The title molecular complex, [Zn(H2O)6](ClO4)2·2C6H8N2O2, consists of neutral 2,5-dimethylpyrazine 1,4-dioxide molecules, a hexaaquazinc(II) cation and perchlorate anions, linked through Coulombic attraction and hydrogen bonding. Half the neutral molecules lie in general positions, and the other half are on inversion centers.
In the title molecular complex, [Mn(NCS)2(H2O)4]·2C5H6N2O2, the manganese(II) atom has a slightly distorted octahedral coordination, formed by four O atoms from the water molecules and two N atoms from the thiocyanate anions. The Mn complrx lies on a special position of site symmetry 2 and 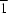 , and the 2-methylpyrazine-1,4-dioxide molecule is located on a mirror plane. The crystal packing is stabilized by intermolecular OH(water)
, and the 2-methylpyrazine-1,4-dioxide molecule is located on a mirror plane. The crystal packing is stabilized by intermolecular OH(water) O←N hydrogen bonds.
O←N hydrogen bonds.
 O←N hydrogen bonds.
O←N hydrogen bonds. The title compound, [Ni(NCS)2(C6H7NO)2(H2O)2]·H2O, consists of a mononuclear complex and an uncoordinated water molecule. The nickel(II) ion has a distorted octahedral coordination, formed by two N atoms from two thiocyanate anions and four O atoms from two water molecules and two 4-methylpyridine N-oxide molecules. The uncoordinated water molecules and the complex are connected by O—H O and O—H
O and O—H S hydrogen bonds.
S hydrogen bonds.
 O and O—H
O and O—H S hydrogen bonds.
S hydrogen bonds. In the title mononuclear complex, [Co(NCS)2(C5H5NO)2(H2O)2], cobalt(II) has a distorted octahedral coordination formed by two N atoms from two thiocyanate anions and four O atoms from two water molecules and two pyridine N-oxide molecules. The complexes are connected to each other by O—H O and O—H
O and O—H S hydrogen bonds.
S hydrogen bonds.
 O and O—H
O and O—H S hydrogen bonds.
S hydrogen bonds. The title structure, (C5H7N2)2[Co(NCS)4], comprises discrete monovalent 4-aminopyridinium cations and divalent tetrathiocyanatocobaltate(II) anions. The cations and anions are linked via N—H S hydrogen bonds [N
S hydrogen bonds [N S = 3.264 (4)–3.640 (6) Å] to form a three-dimensional framework.
S = 3.264 (4)–3.640 (6) Å] to form a three-dimensional framework.
 S hydrogen bonds [N
S hydrogen bonds [N S = 3.264 (4)–3.640 (6) Å] to form a three-dimensional framework.
S = 3.264 (4)–3.640 (6) Å] to form a three-dimensional framework. In the title complex, [Co(C5H4N2O3)6](ClO4)2, six 4-nitropyridine N-oxide (npo) ligands coordinate to the CoII atom via their N-oxide O atoms, resulting in an octahedral CoO6 grouping. The CoII ion occupies an inversion centre. A short contact of 2.841 (12) Å is observed between a pyridine N atom and a perchlorate O atom.
In the title mononuclear complex, [ZnBr2(C6H7NO2)2], the ZnII atom is coordinated by two Br and two O atoms in a distorted tetrahedral geometry. There are π–π stacking interactions between the 4-methoxypridine N-oxide units.
In the mononuclear title complex, [Co(C12H11N2O2)2(C2H6O)2], the six-coordinate CoII atom lies on an inversion centre and is coordinated by six O atoms, two from the ethanol molecules and the remaining four from two chelating 4-acetyl-3-methyl-1-phenyl-5-pyrazolonate monovalent anions. O—H N hydrogen bonds involving the ethanol O—H and the pyrazolonate N atoms stabilize the structure.
N hydrogen bonds involving the ethanol O—H and the pyrazolonate N atoms stabilize the structure.
 N hydrogen bonds involving the ethanol O—H and the pyrazolonate N atoms stabilize the structure.
N hydrogen bonds involving the ethanol O—H and the pyrazolonate N atoms stabilize the structure.
 journal menu
journal menu

















