In the triclinic polymorph of 2-iodo-4-nitroaniline, C6H5IN2O2, space group P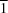 , the molecules are linked by paired N-H
, the molecules are linked by paired N-H O hydrogen bonds into C(8)[R
O hydrogen bonds into C(8)[R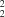 (6)] chains of rings. These chains are linked into sheets by nitro
(6)] chains of rings. These chains are linked into sheets by nitro I interactions, and the sheets are pairwise linked by aromatic
I interactions, and the sheets are pairwise linked by aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) -stacking interactions. In the orthorhombic polymorph, space group Pbca, the molecules are linked by single N-H
-stacking interactions. In the orthorhombic polymorph, space group Pbca, the molecules are linked by single N-H O hydrogen bonds into spiral C(8) chains; the chains are linked by nitro
O hydrogen bonds into spiral C(8) chains; the chains are linked by nitro O interactions into sheets, each of which is linked to its two immediate neighbours by aromatic
O interactions into sheets, each of which is linked to its two immediate neighbours by aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) -stacking interactions, so producing a continuous three-dimensional structure.
-stacking interactions, so producing a continuous three-dimensional structure.
 O hydrogen bonds into C(8)[R
O hydrogen bonds into C(8)[R I interactions, and the sheets are pairwise linked by aromatic
I interactions, and the sheets are pairwise linked by aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) -stacking interactions. In the orthorhombic polymorph, space group Pbca, the molecules are linked by single N-H
-stacking interactions. In the orthorhombic polymorph, space group Pbca, the molecules are linked by single N-H O hydrogen bonds into spiral C(8) chains; the chains are linked by nitro
O hydrogen bonds into spiral C(8) chains; the chains are linked by nitro O interactions into sheets, each of which is linked to its two immediate neighbours by aromatic
O interactions into sheets, each of which is linked to its two immediate neighbours by aromatic ![[pi]](/logos/entities/pi_rmgif.gif) -
-![[pi]](/logos/entities/pi_rmgif.gif) -stacking interactions, so producing a continuous three-dimensional structure.
-stacking interactions, so producing a continuous three-dimensional structure.Molecules of the title compound, C12H10N2O2, are markedly non-planar. There is an intramolecular N-H O hydrogen bond, and the molecules are linked into zigzag chains by a single C-H
O hydrogen bond, and the molecules are linked into zigzag chains by a single C-H O hydrogen bond. Comparisons are made with the supramolecular aggregation in isomeric amino-nitro derivatives, and in some N-methylnitroanilines.
O hydrogen bond. Comparisons are made with the supramolecular aggregation in isomeric amino-nitro derivatives, and in some N-methylnitroanilines.
 O hydrogen bond, and the molecules are linked into zigzag chains by a single C-H
O hydrogen bond, and the molecules are linked into zigzag chains by a single C-H O hydrogen bond. Comparisons are made with the supramolecular aggregation in isomeric amino-nitro derivatives, and in some N-methylnitroanilines.
O hydrogen bond. Comparisons are made with the supramolecular aggregation in isomeric amino-nitro derivatives, and in some N-methylnitroanilines.Molecules of the title compound, C10H20N2O8, adopt a conformation which is almost centrosymmetric. The molecules are disordered over two sets of sites with an occupancy ratio of 0.94:0.06. In the major form, there are two intramolecular O—H O hydrogen bonds [O
O hydrogen bonds [O O 2.756 (4) and 2.765 (4) Å; O—H
O 2.756 (4) and 2.765 (4) Å; O—H O 144 and 146°], in which the two amidic O atoms act as acceptors. In addition, there are four intermolecular O—H
O 144 and 146°], in which the two amidic O atoms act as acceptors. In addition, there are four intermolecular O—H O hydrogen bonds [O
O hydrogen bonds [O O 2.650 (3)–2.666 (3) Å; O—H
O 2.650 (3)–2.666 (3) Å; O—H O 158–171°]; these link each molecule to six others in a continuous sheet structure which contains five distinct ring motifs, two of the S(7) type, two of the R
O 158–171°]; these link each molecule to six others in a continuous sheet structure which contains five distinct ring motifs, two of the S(7) type, two of the R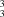 (10) type and one of the R
(10) type and one of the R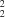 (22) type.
(22) type.
 O hydrogen bonds [O
O hydrogen bonds [O O 2.756 (4) and 2.765 (4) Å; O—H
O 2.756 (4) and 2.765 (4) Å; O—H O 144 and 146°], in which the two amidic O atoms act as acceptors. In addition, there are four intermolecular O—H
O 144 and 146°], in which the two amidic O atoms act as acceptors. In addition, there are four intermolecular O—H O hydrogen bonds [O
O hydrogen bonds [O O 2.650 (3)–2.666 (3) Å; O—H
O 2.650 (3)–2.666 (3) Å; O—H O 158–171°]; these link each molecule to six others in a continuous sheet structure which contains five distinct ring motifs, two of the S(7) type, two of the R
O 158–171°]; these link each molecule to six others in a continuous sheet structure which contains five distinct ring motifs, two of the S(7) type, two of the R
 journal menu
journal menu











