The structure of tetragonal (P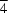 21m) ammonium chlorite, NH4ClO2, has been redetermined by single-crystal X-ray analysis with higher precision. The NH4+ cation is at a site of
21m) ammonium chlorite, NH4ClO2, has been redetermined by single-crystal X-ray analysis with higher precision. The NH4+ cation is at a site of 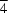 symmetry, while the unique Cl and O atoms are at sites with symmetries mm and m, respectively. The structure consists of corrugated layers of NH4+ cations coordinated by ClO2- anions. The H atoms of the ammonium cation hydrogen bond to four chlorite O atoms. The resultant coordination is almost ideal tetrahedral.
symmetry, while the unique Cl and O atoms are at sites with symmetries mm and m, respectively. The structure consists of corrugated layers of NH4+ cations coordinated by ClO2- anions. The H atoms of the ammonium cation hydrogen bond to four chlorite O atoms. The resultant coordination is almost ideal tetrahedral.
The asymmetric unit of the title compound, [Ru(OH)(NH3)4(NO)][Pd(NO2)4]·H2O, consists of one complex [Ru(NO)(NH3)4OH]2+ cation, two half [Pd(NO2)4]2- complex anions, each anion located on an inversion centre, and a water molecule of crystallization. The crystal structure exhibits an extensive hydrogen-bonding network.
The title compound, K6[Os2(CN)8(SO2)2]·2H2O, was prepared from the reaction of OsO4 with elemental Se and S2Cl2 at 473 K and further reaction with aqueous KCN solution. It contains a centrosymmetric binuclear [Os2S2O4(CN)8]6− cluster anion.
The structure of calcium chlorite, Ca(ClO2)2, has been refined from X-ray powder diffraction data using the Rietveld method. The compound crystallizes in the orthorhombic space group Ccca (Ccce), with Z = 4. The structure is based on separate layers parallel to the ac plane, consisting of calcium cations that are coordinated by chlorite anions; the O atoms form almost ideal square antiprisms. Within the layers, each anion bridges four metal cations. The Ca atoms are located on special positions of 222 symmetry, the Cl atoms lie on twofold axes and the O atoms are in general positions. The compound is isostructural with strontium chlorite, Sr(ClO2)2, and lead chlorite, Pb(ClO2)2.
The structure of strontium chlorite, Sr(ClO2)2, has been refined from X-ray powder diffraction data using the Rietveld method. The compound crystallizes in the orthorhombic space group Ccca (Ccce), with Z = 4. The structure is based on separate layers parallel to the ac plane, consisting of strontium cations that are coordinated by chlorite anions; the O atoms form almost ideal square antiprisms. Within the layers, each anion bridges four metal cations. The Sr atoms are located on special positions of 222 symmetry, the Cl atoms lie on twofold axes and the O atoms are in general positions. The compound is isostructural with calcium chlorite, Ca(ClO2)2, and lead chlorite, Pb(ClO2)2.
The title compound, (C8H20N)[Nb(C6H4S2)2(C6H4S3)], is the major product of the reaction of (Et4N)4[Nb2S4(NCS)8] and disodium benzene-1,2-dithiolate. The coordination environment of the Nb atom contains seven S atoms, viz. four S atoms from two benzene-1,2-dithiolate ligands and three S atoms from 2-disulfanylbenzenethiolate. The Nb-S distances are in the range 2.4391 (6)-2.5932 (7) Å.

 journal menu
journal menu















