The title compound, [Ag2(C7H4NO4)2(C4H10N2)]·H2O, has two similar dinuclear AgI complex molecules, each with a crystallographic inversion center; the asymmetric unit contains half of each molecule and a molecule of water. Each Ag atom in the compound is coordinated by one O atom from a 3-nitrobenzoate anion and one N atom from a piperidine bridge, in a slightly distorted linear geometry.
The title compound, [Cu(C14H12NO)2], is a mononuclear copper(II) compound. The CuII atom, lying on a centre of symmetry, is four-coordinated by two N atoms and two O atoms from two Schiff base ligands, giving a square-planar geometry.
The molecule of the title compound, C14H13NO, is roughly planar and displays a trans configuration with respect to the C=N double bond.
In both of the two independent molecules of the title compound, [Cu(C9H7NO3)(C5H5N)(H2O)], the copper atom is in a square-pyramidal coordination with the apical site occupied by water. The two molecules are related by a non-crystallographic center of inversion. Hydrogen bonds link symmetry-equivalent molecules into zigzag chains that run along the a axis of the orthorhombic unit cell.
The AgI atom in the title compound, [Ag(C5H6N2)2]2(C8H4O4)·10H2O, shows linear coordination. The cation and centrosymmetric anion interact indirectly via the non-coordinated water molecules, to furnish a tightly held three-dimensional structure.
The molecule of the title compound, C6H14N4O4, possesses a crystallographically imposed centre of symmetry. In the crystal packing, the molecules are linked via weak intermolecular C—H O hydrogen bonds into a sheet-like structure parallel to the (10
O hydrogen bonds into a sheet-like structure parallel to the (10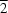 ) plane.
) plane.
 O hydrogen bonds into a sheet-like structure parallel to the (10
O hydrogen bonds into a sheet-like structure parallel to the (10
 journal menu
journal menu















