In the title compound, [Zn2(C7H4O2S)2(C12H8N2)2], the Zn complex forms a centrosymmetric dimer with a Zn Zn separation of 3.580 (1) Å. Each Zn atom is coordinated by two N atoms from the 1,10-phenanthroline (phen) ligand and by three O atoms and one S atom from the thiosalicylate ligands in a highly distorted octahedral geometry, with Zn—N distances of 2.119 (2) and 2.166 (2) Å, Zn—O distances in the range 2.161 (2)–2.329 (2) Å, and a Zn—S distance of 2.3034 (12) Å. There are π–π stacking interactions between the phen rings, forming a stacking arrangement along the c axis in the crystal structure.
Zn separation of 3.580 (1) Å. Each Zn atom is coordinated by two N atoms from the 1,10-phenanthroline (phen) ligand and by three O atoms and one S atom from the thiosalicylate ligands in a highly distorted octahedral geometry, with Zn—N distances of 2.119 (2) and 2.166 (2) Å, Zn—O distances in the range 2.161 (2)–2.329 (2) Å, and a Zn—S distance of 2.3034 (12) Å. There are π–π stacking interactions between the phen rings, forming a stacking arrangement along the c axis in the crystal structure.
 Zn separation of 3.580 (1) Å. Each Zn atom is coordinated by two N atoms from the 1,10-phenanthroline (phen) ligand and by three O atoms and one S atom from the thiosalicylate ligands in a highly distorted octahedral geometry, with Zn—N distances of 2.119 (2) and 2.166 (2) Å, Zn—O distances in the range 2.161 (2)–2.329 (2) Å, and a Zn—S distance of 2.3034 (12) Å. There are π–π stacking interactions between the phen rings, forming a stacking arrangement along the c axis in the crystal structure.
Zn separation of 3.580 (1) Å. Each Zn atom is coordinated by two N atoms from the 1,10-phenanthroline (phen) ligand and by three O atoms and one S atom from the thiosalicylate ligands in a highly distorted octahedral geometry, with Zn—N distances of 2.119 (2) and 2.166 (2) Å, Zn—O distances in the range 2.161 (2)–2.329 (2) Å, and a Zn—S distance of 2.3034 (12) Å. There are π–π stacking interactions between the phen rings, forming a stacking arrangement along the c axis in the crystal structure.In the title molecule, C14H10O4S, all bond lengths and angles are normal. In the crystal structure, strong intermolecular O—H O hydrogen bonds between the carboxyl groups link the molecules into zigzag chains extending along the c axis.
O hydrogen bonds between the carboxyl groups link the molecules into zigzag chains extending along the c axis.
 O hydrogen bonds between the carboxyl groups link the molecules into zigzag chains extending along the c axis.
O hydrogen bonds between the carboxyl groups link the molecules into zigzag chains extending along the c axis.The title compound, C13H14N2·C8H6O4, was obtained by the hydrothermal reaction of CuCl with 1,2,4,5-benzenetetracarboxylic acid and 1,3-bis(4-pyridyl)propane. The 1,3-bis(4-pyridyl)propane molecule lies on a crystallographic twofold axis, and terephthalic acid is on an inversion centre. In the crystal structure, strong intermolecular O—H N hydrogen bonds between the components lead to the formation of zigzag chains extending along [10
N hydrogen bonds between the components lead to the formation of zigzag chains extending along [10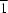 ]. The chains are linked through intermolecular C—H
]. The chains are linked through intermolecular C—H O hydrogen bonds.
O hydrogen bonds.
 N hydrogen bonds between the components lead to the formation of zigzag chains extending along [10
N hydrogen bonds between the components lead to the formation of zigzag chains extending along [10 O hydrogen bonds.
O hydrogen bonds.The title compound, C10H8N2·2C7H5NO4, was obtained as a by-product of the hydrothermal reaction of ZnSO4 with 2-nitrobenzoic acid and 4,4′-bipyridine. The 4,4′-bipyridine molecule lies on an inversion centre. There are O—H N and C—H
N and C—H O hydrogen bonds between the components in the crystal structure.
O hydrogen bonds between the components in the crystal structure.
 N and C—H
N and C—H O hydrogen bonds between the components in the crystal structure.
O hydrogen bonds between the components in the crystal structure.
 journal menu
journal menu













