In the polymeric title compound, [CuI(C10H8N4)]n, the CuI atom is in a four-coordinated tetrahedral geometry, formed by two I atoms and two pyridine N atoms from two different 4,4'-(diazenediyl)dipyridine (4,4'-azpy) ligands. Two ![[mu]](/logos/entities/mu_rmgif.gif) 2-I atoms link two CuI atoms to form a planar rhomboid [Cu2I2] cluster located on an inversion centre, where the distance between two CuI atoms is 2.7781 (15) Å and the Cu-I bond lengths are 2.6290 (13) and 2.7495 (15) Å. The bridging 4,4'-azpy ligands connect the [Cu2I2] clusters into a two-dimensional (2-D) double-layered grid-like network [parallel to the (10
2-I atoms link two CuI atoms to form a planar rhomboid [Cu2I2] cluster located on an inversion centre, where the distance between two CuI atoms is 2.7781 (15) Å and the Cu-I bond lengths are 2.6290 (13) and 2.7495 (15) Å. The bridging 4,4'-azpy ligands connect the [Cu2I2] clusters into a two-dimensional (2-D) double-layered grid-like network [parallel to the (10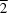 ) plane], with a (4,4)-connected topology. Two 2-D grid-like networks interweave each other by long 4,4'-azpy bridging ligands to form a dense 2-D double-layered network. To the best of our knowledge, this interwoven 2-D
) plane], with a (4,4)-connected topology. Two 2-D grid-like networks interweave each other by long 4,4'-azpy bridging ligands to form a dense 2-D double-layered network. To the best of our knowledge, this interwoven 2-D![[rightwards arrow]](/logos/entities/rarr_rmgif.gif) 2-D network is observed for the first time in [Cu2I2]-organic compounds.
2-D network is observed for the first time in [Cu2I2]-organic compounds.
![[mu]](/logos/entities/mu_rmgif.gif) 2-I atoms link two CuI atoms to form a planar rhomboid [Cu2I2] cluster located on an inversion centre, where the distance between two CuI atoms is 2.7781 (15) Å and the Cu-I bond lengths are 2.6290 (13) and 2.7495 (15) Å. The bridging 4,4'-azpy ligands connect the [Cu2I2] clusters into a two-dimensional (2-D) double-layered grid-like network [parallel to the (10
2-I atoms link two CuI atoms to form a planar rhomboid [Cu2I2] cluster located on an inversion centre, where the distance between two CuI atoms is 2.7781 (15) Å and the Cu-I bond lengths are 2.6290 (13) and 2.7495 (15) Å. The bridging 4,4'-azpy ligands connect the [Cu2I2] clusters into a two-dimensional (2-D) double-layered grid-like network [parallel to the (10![[rightwards arrow]](/logos/entities/rarr_rmgif.gif) 2-D network is observed for the first time in [Cu2I2]-organic compounds.
2-D network is observed for the first time in [Cu2I2]-organic compounds.A one-dimensional polymeric structure of CoII with sulfadiazine and 1,3-bis(pyridin-4-yl)propane is further stabilized by intermolecular hydrogen bonding. The structure is of interest with respect to its electrochemical properties in the reduction reaction of H2O2 to H2O. Investigation of the thermal stability shows that the complex is stable up to 543 K.
A one-dimensional polymeric structure of CdII with sulfaquinoxaline is further stabilized by intermolecular hydrogen bonding. The fluorescence spectrum reveals that the complex emits strong blue fluorescence. The thermal analysis shows that the framework is stable up to 663 K.

 journal menu
journal menu











