In the title mixed-ligand metal-organic polymeric complex, {[Co(NCS)2(C8H12N6)2]·2H2O}n, the asymmetric unit contains a divalent CoII cation, which sits on an inversion centre, two halves of two crystallographically distinct and centrosymmetric 1,4-bis(1,2,4-triazol-1-yl)butane (BTB) ligands, one N-bound thiocyanate ligand and one solvent water molecule. The CoII atom possesses a distorted {CoN6} octahedral geometry, with the equatorial positions taken up by triazole N atoms from four different BTB ligands. The axial positions are filled by thiocyanate N atoms. In the crystal, each CoII atom is linked covalently to four others through the distal donors of the tethering BTB ligands, forming a neutral (4,4)-topology two-dimensional rhomboid grid layer motif, which is coincident with the (1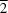 1) crystal planes. Magnetic investigations show that weak antiferromagnetic coupling exists between CoII atoms in the complex.
1) crystal planes. Magnetic investigations show that weak antiferromagnetic coupling exists between CoII atoms in the complex.
In the crystal structure of the title two-dimensional metal-organic polymeric complex, [Cd2Cl4(C8H14N2O4)(H2O)2]n, the asymmetric unit contains a crystallographically independent CdII cation, two chloride ligands, an aqua ligand and half a 2,2'-(piperazine-1,4-diium-1,4-diyl)diacetate (H2PDA) ligand, the piperazine ring centroid of which is located on a crystallographic inversion centre. Each CdII centre is six-coordinated in an octahedral environment by an O atom from an H2PDA ligand and an O atom from an aqua ligand in a trans disposition, and by four chloride ligands arranged in the plane perpendicular to the O-Cd-O axis. The complex forms a two-dimensional layer polymer containing [CdCl2]n chains, which are interconnected into an extensive three-dimensional hydrogen-bonded network by C-H O, C-H
O, C-H Cl and O-H
Cl and O-H O hydrogen bonds.
O hydrogen bonds.
 O, C-H
O, C-H Cl and O-H
Cl and O-H O hydrogen bonds.
O hydrogen bonds.
 journal menu
journal menu






![[mu]](/logos/entities/mu_rmgif.gif)
![[kappa]](/logos/entities/kappa_rmgif.gif)




