the structures of compounds (III) and (IV), but in both compounds, the molecules are linked into chains by ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interactions involving the benzotriazine units. The mechanism of chain formation is the same in both (III) and (IV), and the different orientations of the two chains can be related to the approximate relationship between the unit-cell metrics for (III) and (IV).
stacking interactions involving the benzotriazine units. The mechanism of chain formation is the same in both (III) and (IV), and the different orientations of the two chains can be related to the approximate relationship between the unit-cell metrics for (III) and (IV).
 O hydrogen bond, and both show electronic polarization in the nitrated aryl ring. In both compounds, molecules are linked by a combination of N-H
O hydrogen bond, and both show electronic polarization in the nitrated aryl ring. In both compounds, molecules are linked by a combination of N-H O and C-H
O and C-H O hydrogen bonds to form sheets, which are built from R43(18) rings in (I) and from R44(28) rings in (II). In each of methyl 3-phenyl-1,2,4-benzotriazine-6-carboxylate, C15H11N3O2, (III), and methyl 3-(4-methylphenyl)-1,2,4-benzotriazine-6-carboxylate, C16H13N3O2, (IV), the benzotriazine unit shows naphthalene-type delocalization. There are no hydrogen bonds in the structures of compounds (III) and (IV), but in both compounds, the molecules are linked into chains by
O hydrogen bonds to form sheets, which are built from R43(18) rings in (I) and from R44(28) rings in (II). In each of methyl 3-phenyl-1,2,4-benzotriazine-6-carboxylate, C15H11N3O2, (III), and methyl 3-(4-methylphenyl)-1,2,4-benzotriazine-6-carboxylate, C16H13N3O2, (IV), the benzotriazine unit shows naphthalene-type delocalization. There are no hydrogen bonds in the structures of compounds (III) and (IV), but in both compounds, the molecules are linked into chains by ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interactions involving the benzotriazine units. The mechanism of chain formation is the same in both (III) and (IV), and the different orientations of the two chains can be related to the approximate relationship between the unit-cell metrics for (III) and (IV).
stacking interactions involving the benzotriazine units. The mechanism of chain formation is the same in both (III) and (IV), and the different orientations of the two chains can be related to the approximate relationship between the unit-cell metrics for (III) and (IV).The molecules of 3-amino-4-anilino-1H-isochromen-1-one, C15H12N2O2, (I), and 3-amino-4-[methyl(phenyl)amino]-1H-isochromen-1-one, C16H14N2O2, (II), adopt very similar conformations, with the substituted amino group PhNR, where R = H in (I) and R = Me in (II), almost orthogonal to the adjacent heterocyclic ring. The molecules of (I) are linked into cyclic centrosymmetric dimers by pairs of N-H O hydrogen bonds, while those of (II) are linked into complex sheets by a combination of one three-centre N-H
O hydrogen bonds, while those of (II) are linked into complex sheets by a combination of one three-centre N-H (O)2 hydrogen bond, one two-centre C-H
(O)2 hydrogen bond, one two-centre C-H O hydrogen bond and two C-H
O hydrogen bond and two C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds.
(arene) hydrogen bonds.
 O hydrogen bonds, while those of (II) are linked into complex sheets by a combination of one three-centre N-H
O hydrogen bonds, while those of (II) are linked into complex sheets by a combination of one three-centre N-H (O)2 hydrogen bond, one two-centre C-H
(O)2 hydrogen bond, one two-centre C-H O hydrogen bond and two C-H
O hydrogen bond and two C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds.
(arene) hydrogen bonds.In dibenzylammonium hydrogen maleate [or dibenzylammonium (2Z)-3-carboxyprop-2-enoate], C14H16N+·C4H3O4-, (I), the anion contains a fairly short and nearly linear O-H O hydrogen bond, with an O
O hydrogen bond, with an O ·O distance of 2.4603 (16) Å, but with the H atom clearly offset from the mid-point of the O
·O distance of 2.4603 (16) Å, but with the H atom clearly offset from the mid-point of the O O vector. The counter-ions in (I) are linked by two N-H
O vector. The counter-ions in (I) are linked by two N-H O hydrogen bonds to form C22(6) chains and these chains are weakly linked into sheets by a C-H
O hydrogen bonds to form C22(6) chains and these chains are weakly linked into sheets by a C-H O hydrogen bond. Bis(dibenzylamino)methane, C29H30N2, (II), crystallizes with two independent molecules lying across twofold rotation axes in the space group C2/c, and the molecules are conformationally chiral; there are no direction-specific intermolecular interactions in the crystal structure of (II).
O hydrogen bond. Bis(dibenzylamino)methane, C29H30N2, (II), crystallizes with two independent molecules lying across twofold rotation axes in the space group C2/c, and the molecules are conformationally chiral; there are no direction-specific intermolecular interactions in the crystal structure of (II).
 O hydrogen bond, with an O
O hydrogen bond, with an O ·O distance of 2.4603 (16) Å, but with the H atom clearly offset from the mid-point of the O
·O distance of 2.4603 (16) Å, but with the H atom clearly offset from the mid-point of the O O vector. The counter-ions in (I) are linked by two N-H
O vector. The counter-ions in (I) are linked by two N-H O hydrogen bonds to form C22(6) chains and these chains are weakly linked into sheets by a C-H
O hydrogen bonds to form C22(6) chains and these chains are weakly linked into sheets by a C-H O hydrogen bond. Bis(dibenzylamino)methane, C29H30N2, (II), crystallizes with two independent molecules lying across twofold rotation axes in the space group C2/c, and the molecules are conformationally chiral; there are no direction-specific intermolecular interactions in the crystal structure of (II).
O hydrogen bond. Bis(dibenzylamino)methane, C29H30N2, (II), crystallizes with two independent molecules lying across twofold rotation axes in the space group C2/c, and the molecules are conformationally chiral; there are no direction-specific intermolecular interactions in the crystal structure of (II).The molecules contain an intramolecular π–π stacking interaction, and C—H O and C—H
O and C—H N hydrogen bonds link the molecules to form a bilayer having tert-butyl groups on both faces.
N hydrogen bonds link the molecules to form a bilayer having tert-butyl groups on both faces.
 O and C—H
O and C—H N hydrogen bonds link the molecules to form a bilayer having tert-butyl groups on both faces.
N hydrogen bonds link the molecules to form a bilayer having tert-butyl groups on both faces.Two very closely related pyrrolo[3,4-c]pyrrole derivatives form hydrogen-bonded sheets using a different combination of C-H N, C-H
N, C-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) interactions.
interactions.
 N, C-H
N, C-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) interactions.
interactions.An unexpected but high-yield reaction product forms sheets of ![[pi]](/logos/entities/pi_rmgif.gif) -stacked hydrogen-bonded dimers.
-stacked hydrogen-bonded dimers.
![[pi]](/logos/entities/pi_rmgif.gif) -stacked hydrogen-bonded dimers.
-stacked hydrogen-bonded dimers.The central C-C-C angle is 120.3 (2)° and the molecules are linked by N-H O hydrogen bonds into a ribbon containing edge-fused S(8) and
O hydrogen bonds into a ribbon containing edge-fused S(8) and 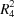 (8) rings.
(8) rings.
 O hydrogen bonds into a ribbon containing edge-fused S(8) and
O hydrogen bonds into a ribbon containing edge-fused S(8) and Hydrogen-bonded sheets of organic molecules are linked into a three-dimensional layer structure by cyclic centrosymmetric water tetramers.
Corrigendum to Acta Cryst. (2009), C65, o549-o552
.
A tetrahydropyrimidin-4(3H)-one, prepared by mild oxidation of the corresponding hexahydro analogue, forms hydrogen-bonded 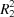 (8) dimers.
(8) dimers.

 journal menu
journal menu


























