The bond distances in the title compound, C11H16N4, provide evidence for peripheral delocalization of ![[pi]](/logos/entities/pi_rmgif.gif) electrons. The molecules are linked by two independent N-H
electrons. The molecules are linked by two independent N-H N hydrogen bonds into a three-dimensional framework structure.
N hydrogen bonds into a three-dimensional framework structure.
![[pi]](/logos/entities/pi_rmgif.gif) electrons. The molecules are linked by two independent N-H
electrons. The molecules are linked by two independent N-H N hydrogen bonds into a three-dimensional framework structure.
N hydrogen bonds into a three-dimensional framework structure.In the title compound, C13H10O4, prepared by the condensation reaction between phthalic anhydride and pentane-2,4-dione, one of the acetyl groups in the pentane-2,4-dione fragment is almost coplanar with the isobenzofuran ring system, while the other group is twisted out of this plane by almost 80°. The corresponding C-C and C-O distances in the two acetyl units are consistent with dipolar delocalization into the coplanar unit only. There is no supramolecular aggregation of the molecules, but a number of rather short intermolecular contacts of C-H O, O
O, O O and O
O and O C types occur.
C types occur.
 O, O
O, O O and O
O and O C types occur.
C types occur.In both 2,5-dimethyl-6,7-dihydrobenzo[h]pyrazolo[1,5-a]quinazoline, C16H15N3, (I), and 2-tert-butyl-5-methyl-6,7-dihydrobenzo[h]pyrazolo[1,5-a]quinazoline, C19H21N3, (II), which crystallizes with Z' = 2 in the space group P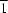 , the non-aromatic carbocyclic rings adopt screw-boat conformations. The molecules of (I) are linked into chains of rings by a combination of C-H
, the non-aromatic carbocyclic rings adopt screw-boat conformations. The molecules of (I) are linked into chains of rings by a combination of C-H N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.
(arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.
 N and C-H
N and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.
(arene) hydrogen bonds, while in (II) there are no hydrogen bonds of any kind.The title compound, C30H26ClN3, crystallizes with Z' = 3 in the P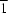 space group. The three independent molecules have very similar, although not identical, conformations, with almost perfect screw-boat forms adopted by the non-aromatic carbocyclic rings. Four independent C-H
space group. The three independent molecules have very similar, although not identical, conformations, with almost perfect screw-boat forms adopted by the non-aromatic carbocyclic rings. Four independent C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds link the molecules into centrosymmetric six-component aggregates.
(arene) hydrogen bonds link the molecules into centrosymmetric six-component aggregates.

![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds link the molecules into centrosymmetric six-component aggregates.
(arene) hydrogen bonds link the molecules into centrosymmetric six-component aggregates.In the title compound, C23H24N4O3, the pyrimidine ring adopts an almost perfect boat conformation, and the bond distances provide evidence for some polarization of the molecular-electronic structure. Two independent N-H O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
 O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
 journal menu
journal menu





 access
access







