The crystal structure of the title compound, (NH4)3[Sb2I9], which crystallizes in the monoclinic space group P21/n, resembles that of hexagonal Cs3[Sb2I9], which crystallizes in space group P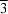 m1. The title compound consists of double layers parallel to (001). Each layer is made up of distorted corner-sharing SbI6 octahedra. One-third of the ammonium cations reside within six-membered rings formed by corner-sharing SbI6 octahedra within the layers. The other ammonium cations are situated within three-membered rings built from corner-sharing SbI6 octahedra near the edges of the layers.
m1. The title compound consists of double layers parallel to (001). Each layer is made up of distorted corner-sharing SbI6 octahedra. One-third of the ammonium cations reside within six-membered rings formed by corner-sharing SbI6 octahedra within the layers. The other ammonium cations are situated within three-membered rings built from corner-sharing SbI6 octahedra near the edges of the layers.
The title compound, (C5H6N)2[CdBr4]·H2O, consists of discrete anions, cations and solvent water molecules. Both pyridinium cations and the tetrabromocadmate anion possess a crystallographically imposed mirror symmetry. The solvent water molecule and one pyridinium cation form intermolecular N—H O and O—H
O and O—H Br hydrogen bonds, giving a three-dimensional hydrogen-bonded structure.
Br hydrogen bonds, giving a three-dimensional hydrogen-bonded structure.
 O and O—H
O and O—H Br hydrogen bonds, giving a three-dimensional hydrogen-bonded structure.
Br hydrogen bonds, giving a three-dimensional hydrogen-bonded structure. In the title compound, [NiCl2(C5H5N)4]·0.76H2O, the nickel(II) ion is coordinated in a trans fashion by two Cl− ions and four pyridine molecules. The molecule possesses crystallographic 222 symmetry. The Ni atom has a distorted octahedral geometry, with long Ni—Cl bonds. In the crystal structure, O—H Cl hydrogen bonding forms a three-dimensional network.
Cl hydrogen bonding forms a three-dimensional network.
 Cl hydrogen bonding forms a three-dimensional network.
Cl hydrogen bonding forms a three-dimensional network.The title compound, (C3H5N2)2[SnCl6], contains discrete [SnCl6]2− anions and two imidazolium (C3H5N2+) cations. The SnIV atom is located on a center of inversion and is octahedrally coordinated by six Cl− ions. N—H Cl hydrogen bonds form a three-dimensional hydrogen-bonded structure.
Cl hydrogen bonds form a three-dimensional hydrogen-bonded structure.
 Cl hydrogen bonds form a three-dimensional hydrogen-bonded structure.
Cl hydrogen bonds form a three-dimensional hydrogen-bonded structure. The title compound, (C5H6N)2[CoBr4], contains discrete [CoBr4]2- and pyridinium C5H6N+ ions. Hydrogen bonds give rise to a three-dimensional network structure.
The title compound, (C3H5N2)2[CoCl4], contains discrete [CoCl4]2− anions and two imidazolium C3H5N2+ cations. Various N—H Cl hydrogen bonds form a three-dimensional network.
Cl hydrogen bonds form a three-dimensional network.
 Cl hydrogen bonds form a three-dimensional network.
Cl hydrogen bonds form a three-dimensional network. In the title compound, [Co(C3H4N2)4(H2O)2]Br2, the Co atom is situated on an inversion center. It is coordinated by four N atoms of four imidazole ligands and two O atoms of water molecules and has an octahedral geometry. In the crystal structure, water molecules, imidazole NH groups and Br atoms contribute to the formation of a three-dimensional hydrogen-bonded network.
The structure of the title compound, [NiBr2(C3H4N2)2(H2O)2], consists of monomers with inversion symmetry. The three monodentate ligands (imidazole, bromine and aqua), together with their symmetry equivalents, define an almost perfect octahedral coordination. Hydrogen-bonding interactions via the NH group of imidazole, Br and aqua H atoms lead to a three-dimensional network.

 journal menu
journal menu

















