Six closely related pyrazolo[3,4-b]pyridine derivatives, namely 6-chloro-3-methyl-1,4-diphenylpyrazolo[3,4-b]pyridine-5-carbaldehyde, C20H14ClN3O, (I), 6-chloro-3-methyl-4-(4-methylphenyl)-1-phenylpyrazolo[3,4-b]pyridine-5-carbaldehyde, C21H16ClN3O, (II), 6-chloro-4-(4-chlorophenyl)-3-methyl-1-phenylpyrazolo[3,4-b]pyridine-5-carbaldehyde, C20H13Cl2N3O, (III), 4-(4-bromophenyl)-6-chloro-3-methyl-1-phenylpyrazolo[3,4-b]pyridine-5-carbaldehyde, C20H13BrClN3O, (IV), 6-chloro-4-(4-methoxyphenyl)-3-methyl-1-phenylpyrazolo[3,4-b]pyridine-5-carbaldehyde, C21H16ClN3O2, (V), and 6-chloro-3-methyl-4-(4-nitrophenyl)-1-phenylpyrazolo[3,4-b]pyridine-5-carbaldehyde, C20H13ClN4O3, (VI), which differ only in the identity of a single small substituent on one of the aryl rings, crystallize in four different space groups spanning three crystal systems. The molecules of (I) are linked into a chain of rings by a combination of C-H N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds; those of (II), (IV) and (V), which all crystallize in the space group P
(arene) hydrogen bonds; those of (II), (IV) and (V), which all crystallize in the space group P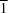 , are each linked by two independent C-H
, are each linked by two independent C-H O hydrogen bonds to form chains of edge-fused rings running in different directions through the three unit cells; the molecules of (III) are linked into complex sheets by a combination of two C-H
O hydrogen bonds to form chains of edge-fused rings running in different directions through the three unit cells; the molecules of (III) are linked into complex sheets by a combination of two C-H O hydrogen bonds and one C-H
O hydrogen bonds and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond; finally, the molecules of (VI) are linked by a single C-H
(arene) hydrogen bond; finally, the molecules of (VI) are linked by a single C-H O hydrogen bond to form a simple chain.
O hydrogen bond to form a simple chain.
 N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds; those of (II), (IV) and (V), which all crystallize in the space group P
(arene) hydrogen bonds; those of (II), (IV) and (V), which all crystallize in the space group P O hydrogen bonds to form chains of edge-fused rings running in different directions through the three unit cells; the molecules of (III) are linked into complex sheets by a combination of two C-H
O hydrogen bonds to form chains of edge-fused rings running in different directions through the three unit cells; the molecules of (III) are linked into complex sheets by a combination of two C-H O hydrogen bonds and one C-H
O hydrogen bonds and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond; finally, the molecules of (VI) are linked by a single C-H
(arene) hydrogen bond; finally, the molecules of (VI) are linked by a single C-H O hydrogen bond to form a simple chain.
O hydrogen bond to form a simple chain.The molecules of the title compound, C20H15BrClNO, are linked into chains by a C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond, in which the acceptor is the brominated ring of the naphthalene unit, and these chains are linked by an aromatic
(arene) hydrogen bond, in which the acceptor is the brominated ring of the naphthalene unit, and these chains are linked by an aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction, again involving the naphthalene unit, into a sheet structure.
stacking interaction, again involving the naphthalene unit, into a sheet structure.

![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond, in which the acceptor is the brominated ring of the naphthalene unit, and these chains are linked by an aromatic
(arene) hydrogen bond, in which the acceptor is the brominated ring of the naphthalene unit, and these chains are linked by an aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interaction, again involving the naphthalene unit, into a sheet structure.
stacking interaction, again involving the naphthalene unit, into a sheet structure.The closely related compounds cis-(2RS,4SR)-2-(thiophen-2-yl)-2,3,4,5-tetrahydro-1H-1-benzazepin-4-ol, C14H15NOS, (I), and cis-(2RS,4SR)-2-(5-methylthiophen-2-yl)-2,3,4,5-tetrahydro-1H-1-benzazepin-4-ol, C15H17NOS, (II), both crystallize with Z' = 2 in the space group P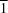 . In (I), the thienyl substituent in one of the two independent molecules is disordered over two sets of atomic sites having occupancies of 0.856 (2) and 0.144 (2). In both compounds, the two independent hydroxy O atoms are both within 2.8 Å of the hydroxy O atoms of two neighbouring molecules, and all of the hydroxy H atoms are disordered, each over two sites. The resulting O-H
. In (I), the thienyl substituent in one of the two independent molecules is disordered over two sets of atomic sites having occupancies of 0.856 (2) and 0.144 (2). In both compounds, the two independent hydroxy O atoms are both within 2.8 Å of the hydroxy O atoms of two neighbouring molecules, and all of the hydroxy H atoms are disordered, each over two sites. The resulting O-H O hydrogen bonds generate two similar but distinct C44(8) chains, depending upon which hydroxy H-atom sites are occupied or vacant, with full correlation of the hydroxy H-atom occupancies within each chain. Comparisons are made with the supramolecular assembly in some related compounds.
O hydrogen bonds generate two similar but distinct C44(8) chains, depending upon which hydroxy H-atom sites are occupied or vacant, with full correlation of the hydroxy H-atom occupancies within each chain. Comparisons are made with the supramolecular assembly in some related compounds.
 O hydrogen bonds generate two similar but distinct C44(8) chains, depending upon which hydroxy H-atom sites are occupied or vacant, with full correlation of the hydroxy H-atom occupancies within each chain. Comparisons are made with the supramolecular assembly in some related compounds.
O hydrogen bonds generate two similar but distinct C44(8) chains, depending upon which hydroxy H-atom sites are occupied or vacant, with full correlation of the hydroxy H-atom occupancies within each chain. Comparisons are made with the supramolecular assembly in some related compounds.The structures are reported of nine closely related tetrahydro-1,4-epoxy-1-benzazepines carrying pendant heterocyclic substituents, namely: 2-exo-(5-nitrofuran-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C14H12N2O4, (I), 7-fluoro-2-exo-(1-methyl-1H-pyrrol-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C15H15FN2O, (II), 7-fluoro-2-exo-(5-methylfuran-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C15H14FNO2, (III), 7-fluoro-2-exo-(3-methylthiophen-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C15H14FNOS, (IV), 7-fluoro-2-exo-(5-methylthiophen-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C15H14FNOS, (V), 7-chloro-2-exo-(5-methylfuran-2-yl)-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C15H14ClNO2, (VI), 2-exo-(5-methylfuran-2-yl)-7-trifluoromethoxy-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C16H14F3NO3, (VII), 2-exo-(3-methylthiophen-2-yl)-7-trifluoromethoxy-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C16H14F3NO2S, (VIII), and 2-exo-(5-nitrofuran-2-yl)-7-trifluoromethoxy-2,3,4,5-tetrahydro-1,4-epoxy-1H-1-benzazepine, C15H11F3N2O5, (IX). All nine compounds crystallize in centrosymmetric space groups as racemic mixtures with configuration (2RS,4SR). There are no direction-specific interactions between the molecules in (V). The molecules in (III), (IV), (VI) and (VII) are linked into simple chains, by means of a single C-H O hydrogen bond in each of (III), (VI) and (VII), and by means of a single C-H
O hydrogen bond in each of (III), (VI) and (VII), and by means of a single C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond in (IV), while the molecules in (VIII) are linked into a chain of rings. In each of (I) and (II), a combination of one C-H
(arene) hydrogen bond in (IV), while the molecules in (VIII) are linked into a chain of rings. In each of (I) and (II), a combination of one C-H O hydrogen bond and one C-H
O hydrogen bond and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond links the molecules into sheets, albeit of completely different construction in the two compounds. In (IX), the sheet structure is built from a combination of four independent C-H
(arene) hydrogen bond links the molecules into sheets, albeit of completely different construction in the two compounds. In (IX), the sheet structure is built from a combination of four independent C-H O hydrogen bonds and one C-H
O hydrogen bonds and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond. Comparisons are made with some related compounds.
(arene) hydrogen bond. Comparisons are made with some related compounds.
 O hydrogen bond in each of (III), (VI) and (VII), and by means of a single C-H
O hydrogen bond in each of (III), (VI) and (VII), and by means of a single C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond in (IV), while the molecules in (VIII) are linked into a chain of rings. In each of (I) and (II), a combination of one C-H
(arene) hydrogen bond in (IV), while the molecules in (VIII) are linked into a chain of rings. In each of (I) and (II), a combination of one C-H O hydrogen bond and one C-H
O hydrogen bond and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond links the molecules into sheets, albeit of completely different construction in the two compounds. In (IX), the sheet structure is built from a combination of four independent C-H
(arene) hydrogen bond links the molecules into sheets, albeit of completely different construction in the two compounds. In (IX), the sheet structure is built from a combination of four independent C-H O hydrogen bonds and one C-H
O hydrogen bonds and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond. Comparisons are made with some related compounds.
(arene) hydrogen bond. Comparisons are made with some related compounds.(2RS,4RS)-7-Fluoro-2-(2-phenylethyl)-2,3,4,5-tetrahydro-1H-1,4-epoxy-1-benzazepine, C18H18FNO, (I), and (2RS,4RS)-7-chloro-2-(2-phenylethyl)-2,3,4,5-tetrahydro-1H-1,4-epoxy-1-benzazepine, C18H18ClNO, (II), are isomorphous but not strictly isostructural, as the slight differences in the unit-cell dimensions and molecular conformations are sufficient to preclude, in the structure of (I), the direction-specific intermolecular interactions present in the structure of (II), where the molecules are linked into sheets by a combination of C-H N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds.
(arene) hydrogen bonds.
 N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds.
(arene) hydrogen bonds.In both 2-amino-6-methoxy-4-(4-methylanilino)-5-nitrosopyrimidine, C12H13N5O2, (I), and ethyl N-[4-(1-adamantylamino)-2-amino-5-nitrosopyrimidin-6-yl]-3-aminopropionate, C19H28N6O3, (II), the nitrosopyrimidine unit is planar and the bond distances provide evidence for significant polarization of the electronic structures. In (II), the ethoxycarbonyl fragment of the molecule is disordered over two sets of sites with occupancies of 0.910 (4) and 0.090 (4). In the molecules of both compounds, there is an intramolecular N-H O hydrogen bond. The molecules of (I) are linked into a chain of rings by a combination of N-H
O hydrogen bond. The molecules of (I) are linked into a chain of rings by a combination of N-H O and C-H
O and C-H O hydrogen bonds, while the molecules of (II) are linked by a two-centre N-H
O hydrogen bonds, while the molecules of (II) are linked by a two-centre N-H N hydrogen bond and a three-centre N-H
N hydrogen bond and a three-centre N-H (N,O) hydrogen bond to form sheets containing four distinct types of ring.
(N,O) hydrogen bond to form sheets containing four distinct types of ring.
 O hydrogen bond. The molecules of (I) are linked into a chain of rings by a combination of N-H
O hydrogen bond. The molecules of (I) are linked into a chain of rings by a combination of N-H O and C-H
O and C-H O hydrogen bonds, while the molecules of (II) are linked by a two-centre N-H
O hydrogen bonds, while the molecules of (II) are linked by a two-centre N-H N hydrogen bond and a three-centre N-H
N hydrogen bond and a three-centre N-H (N,O) hydrogen bond to form sheets containing four distinct types of ring.
(N,O) hydrogen bond to form sheets containing four distinct types of ring.1,5-Bis(4-chlorophenyl)-3-(2-oxo-1,2-dihydroquinolin-3-yl)pentane-1,5-dione, (Ia), and 1,5-bis(2-chlorophenyl)-3-(2-oxo-1,2-dihydroquinolin-3-yl)pentane-1,5-dione, (Ib), crystallize as an 84:16 mixture, 0.84C26H19Cl2NO3·0.16C26H19Cl2NO3, in the space group I41/a, where the molecules of the two isomers occupy very similar sites in the unit cell. A combination of one N—H O hydrogen bond and one C—H
O hydrogen bond and one C—H O hydrogen bond links the molecules, regardless of isomeric form, into a single three-dimensional framework structure. The molecules of (9RS,10RS)-8,9-bis(4-chlorobenzyl)-10-(2-oxo-1,2-dihydroquinolin-3-yl)-5,6,9,10-tetrahydrophenanthridine, C36H22Cl2N2O4, (II), are linked by two hydrogen bonds, one each of the N—H
O hydrogen bond links the molecules, regardless of isomeric form, into a single three-dimensional framework structure. The molecules of (9RS,10RS)-8,9-bis(4-chlorobenzyl)-10-(2-oxo-1,2-dihydroquinolin-3-yl)-5,6,9,10-tetrahydrophenanthridine, C36H22Cl2N2O4, (II), are linked by two hydrogen bonds, one each of the N—H O and C—H
O and C—H O types, into a molecular ribbon in which centrosymmetric rings of R22(18) and R44(24) types alternate. The hydrogen-bonded ribbons enclose channels, which contain highly disordered solvent molecules.
O types, into a molecular ribbon in which centrosymmetric rings of R22(18) and R44(24) types alternate. The hydrogen-bonded ribbons enclose channels, which contain highly disordered solvent molecules.
 O hydrogen bond and one C—H
O hydrogen bond and one C—H O hydrogen bond links the molecules, regardless of isomeric form, into a single three-dimensional framework structure. The molecules of (9RS,10RS)-8,9-bis(4-chlorobenzyl)-10-(2-oxo-1,2-dihydroquinolin-3-yl)-5,6,9,10-tetrahydrophenanthridine, C36H22Cl2N2O4, (II), are linked by two hydrogen bonds, one each of the N—H
O hydrogen bond links the molecules, regardless of isomeric form, into a single three-dimensional framework structure. The molecules of (9RS,10RS)-8,9-bis(4-chlorobenzyl)-10-(2-oxo-1,2-dihydroquinolin-3-yl)-5,6,9,10-tetrahydrophenanthridine, C36H22Cl2N2O4, (II), are linked by two hydrogen bonds, one each of the N—H O and C—H
O and C—H O types, into a molecular ribbon in which centrosymmetric rings of R22(18) and R44(24) types alternate. The hydrogen-bonded ribbons enclose channels, which contain highly disordered solvent molecules.
O types, into a molecular ribbon in which centrosymmetric rings of R22(18) and R44(24) types alternate. The hydrogen-bonded ribbons enclose channels, which contain highly disordered solvent molecules.In the title compound, C4H6N4S·0.5H2O, there are two independent pyrimidinethione units, both of which lie across mirror planes in the space group Cmca. Hence, the H atoms bonded to the ring N atoms in each molecule are disordered over two symmetry-related sites, each having an occupancy of 0.5. The water molecule lies across a twofold rotation axis parallel to [010]. The molecular components of (I) are linked by seven independent hydrogen bonds, of N-H N, N-H
N, N-H S, N-H
S, N-H O and O-H
O and O-H S types. A combination of disordered N-H
S types. A combination of disordered N-H N hydrogen bonds and ordered N-H
N hydrogen bonds and ordered N-H S hydrogen bonds links the pyrimidinethione units into a continuous tubular structure. The water molecule acts as both a double donor of hydrogen bonds and a double acceptor, forming hydrogen bonds with components of four distinct pyrimidinethione tubes, thus linking these tubes into a three-dimensional structure.
S hydrogen bonds links the pyrimidinethione units into a continuous tubular structure. The water molecule acts as both a double donor of hydrogen bonds and a double acceptor, forming hydrogen bonds with components of four distinct pyrimidinethione tubes, thus linking these tubes into a three-dimensional structure.
 N, N-H
N, N-H S, N-H
S, N-H O and O-H
O and O-H S types. A combination of disordered N-H
S types. A combination of disordered N-H N hydrogen bonds and ordered N-H
N hydrogen bonds and ordered N-H S hydrogen bonds links the pyrimidinethione units into a continuous tubular structure. The water molecule acts as both a double donor of hydrogen bonds and a double acceptor, forming hydrogen bonds with components of four distinct pyrimidinethione tubes, thus linking these tubes into a three-dimensional structure.
S hydrogen bonds links the pyrimidinethione units into a continuous tubular structure. The water molecule acts as both a double donor of hydrogen bonds and a double acceptor, forming hydrogen bonds with components of four distinct pyrimidinethione tubes, thus linking these tubes into a three-dimensional structure.The racemic title dipyrrolopyrrolizine compound crystallizes from dimethylformamide as a disolvate, C55H39Cl2N7O6·2C3H7NO. None of the four fused heterocyclic rings is planar; one adopts an envelope conformation, two others adopt half-chair conformations and the fourth adopts a conformation intermediate between an envelope and a half-chair. The arrangement of the ring fusions is such as to preclude the possibility of internal mirror symmetry. The three independent molecular components are weakly linked by C-H O hydrogen bonds, and the dipyrrolopyrrolizine molecules are linked by a combination of four C-H
O hydrogen bonds, and the dipyrrolopyrrolizine molecules are linked by a combination of four C-H O and one C-H
O and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond to form a three-dimensional framework, from which the dimethylformamide solvent molecules are pendent. However, aromatic
(arene) hydrogen bond to form a three-dimensional framework, from which the dimethylformamide solvent molecules are pendent. However, aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interactions are absent in the structure.
stacking interactions are absent in the structure.
 O hydrogen bonds, and the dipyrrolopyrrolizine molecules are linked by a combination of four C-H
O hydrogen bonds, and the dipyrrolopyrrolizine molecules are linked by a combination of four C-H O and one C-H
O and one C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond to form a three-dimensional framework, from which the dimethylformamide solvent molecules are pendent. However, aromatic
(arene) hydrogen bond to form a three-dimensional framework, from which the dimethylformamide solvent molecules are pendent. However, aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) stacking interactions are absent in the structure.
stacking interactions are absent in the structure.The non-H atoms in the organic component of the title compound, C8H7N3OS2·H2O, are almost coplanar, as the dihedral angle between the two ring planes is only 1.8 (2)°; there is a wide C-C-C angle of 127.8 (3)° at the methine C atom linking the two rings. The molecular components are linked into a three-dimensional framework structure by two-centre hydrogen bonds of N-H O and O-H
O and O-H N types, together with a three-centre O-H
N types, together with a three-centre O-H (N,S) system. Comparisons are made with some (Z)-5-arylmethylidene-2-sulfanylidene-1,3-thiazolidin-4-ones.
(N,S) system. Comparisons are made with some (Z)-5-arylmethylidene-2-sulfanylidene-1,3-thiazolidin-4-ones.
 O and O-H
O and O-H N types, together with a three-centre O-H
N types, together with a three-centre O-H (N,S) system. Comparisons are made with some (Z)-5-arylmethylidene-2-sulfanylidene-1,3-thiazolidin-4-ones.
(N,S) system. Comparisons are made with some (Z)-5-arylmethylidene-2-sulfanylidene-1,3-thiazolidin-4-ones.
 journal menu
journal menu


















