The title compound, 7-chloro-8-fluoro-4-phenyl-2,3,3a,4,5,9b-hexahydrofuro[3,2-c]quinoline, C17H15ClFNO, was obtained from the imine Diels-Alder reaction of an N-benzylideneaniline and 2,3-dihydrofuran catalyzed by ZrCl4. An isomer that crystallizes in the centrosymmetric monoclinic space group C2/c is presented. The furan ring is in a twist conformation, while the pyridine ring is in a sofa conformation. An intermolecular hydrogen bond between the pyridine NH group and the O atom of the furan ring in a neighboring molecule forms chains along the b axis.
The title compound, C15H24O5, was isolated from the flowers of Parthenium hysterophorus. The crystal structure determination confirms the relative stereochemistry unambiguously. The packing is stabilized by O—H O hydrogen bonding.
O hydrogen bonding.
 O hydrogen bonding.
O hydrogen bonding.The title compound, 4-(4-chlorophenyl)-2,3,3a,4,5,9b-hexahydrofuro[3,2-c]quinoline, C17H16ClNO, was obtained as one of the diastereoisomers from the imino Diels–Alder reaction of an imine and dihydrofuran. The molecule is a cis (endo) cycloadduct isomer. The tetrahydrofuran ring adopts an envelope conformation, while the N-heterocyclic ring is in a sofa conformation. Intermolecular N—H O hydrogen bonds lead to chains of molecules running along the a axis.
O hydrogen bonds lead to chains of molecules running along the a axis.
 O hydrogen bonds lead to chains of molecules running along the a axis.
O hydrogen bonds lead to chains of molecules running along the a axis.The piperidone ring of the title compound, C31H28N2O4, adopts a twist boat conformation. The crystal packing is characterized by a layered arrangement of molecules held together by C—H O hydrogen bonds, in which each of the nitroso and carbonyl O atoms participate. No significant aryl–aryl interactions are observed.
O hydrogen bonds, in which each of the nitroso and carbonyl O atoms participate. No significant aryl–aryl interactions are observed.
 O hydrogen bonds, in which each of the nitroso and carbonyl O atoms participate. No significant aryl–aryl interactions are observed.
O hydrogen bonds, in which each of the nitroso and carbonyl O atoms participate. No significant aryl–aryl interactions are observed. The structure of bharangin triacetate [systematic name: 6,10-bis(acetyloxy)-8-isopropyl-4,4,11a-trimethyl-2-oxo-2,3,4,6,11,11a-hexahydrobenzo[5,6]cyclohepta[1,2-b]pyran-9-yl acetate], C26H32O8, a tricyclic diterpenoid, contains a seven-membered cycloheptadiene ring forming two planar parts, a six-membered δ-lactone ring in a boat conformation and a planar benzene ring. The structure is stabilized by intra- and intermolecular C—H O interactions.
O interactions.
 O interactions.
O interactions. The title compound, 5-(4-nitrophenyl)-3,4,4a,5,6,10b-hexahydro-2H-pyrano[3,2-c]quinoline, C18H18N2O3, was obtained from the Diels–Alder reaction of N-benylideneaniline and dihydropyran, catalyzed by zirconium tetrachloride. This molecule is an exo-cycloadduct isomer. The pyran ring adopts a chair conformation, while the N-heterocyclic ring prefers a half-chair conformation. Molecules are associated into centrosymmetric dimers by N—H O hydrogen bonds.
O hydrogen bonds.
 O hydrogen bonds.
O hydrogen bonds. The title structure, 15,18-dimethyl-5-dioxo-14-oxapentacyclo[11.4.1.02,15.04,13.06,11]octadeca-3,6,8,10-tetraene-18-carbaldehyde, C20H18O4, is a novel naphthoquinone, isolated from Stereospermum personatum. In the bicyclic system, all three six-membered rings adopt a boat conformation. Two intermolecular C=O H—C interactions link the molecules into a chain.
H—C interactions link the molecules into a chain.
 H—C interactions link the molecules into a chain.
H—C interactions link the molecules into a chain. The crystal structure of scandenin [systematic name: 4-hydroxy-3-(4-hydroxyphenyl)-5-methoxy-8,8-dimethyl-6-(3-methyl-2-butenyl)-2H,8H-pyrano[2,3f]chromen-2-one], C26H26O6, has been determined. The compound crystallizes in the monoclinic space group P21/c with two independent but chemically identical molecules in the asymmetric unit. In both molecules, the coumarin moiety is planar and the angularly fused pyran ring is in a sofa conformation. The crystal structure is stabilized by intra- and intermolecular O—H O hydrogen bonds, with O
O hydrogen bonds, with O O distances in the range 2.601 (1)–2.699 (2) Å.
O distances in the range 2.601 (1)–2.699 (2) Å.
 O hydrogen bonds, with O
O hydrogen bonds, with O O distances in the range 2.601 (1)–2.699 (2) Å.
O distances in the range 2.601 (1)–2.699 (2) Å. A stereoisomer of bharangin triacetate [systematic name: 6,10-bis(acetyloxy)-8-isopropyl-4,4,11a-trimethyl-2-oxo-2,3,4,6,11,11a-hexahydrobenzo[5,6]cyclohepta[1,2-b]pyran-9-yl acetate], C26H32O8, a tricyclic diterpenoid, is reported. There is a 0.56 (3):0.44 (3) disorder in the terminal isopropyl side chain. The title compound and a previously reported stereoisomer, crystallizing in the same space group, exhibit very similar molecular conformations, but differ in the orientation of one of the acetate groups, resulting in a different configuration at the atom to which the acetate group is attached. Consequently, different C—H O interactions are observed.
O interactions are observed.
 O interactions are observed.
O interactions are observed. In the title compound, C15H11ClN2O, the dihedral angle between the pyridyl and phenyl rings is 38.7 (4)°. The molecules are linked via O—H N and C—H
N and C—H N interactions, forming infinite chains running along the b axis. The structure is further stabilized by weak C—H
N interactions, forming infinite chains running along the b axis. The structure is further stabilized by weak C—H π interactions.
π interactions.
 N and C—H
N and C—H N interactions, forming infinite chains running along the b axis. The structure is further stabilized by weak C—H
N interactions, forming infinite chains running along the b axis. The structure is further stabilized by weak C—H π interactions.
π interactions.In the title compound, C11H11ClN2O, molecules are linked via O—H N hydrogen bonds, forming infinite chains running along the c axis with a graph-set motif of C(6). The structure is further stabilized by weak π–π and C—H
N hydrogen bonds, forming infinite chains running along the c axis with a graph-set motif of C(6). The structure is further stabilized by weak π–π and C—H π interactions.
π interactions.
 N hydrogen bonds, forming infinite chains running along the c axis with a graph-set motif of C(6). The structure is further stabilized by weak π–π and C—H
N hydrogen bonds, forming infinite chains running along the c axis with a graph-set motif of C(6). The structure is further stabilized by weak π–π and C—H π interactions.
π interactions. The structure of the clerodane diterpene C21H30O5, with modest antibacterial activity and isolated from the plant Pulicaria wightiana, was established to be methyl 6α-hydroxy-3,13-clerodadien-15,16-olid-18-oate. The fused six-membered rings of the decalin system have sofa and chair conformations. The furanone side chain is in an antiperiplanar conformation. The molecules in the crystal structure are stabilized by an intramolecular O—H O hydrogen bond and a chain of intermolecular C—H
O hydrogen bond and a chain of intermolecular C—H O interactions.
O interactions.
 O hydrogen bond and a chain of intermolecular C—H
O hydrogen bond and a chain of intermolecular C—H O interactions.
O interactions.The title compound, 1-[2-hydroxy-4-methoxy-5-(3-methylbut-2-enyl)phenyl]-3-(4-hydroxyphenyl)prop-2-en-1-one (also known as bavachalcone), C21H22O4, is a polymorphic form of a previously reported structure [Bhalla et al. (1968). Tetrahedron Lett. 20, 2401–2406]. The molecules in both polymorphs exhibit nearly identical conformations. In each polymorph, strong intra- and intermolecular hydrogen-bonded chains, involving the hydroxyl groups, are formed. A weak C—H O interaction is seen in the present structure, but was not observed in the previously reported polymorph.
O interaction is seen in the present structure, but was not observed in the previously reported polymorph.
 O interaction is seen in the present structure, but was not observed in the previously reported polymorph.
O interaction is seen in the present structure, but was not observed in the previously reported polymorph. 6-Methyl-5-(N-methylcarbamoyl)-4-phenyl-1,2,3,4-tetrahydropyrimidine-2-thione dimethyl sulfoxide solvate, C13H15N3OS·C2H6OS, crystallizes with one molecule of each species in the asymmetric unit. The dihydropyridimine group adopts a flattened boat conformation. An N—H O hydrogen bond exists between the N atom of the carbamoyl side chain and the carbonyl O atom of the solvent molecule. In addition, intermolecular N—H
O hydrogen bond exists between the N atom of the carbamoyl side chain and the carbonyl O atom of the solvent molecule. In addition, intermolecular N—H O and N—H
O and N—H S hydrogen bonding is observed in the crystal structure.
S hydrogen bonding is observed in the crystal structure.
 O hydrogen bond exists between the N atom of the carbamoyl side chain and the carbonyl O atom of the solvent molecule. In addition, intermolecular N—H
O hydrogen bond exists between the N atom of the carbamoyl side chain and the carbonyl O atom of the solvent molecule. In addition, intermolecular N—H O and N—H
O and N—H S hydrogen bonding is observed in the crystal structure.
S hydrogen bonding is observed in the crystal structure.The title compound, C17H16N2O3, is one of the diastereoisomers formed as the result of the Diels–Alder reaction of 1-(4-nitrophenyl)methylidene]-N-phenylamine and dihydrofuran catalyzed by zirconium tetrachloride. The furan ring adopts an envelope conformation, while the N-heterocyclic ring prefers a sofa conformation. Molecules are linked into centrosymmetric dimers by N—H O hydrogen-bond interactions.
O hydrogen-bond interactions.
 O hydrogen-bond interactions.
O hydrogen-bond interactions.In the title compound, C28H23NO4, the pyrrolidine ring adopts a twist conformation and the pyran ring in the chromanone system adopts a half-chair conformation. The molecules associate via C—H O interactions to form R22(18) dimers.
O interactions to form R22(18) dimers.
 O interactions to form R22(18) dimers.
O interactions to form R22(18) dimers.In the title compound, C28H24N2O4S, the indole ring system is planar, but the pyrrolidine and thiazole rings adopt an envelope and a twist conformation, respectively. The indole system makes a dihedral angle of 55.7 (1)° with the methoxyphenyl ring. The molecular packing in the crystal structure is stabilized by C—H O and N—H
O and N—H O hydrogen bonds.
O hydrogen bonds.
 O and N—H
O and N—H O hydrogen bonds.
O hydrogen bonds.In the title compound, C26H24N2O3, the hydrogenated pyrrole ring adopts a twist conformation and the pyran ring adopts a twist–boat conformation. The molecular packing is stabilized by N—H O and C—H
O and C—H O intermolecular hydrogen bonds and also by weak intermolecular π–π interactions.
O intermolecular hydrogen bonds and also by weak intermolecular π–π interactions.
 O and C—H
O and C—H O intermolecular hydrogen bonds and also by weak intermolecular π–π interactions.
O intermolecular hydrogen bonds and also by weak intermolecular π–π interactions.In the title compound, C25H20O3, two of the benzene rings are almost perpendicular to each other, with a dihedral angle of 88.1 (1)°. The molecular packing is stabilized by intermolecular C—H O and π–π interactions, in addition to van der Waals forces.
O and π–π interactions, in addition to van der Waals forces.
 O and π–π interactions, in addition to van der Waals forces.
O and π–π interactions, in addition to van der Waals forces.In the structure of N-butyl-N-o-tolylbenzenesulfonamide, C17H21NO2S, the mean planes of the benzene rings are inclined to each other at 40.3 (1)°. The geometry around the S atom is considerably distorted from ideal tetrahedral geometry. Centrosymmetrically related pairs of molecules dimerize through C—H O hydrogen bonds. The crystal packing is stabilized by intermolecular C—H
O hydrogen bonds. The crystal packing is stabilized by intermolecular C—H O hydrogen bonding.
O hydrogen bonding.
 O hydrogen bonds. The crystal packing is stabilized by intermolecular C—H
O hydrogen bonds. The crystal packing is stabilized by intermolecular C—H O hydrogen bonding.
O hydrogen bonding.The title compound, 4-methoxy-12-methyl-12-azatetracyclo[9.31,10.02,7]heptadeca-2(7),3,5-triene, C18H25NO, has a T-shaped configuration, with the benzene ring (A) and the fused carbocyclic ring (C) forming the upright stock; the other carbocyclic ring (D) and the piperidine ring (E) are in the arm positions. Rings C, D and E have sofa, chair and chair conformations, respectively. In the crystal packing, the molecules are joined through C—H O hydrogen bonding into chains running along the c axis.
O hydrogen bonding into chains running along the c axis.
 O hydrogen bonding into chains running along the c axis.
O hydrogen bonding into chains running along the c axis.The title compound, C28H22ClNO3, has two crystallographically independent molecules in the asymmetric unit. The pyrrolidine ring (A) of the pyrrolizine system adopts an envelope conformation in both molecules, whereas the other five-membered ring (B) adopts an envelope conformation in one molecule and a twist conformation in the other. The molecular packing is stabilized by intermolecular C—H O, C—H
O, C—H π and π–π interactions.
π and π–π interactions.
 O, C—H
O, C—H π and π–π interactions.
π and π–π interactions.In the title compound, C30H25NO3S, the phenylsulfonyl and methoxyphenyl groups are almost perpendicular to the indole unit, whereas the other phenyl ring is inclined at an angle of 68.9 (1)° to it. The benzene ring of the phenylsulfonyl substituent makes a dihedral angle of 68.0 (1)° with the benzene ring of the methoxyphenyl substituent. The molecular packing in the crystal is stabilized by weak intra- and intermolecular C—H O interactions.
O interactions.
 O interactions.
O interactions.In the title compound, C29H25NO2S2, the benzene rings of the phenylsulfonyl and the dimethylbenzyl substituents are almost perpendicular to the indole unit, whereas the dihedral angle between the phenyl rings of the phenylsulfanyl and phenylsulfonyl groups is 71.2 (1)°. The molecules in the crystal structure are held together by van der Waals, C—H O and C—H
O and C—H S interactions.
S interactions.
 O and C—H
O and C—H S interactions.
S interactions.In the title compound, C30H24ClNO3S, the phenylsulfonyl, chlorophenyl and methoxyphenyl rings are each almost perpendicular to the indole ring system. The crystal packing shows centrosymmetrically related pairs of molecules. Intermolecular C—H O interactions are present.
O interactions are present.
 O interactions are present.
O interactions are present.In the title compound, C32H25NO4, the mean plane through the pyrrolizidine system is almost perpendicular [dihedral angle 88.9 (1)°] to that through one of the acenaphthene units, and the dihedral angle between the pyrrolizidine and the second acenaphthene system is 81.6 (1)°. In the pyrrolizidine system, one of the rings is in a half-chair conformation while the other is in an envelope conformation. The overall conformation is twisted about the fusion bond. The crystal structure is stabilized by a C—H O intermolecular hydrogen bond.
O intermolecular hydrogen bond.
 O intermolecular hydrogen bond.
O intermolecular hydrogen bond.The title compound, C34H32N2, is formed as a result of the Diels-Alder reaction of N-benzylideneaniline and cyclohexen-2-one, catalyzed by zirconium tetrachloride. The molecule exists as the Z isomer. All three rings in the azabicyclo[2.2.2]octane system adopt boat conformations. The crystal packing is stabilized by van der Waals forces.
In the title compound, C19H21BrN2O2S, the fused pyrrolidine rings adopt envelope conformations. The molecular packing is stabilized by weak intermolecular C—H O interactions and van der Waals forces.
O interactions and van der Waals forces.
 O interactions and van der Waals forces.
O interactions and van der Waals forces.In the crystal structure of the title compound {systematic name: 4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-hydroxy-1-[2-(2-methyl-4-oxo-3,4,6,7,8,9-hexahydro-2H-pyrido[1,2-a]pyrimidin-3-yl)ethyl]piperidine N-oxide hydrogen peroxide methanol solvate}, C23H27FN4O3·H2O2·CH3OH, the asymmetric unit contains one molecule of risperidone N-oxide, one methanol solvent molecule and one hydrogen peroxide molecule. The piperidine ring adopts a chair conformation, while the tetrahydropyridine ring has a sofa conformation. The hydrogen peroxide molecule links the risperidone molecules to form a chain. O—H O and C—H
O and C—H O interactions stabilize the crystal packing.
O interactions stabilize the crystal packing.
 O and C—H
O and C—H O interactions stabilize the crystal packing.
O interactions stabilize the crystal packing.The crystal structure of the title compound, C17H21N4S+·C6H4NO2−, [systematic name: 1-methyl-4-(2-methyl-10H-thieno[2,3-b][1,5]benzodiazepin-4-yl)hexahydropyrazin-1-ium nicotinate] is reported. The central seven-membered heterocycle is in a boat conformation, while the piperazine ring displays a chair conformation with its methyl group oriented equatorially. The coulombic interaction between olanzapinium and nicotinate ions is supplemented by intra- and intermolecular N—H O hydrogen bonds, forming infinite chains along the c axis.
O hydrogen bonds, forming infinite chains along the c axis.
 O hydrogen bonds, forming infinite chains along the c axis.
O hydrogen bonds, forming infinite chains along the c axis.Ezetimibe monohydrate or 1-(4-fluorophenyl)-3(R)-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4(S)-(4-hydroxyphenyl)azetidin-2-one monohydrate, C24H21F2NO3·H2O, belongs to a class of agents that inhibit cholesterol absorption in the intestine. The solvent water molecule present in the structure is involved in several hydrogen bonds.
4′-(4-Methoxyphenyl)-1′-methyl-9H-fluorene-9-spiro-3′-pyrrolidine-2′-spiro-2′′-indan-1′′,3′′-dione
In the title compound, C32H25NO3, the pyrrolidine ring and the five-membered ring of the indan moiety adopt twist and envelope conformations, respectively. The molecules in the crystal structure are linked by an intermolecular C—H O hydrogen bond, forming chains running along the [
O hydrogen bond, forming chains running along the [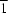 01] direction.
01] direction.
 O hydrogen bond, forming chains running along the [
O hydrogen bond, forming chains running along the [In the title compound, C24H17N3O, the dihedral angles between the planes of the two indole ring systems and the oxindole group are 77.7 (1) and 71.9 (1)°. The molecular packing in the crystal structure is stabilized by an intermolecular N—H O hydrogen bond.
O hydrogen bond.
 O hydrogen bond.
O hydrogen bond.The title compound, C27H22N2O3, comprises an indeno-quinoxaline system linked via a spiro ring junction to a hexahydro-1H-pyrrolizine unit. This in turn carries an indene-1,3-dione linked again by a spiro junction through the five-membered indenone ring. The pyrrolizidine moiety is folded and twisted about the N—C bond common to the two five-membered rings. In the crystal packing, an R22(20) graph set involves a dimeric C—H O hydrogen bond and ring motif. The packing is further stabilized by C—H
O hydrogen bond and ring motif. The packing is further stabilized by C—H O and weak π–π interactions.
O and weak π–π interactions.
 O hydrogen bond and ring motif. The packing is further stabilized by C—H
O hydrogen bond and ring motif. The packing is further stabilized by C—H O and weak π–π interactions.
O and weak π–π interactions.Quetiapine hemifumarate (systematic name: 1-[2-(2-hydroxyethoxy)ethyl]-4-(dibenzo[b,f][1,4]thiazepin-11-yl)piperazinium hemifumarate), C21H26O2N3S+·0.5C4H2O42−, a new dibenzothiazepine antipsychotic, has international approvals for the treatment of schizophrenia. In the tricyclic framework, the central thiazepine ring has a boat conformation and the dihedral angle between the planar benzene rings is 108.6 (1)°. The protonated piperazine ring exhibits a chair conformation with its ethoxyethanol side chain oriented equatorially. The fumarate anion possesses a centre of symmetry. The quetiapinium and fumarate ions are connected by O—H O and N—H
O and N—H O hydrogen bonds.
O hydrogen bonds.
 O and N—H
O and N—H O hydrogen bonds.
O hydrogen bonds.The title compound, C32H28N2, was obtained from the Diels-Alder reaction of N-benzylideneaniline and cyclohexen-2-one catalysed by zirconium tetrachloride. All three rings in the azabicyclo[2.2.2] ring system adopt boat conformations. The crystal packing is stabilized by van der Waals forces.
The structure of the title cycloadduct, C20H18N3O3Cl, has been determined. The inversion-related molecules are held together by N—H O hydrogen bonds, forming R22(8) rings. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.
O hydrogen bonds, forming R22(8) rings. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.
 O hydrogen bonds, forming R22(8) rings. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.
O hydrogen bonds, forming R22(8) rings. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.In the title cycloadduct, C20H19N3O3, the molecules form centrosymmetric dimers linked by N—H O hydrogen bonds, forming an R22(8) ring. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.
O hydrogen bonds, forming an R22(8) ring. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.
 O hydrogen bonds, forming an R22(8) ring. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.
O hydrogen bonds, forming an R22(8) ring. The overall conformation of the pyrrolizidine nucleus is folded about the bridging bond.In the title compound, C20H20O6, the biphenyl twist angle is 48.2 (1)°. All three ester groups are planar and they make dihedral angles of 63.6 (1), 56.8 (1) and 41.6 (1)° with the attached benzene ring. C—H O interactions contribute to both molecular and crystal stability.
O interactions contribute to both molecular and crystal stability.
 O interactions contribute to both molecular and crystal stability.
O interactions contribute to both molecular and crystal stability.In the title compound, C22H21NO5, the pyrrolidine ring and the five-membered ring in the indene group have envelope conformations, while the furan ring adopts a twist conformation. Weak intermolecular C—H O interactions link the molecules into centrosymmetric dimers. The crystal packing is further stabilized by van der Waals forces.
O interactions link the molecules into centrosymmetric dimers. The crystal packing is further stabilized by van der Waals forces.
 O interactions link the molecules into centrosymmetric dimers. The crystal packing is further stabilized by van der Waals forces.
O interactions link the molecules into centrosymmetric dimers. The crystal packing is further stabilized by van der Waals forces.In the title compound, C35H31N3O2, the dihedral angle between the two indole groups is 76.4 (1)°. The molecular structure is stabilized by an intramolecular π–π interaction. The molecular packing in the crystal structure is stabilized by strong N—H O hydrogen bonds and weak N—H
O hydrogen bonds and weak N—H π interactions.
π interactions.
 O hydrogen bonds and weak N—H
O hydrogen bonds and weak N—H π interactions.
π interactions.The pyrrolidine ring of the title compound, C34H32N4O2S, adopts an envelope conformation. The packing is stabilized by intermolecular C—H O and N—H
O and N—H N interactions.
N interactions.
 O and N—H
O and N—H N interactions.
N interactions.The molecule of the title compound, C24H21F2NO2, has a T-shaped form in the crystal structure. The central tetrahydropyran ring shows a chair conformation. All substituents are equatorially attached to this ring. The crystal packing is stabilized by N—H O, O—H
O, O—H O and C—H
O and C—H π(arene) interactions.
π(arene) interactions.
 O, O—H
O, O—H O and C—H
O and C—H π(arene) interactions.
π(arene) interactions.In the title compound, C16H17NO, the cyclohexanone ring adopts an envelope conformation.
In the title compound, C19H19N3O3, the oxindole residue and the 4-methylphenyl ring are almost perpendicular to the pyrrolidine ring. The pyrrolidine ring adopts an envelope conformation. The molecular packing is stabilized by N—H O hydrogen bonds and C—H
O hydrogen bonds and C—H O interactions, in addition to van der Waals forces.
O interactions, in addition to van der Waals forces.
 O hydrogen bonds and C—H
O hydrogen bonds and C—H O interactions, in addition to van der Waals forces.
O interactions, in addition to van der Waals forces.In the title compound, C34H31ClN2O3, the pyrrolidine ring adopts an envelope conformation. The molecular conformation is stabilized only by an intramolecular C—H O interaction.
O interaction.
 O interaction.
O interaction.In the title compound, C26H27NO4, the central ring of the xanthene moiety is planar and the two outer rings are in half-boat conformations. The molecular packing in the crystal structure is stabilized by N—H O and C—H
O and C—H O hydrogen bonds in addition to van der Waals forces.
O hydrogen bonds in addition to van der Waals forces.
 O and C—H
O and C—H O hydrogen bonds in addition to van der Waals forces.
O hydrogen bonds in addition to van der Waals forces.Lamotriginium benzoate dimethylformamide solvate [systematic name: 3,5-diamino-6-(2,3-dichlorophenyl)-1,2,4-triazin-2-ium benzoate dimethylformamide solvate], C9H8Cl2N5+·C7H5O2−·C3H7NO, is protonated on the triazine ring. The dihedral angle between the dichlorobenzene and triazine rings is 89.6 (1)°. The dimethylformamide solvent molecule is disordered over two sites. N—H O and N—H
O and N—H N hydrogen bonding stabilizes the crystal structure.
N hydrogen bonding stabilizes the crystal structure.
 O and N—H
O and N—H N hydrogen bonding stabilizes the crystal structure.
N hydrogen bonding stabilizes the crystal structure.In the title compound, C19H19Cl2N3O2, the piperidine ring adopts a distorted boat conformation. In the solid state, the molecules exist as O—H N-hydrogen-bonded centrosymmetric dimers.
N-hydrogen-bonded centrosymmetric dimers.
 N-hydrogen-bonded centrosymmetric dimers.
N-hydrogen-bonded centrosymmetric dimers.The title compound, C13H14N4O3S·H2O, is an isomer of 1,4-dihydro-6-methyl-5-(N-methylcarbamoyl)-4-(2-nitrophenyl)pyrimidine-2(3H)-thione. The nitro group is coplanar with the benzene ring to which it is attached. Intermolecular hydrogen bonds stabilize the crystal structure.
The title compound, C26H23NO, was obtained from the Diels-Alder reaction of N-benzylideneaniline and cyclohexen-2-one catalyzed by zirconium tetrachloride. The molecule is the Z isomer. All three six-membered rings in the 2-azabicyclo[2.2.2]octane system adopt the expected boat conformation. In the crystal packing, the phenyl groups are aggregated in hydrophobic layers at y = 0 and 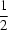 .
.
The benzimidazole ring system in the title molecule, C8H8N2S, is planar The imidazole ring does not exhibit a delocalized aromatic bond system. Molecules are linked by N—H N hydrogen bonds along the c axis. The crystal structure is further stabilized by C—H
N hydrogen bonds along the c axis. The crystal structure is further stabilized by C—H π interactions.
π interactions.
 N hydrogen bonds along the c axis. The crystal structure is further stabilized by C—H
N hydrogen bonds along the c axis. The crystal structure is further stabilized by C—H π interactions.
π interactions.In the crystal structure of the title compound, C23H22N2O3, the dihydropyrimidine (DHPM) ring adopts a sofa conformation. The methoxy-substituted benzene ring is oriented equatorially and the phenylethyl group has a fully extended conformation with respect to the DHPM ring. Four adjacent molecules are linked via N—H O and O—H
O and O—H O hydrogen-bonding interactions, generating a tetrameric unit. The crystal structure is further stabilized by C—H
O hydrogen-bonding interactions, generating a tetrameric unit. The crystal structure is further stabilized by C—H π(arene) interactions.
π(arene) interactions.
 O and O—H
O and O—H O hydrogen-bonding interactions, generating a tetrameric unit. The crystal structure is further stabilized by C—H
O hydrogen-bonding interactions, generating a tetrameric unit. The crystal structure is further stabilized by C—H π(arene) interactions.
π(arene) interactions.4′-(4-Bromobenzoyl)-1′-methyldispiro[indole-3(2H),2′-pyrrolidine-3′,3′′(2′′H)-indole]-2,2′′-dione
In the title compound, C26H20BrN3O3, the central pyrrolidine ring adopts an envelope conformation. In the crystal structure, the molecules exist as centrosymmetric N—H O hydrogen-bonded dimers. The dimers are linked via C—H
O hydrogen-bonded dimers. The dimers are linked via C—H O hydrogen bonds, forming a chain along the b axis.
O hydrogen bonds, forming a chain along the b axis.
 O hydrogen-bonded dimers. The dimers are linked via C—H
O hydrogen-bonded dimers. The dimers are linked via C—H O hydrogen bonds, forming a chain along the b axis.
O hydrogen bonds, forming a chain along the b axis.In the title compound, C19H19Cl2N3O2, the piperidine ring adopts a distorted boat conformation. In the solid state, the molecules exist as O—H N hydrogen-bonded centrosymmetric dimers.
N hydrogen-bonded centrosymmetric dimers.
 N hydrogen-bonded centrosymmetric dimers.
N hydrogen-bonded centrosymmetric dimers.In the title compound, C18H19N3O2·H2O, the piperidine ring adopts a distorted boat conformation. In the solid state, the molecules are linked by intermolecular O—H O and O—H
O and O—H N hydrogen bonds involving water molecules.
N hydrogen bonds involving water molecules.
 O and O—H
O and O—H N hydrogen bonds involving water molecules.
N hydrogen bonds involving water molecules.
 journal menu
journal menu


































































