The cation of the title salt, C12H9N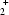 ·C8H4NO
·C8H4NO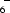 , is a monoprotonated 1,10-phenanthroline molecule, whereas its anion is a monodeprotonated 4-nitrophthalic acid molecule. There is a strong, almost symmetrical, intramolecular hydrogen bond linking the O atoms of neighbouring carboxylate groups within the anion [O—H = 1.13 (4) Å, H
, is a monoprotonated 1,10-phenanthroline molecule, whereas its anion is a monodeprotonated 4-nitrophthalic acid molecule. There is a strong, almost symmetrical, intramolecular hydrogen bond linking the O atoms of neighbouring carboxylate groups within the anion [O—H = 1.13 (4) Å, H O = 1.27 (4) Å, O
O = 1.27 (4) Å, O O = 2.395 (3) Å and O—H
O = 2.395 (3) Å and O—H O = 173 (4)°]. An N—H
O = 173 (4)°]. An N—H O bond [N—H = 1.00 (3) Å, H
O bond [N—H = 1.00 (3) Å, H O = 1.70 (3) Å, N
O = 1.70 (3) Å, N O = 2.668 (3) Å and N—H
O = 2.668 (3) Å and N—H O = 160 (3)°] is responsible for formation of ion pairs, which are further loosely aggregated into a three-dimensional framework via C—H
O = 160 (3)°] is responsible for formation of ion pairs, which are further loosely aggregated into a three-dimensional framework via C—H O and π–π interactions.
O and π–π interactions.
 O = 1.27 (4) Å, O
O = 1.27 (4) Å, O O = 2.395 (3) Å and O—H
O = 2.395 (3) Å and O—H O = 173 (4)°]. An N—H
O = 173 (4)°]. An N—H O bond [N—H = 1.00 (3) Å, H
O bond [N—H = 1.00 (3) Å, H O = 1.70 (3) Å, N
O = 1.70 (3) Å, N O = 2.668 (3) Å and N—H
O = 2.668 (3) Å and N—H O = 160 (3)°] is responsible for formation of ion pairs, which are further loosely aggregated into a three-dimensional framework via C—H
O = 160 (3)°] is responsible for formation of ion pairs, which are further loosely aggregated into a three-dimensional framework via C—H O and π–π interactions.
O and π–π interactions.In the title salt, C12H9N2+·C8H4NO6−, one N atom of 1,10-phenanthroline is protonated [N—H = 1.00 (2) Å], while the other N atom is not. Protonation of the N atom causes the C—N—C angle to increase [122.3 (2)° in the protonated ring versus 116.2 (2)° in the unprotonated ring]. In the crystal structure, N—H O and O—H
O and O—H O hydrogen bonds and van der Waals forces stabilize the packing of the ions.
O hydrogen bonds and van der Waals forces stabilize the packing of the ions.
 O and O—H
O and O—H O hydrogen bonds and van der Waals forces stabilize the packing of the ions.
O hydrogen bonds and van der Waals forces stabilize the packing of the ions. In the title compound, C11H11N2O7P, compression of the P—O—C angles is observed, suggesting that some strain is probably present. The configuration around phosphorus is distorted tetrahedral. In the crystal structure, N—H O and C—H
O and C—H O hydrogen-bond contacts and van der Waals forces stabilize the packing of the compound.
O hydrogen-bond contacts and van der Waals forces stabilize the packing of the compound.
 O and C—H
O and C—H O hydrogen-bond contacts and van der Waals forces stabilize the packing of the compound.
O hydrogen-bond contacts and van der Waals forces stabilize the packing of the compound.In the title salt, C5H13N2+·C7H5INO2−, the packing of the ions is stabilized by N—H O hydrogen bonds and van der Waals forces.
O hydrogen bonds and van der Waals forces.
 O hydrogen bonds and van der Waals forces.
O hydrogen bonds and van der Waals forces.In the crystal structure of the title compound, C11H11N2O7P, the carboxamide molecule and the solvent molecule of crystallization each possess mirror symmetry; for the former, the mirror plane bisects the two-ring OP(OCH2)3C cage. The packing is stabilized by an intermolecular N—H O hydrogen bond and a number of C—H
O hydrogen bond and a number of C—H O hydrogen bonds.
O hydrogen bonds.
 O hydrogen bond and a number of C—H
O hydrogen bond and a number of C—H O hydrogen bonds.
O hydrogen bonds.
 journal menu
journal menu














