Two polymorphs of bis(2-carbamoylguanidinium) fluorophosphonate dihydrate, 2C2H7N4O+·FO3P2-·2H2O, are presented. Polymorph (I), crystallizing in the space group Pnma, is slightly less densely packed than polymorph (II), which crystallizes in Pbca. In (I), the fluorophosphonate anion is situated on a crystallographic mirror plane and the O atom of the water molecule is disordered over two positions, in contrast with its H atoms. The hydrogen-bond patterns in both polymorphs share similar features. There are O-H O and N-H
O and N-H O hydrogen bonds in both structures. The water molecules donate their H atoms to the O atoms of the fluorophosphonates exclusively. The water molecules and the fluorophosphonates participate in the formation of R44(10) graph-set motifs. These motifs extend along the a axis in each structure. The water molecules are also acceptors of either one [in (I) and (II)] or two [in (II)] N-H
O hydrogen bonds in both structures. The water molecules donate their H atoms to the O atoms of the fluorophosphonates exclusively. The water molecules and the fluorophosphonates participate in the formation of R44(10) graph-set motifs. These motifs extend along the a axis in each structure. The water molecules are also acceptors of either one [in (I) and (II)] or two [in (II)] N-H O hydrogen bonds. The water molecules are significant building elements in the formation of a three-dimensional hydrogen-bond network in both structures. Despite these similarities, there are substantial differences between the hydrogen-bond networks of (I) and (II). The N-H
O hydrogen bonds. The water molecules are significant building elements in the formation of a three-dimensional hydrogen-bond network in both structures. Despite these similarities, there are substantial differences between the hydrogen-bond networks of (I) and (II). The N-H O and O-H
O and O-H O hydrogen bonds in (I) are stronger and weaker, respectively, than those in (II). Moreover, in (I), the shortest N-H
O hydrogen bonds in (I) are stronger and weaker, respectively, than those in (II). Moreover, in (I), the shortest N-H O hydrogen bonds are shorter than the shortest O-H
O hydrogen bonds are shorter than the shortest O-H O hydrogen bonds, which is an unusual feature. The properties of the hydrogen-bond network in (II) can be related to an unusually long P-O bond length for an unhydrogenated fluorophosphonate anion that is present in this structure. In both structures, the N-H
O hydrogen bonds, which is an unusual feature. The properties of the hydrogen-bond network in (II) can be related to an unusually long P-O bond length for an unhydrogenated fluorophosphonate anion that is present in this structure. In both structures, the N-H F interactions are far weaker than the N-H
F interactions are far weaker than the N-H O hydrogen bonds. It follows from the structure analysis that (II) seems to be thermodynamically more stable than (I).
O hydrogen bonds. It follows from the structure analysis that (II) seems to be thermodynamically more stable than (I).
 O and N-H
O and N-H O hydrogen bonds in both structures. The water molecules donate their H atoms to the O atoms of the fluorophosphonates exclusively. The water molecules and the fluorophosphonates participate in the formation of R44(10) graph-set motifs. These motifs extend along the a axis in each structure. The water molecules are also acceptors of either one [in (I) and (II)] or two [in (II)] N-H
O hydrogen bonds in both structures. The water molecules donate their H atoms to the O atoms of the fluorophosphonates exclusively. The water molecules and the fluorophosphonates participate in the formation of R44(10) graph-set motifs. These motifs extend along the a axis in each structure. The water molecules are also acceptors of either one [in (I) and (II)] or two [in (II)] N-H O hydrogen bonds. The water molecules are significant building elements in the formation of a three-dimensional hydrogen-bond network in both structures. Despite these similarities, there are substantial differences between the hydrogen-bond networks of (I) and (II). The N-H
O hydrogen bonds. The water molecules are significant building elements in the formation of a three-dimensional hydrogen-bond network in both structures. Despite these similarities, there are substantial differences between the hydrogen-bond networks of (I) and (II). The N-H O and O-H
O and O-H O hydrogen bonds in (I) are stronger and weaker, respectively, than those in (II). Moreover, in (I), the shortest N-H
O hydrogen bonds in (I) are stronger and weaker, respectively, than those in (II). Moreover, in (I), the shortest N-H O hydrogen bonds are shorter than the shortest O-H
O hydrogen bonds are shorter than the shortest O-H O hydrogen bonds, which is an unusual feature. The properties of the hydrogen-bond network in (II) can be related to an unusually long P-O bond length for an unhydrogenated fluorophosphonate anion that is present in this structure. In both structures, the N-H
O hydrogen bonds, which is an unusual feature. The properties of the hydrogen-bond network in (II) can be related to an unusually long P-O bond length for an unhydrogenated fluorophosphonate anion that is present in this structure. In both structures, the N-H F interactions are far weaker than the N-H
F interactions are far weaker than the N-H O hydrogen bonds. It follows from the structure analysis that (II) seems to be thermodynamically more stable than (I).
O hydrogen bonds. It follows from the structure analysis that (II) seems to be thermodynamically more stable than (I).The title compounds, 2-carbamoylguanidinium hydrogen fluorophosphonate, C2H7N4O+·HFO3P-, (I), 2-carbamoylguanidinium-hydrogen fluorophosphonate-hydrogen phosphite (1/0.76/0.24), C2H7N4O+·0.76HFO3P-·0.24H2O3P-, (II), and 2-carbamoylguanidinium-hydrogen fluorophosphonate-hydrogen phosphite (1/0.115/0.885), C2H7N4O+·0.115HFO3P-·0.885H2O3P-, (III), are isostructural with guanylurea hydrogen phosphite, C2H7N4O+·H2O3P- [Fridrichová, Nemec, Císarová & Nemec (2010). CrystEngComm, 12, 2054-2056]. They constitute structures where the hydrogen phosphite anion has been fully or partially replaced by hydrogen fluorophosphonate. The title structures are the fourth example of isostructural compounds which differ by the presence of hydrogen fluorophosphonate and hydrogen phosphite or fluorophosphonate and phosphite anions. Moreover, the present study reports structures with these mixed anions for the first time. In the reported mixed salts, the P and O atoms of either anion overlap almost exactly, as can be judged by comparison of their equivalent isotropic displacement parameters, while the P-F and P-H directions are almost parallel. There are strong O-H O hydrogen bonds between the anions, as well as strong N-H
O hydrogen bonds between the anions, as well as strong N-H O hydrogen bonds between the 2-carbamoylguanidinium cations in the title structures. Altogether they form a three-dimensional hydrogen-bond pattern. Interestingly, rare N-H
O hydrogen bonds between the 2-carbamoylguanidinium cations in the title structures. Altogether they form a three-dimensional hydrogen-bond pattern. Interestingly, rare N-H F interactions are also present in the title structures. Another exceptional feature concerns the P-O(H) distances, which are about as long as the P-F distance. The dependence of P-F distances on the longest P-O distances in FO3P2- or HFO3P- is presented. The greater content of hydrogen phosphite in the mixed crystals causes a larger deformation of the cations from planarity.
F interactions are also present in the title structures. Another exceptional feature concerns the P-O(H) distances, which are about as long as the P-F distance. The dependence of P-F distances on the longest P-O distances in FO3P2- or HFO3P- is presented. The greater content of hydrogen phosphite in the mixed crystals causes a larger deformation of the cations from planarity.
 O hydrogen bonds between the anions, as well as strong N-H
O hydrogen bonds between the anions, as well as strong N-H O hydrogen bonds between the 2-carbamoylguanidinium cations in the title structures. Altogether they form a three-dimensional hydrogen-bond pattern. Interestingly, rare N-H
O hydrogen bonds between the 2-carbamoylguanidinium cations in the title structures. Altogether they form a three-dimensional hydrogen-bond pattern. Interestingly, rare N-H F interactions are also present in the title structures. Another exceptional feature concerns the P-O(H) distances, which are about as long as the P-F distance. The dependence of P-F distances on the longest P-O distances in FO3P2- or HFO3P- is presented. The greater content of hydrogen phosphite in the mixed crystals causes a larger deformation of the cations from planarity.
F interactions are also present in the title structures. Another exceptional feature concerns the P-O(H) distances, which are about as long as the P-F distance. The dependence of P-F distances on the longest P-O distances in FO3P2- or HFO3P- is presented. The greater content of hydrogen phosphite in the mixed crystals causes a larger deformation of the cations from planarity.In N,N′-di-tert-butyl-N′′,N′′-dimethylphosphoric triamide, C10H26N3OP, (I), and N,N′,N′′,N′′′-tetra-tert-butoxybis(phosphonic diamide), C16H40N4O3P2, (II), the extended structures are mediated by P(O) (H—N)2 interactions. The asymmetric unit of (I) consists of six independent molecules which aggregate through P(O)
(H—N)2 interactions. The asymmetric unit of (I) consists of six independent molecules which aggregate through P(O) (H—N)2 hydrogen bonds, giving R21(6) loops and forming two independent chains parallel to the a axis. Of the 12 independent tert-butyl groups, five are disordered over two different positions with occupancies ranging from
(H—N)2 hydrogen bonds, giving R21(6) loops and forming two independent chains parallel to the a axis. Of the 12 independent tert-butyl groups, five are disordered over two different positions with occupancies ranging from 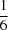 to
to 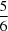 . In the structure of (II), the asymmetric unit contains one molecule. P(O)
. In the structure of (II), the asymmetric unit contains one molecule. P(O) (H—N)2 hydrogen bonds give S(6) and R22(8) rings, and the molecules form extended chains parallel to the c axis. The structures of (I) and (II), along with similar structures having (N)P(O)(NH)2 and (NH)2P(O)(O)P(O)(NH)2 skeletons extracted from the Cambridge Structural Database, are used to compare hydrogen-bond patterns in these families of phosphoramidates. The strengths of P(O)[
(H—N)2 hydrogen bonds give S(6) and R22(8) rings, and the molecules form extended chains parallel to the c axis. The structures of (I) and (II), along with similar structures having (N)P(O)(NH)2 and (NH)2P(O)(O)P(O)(NH)2 skeletons extracted from the Cambridge Structural Database, are used to compare hydrogen-bond patterns in these families of phosphoramidates. The strengths of P(O)[ H—N]x (x = 1, 2 or 3) hydrogen bonds are also analysed, using these compounds and previously reported structures with (N)2P(O)(NH) and P(O)(NH)3 fragments.
H—N]x (x = 1, 2 or 3) hydrogen bonds are also analysed, using these compounds and previously reported structures with (N)2P(O)(NH) and P(O)(NH)3 fragments.
 (H—N)2 interactions. The asymmetric unit of (I) consists of six independent molecules which aggregate through P(O)
(H—N)2 interactions. The asymmetric unit of (I) consists of six independent molecules which aggregate through P(O) (H—N)2 hydrogen bonds, giving R21(6) loops and forming two independent chains parallel to the a axis. Of the 12 independent tert-butyl groups, five are disordered over two different positions with occupancies ranging from
(H—N)2 hydrogen bonds, giving R21(6) loops and forming two independent chains parallel to the a axis. Of the 12 independent tert-butyl groups, five are disordered over two different positions with occupancies ranging from  (H—N)2 hydrogen bonds give S(6) and R22(8) rings, and the molecules form extended chains parallel to the c axis. The structures of (I) and (II), along with similar structures having (N)P(O)(NH)2 and (NH)2P(O)(O)P(O)(NH)2 skeletons extracted from the Cambridge Structural Database, are used to compare hydrogen-bond patterns in these families of phosphoramidates. The strengths of P(O)[
(H—N)2 hydrogen bonds give S(6) and R22(8) rings, and the molecules form extended chains parallel to the c axis. The structures of (I) and (II), along with similar structures having (N)P(O)(NH)2 and (NH)2P(O)(O)P(O)(NH)2 skeletons extracted from the Cambridge Structural Database, are used to compare hydrogen-bond patterns in these families of phosphoramidates. The strengths of P(O)[ H—N]x (x = 1, 2 or 3) hydrogen bonds are also analysed, using these compounds and previously reported structures with (N)2P(O)(NH) and P(O)(NH)3 fragments.
H—N]x (x = 1, 2 or 3) hydrogen bonds are also analysed, using these compounds and previously reported structures with (N)2P(O)(NH) and P(O)(NH)3 fragments.
 journal menu
journal menu











