In the title compound, [Cu3(C10H9N3)3(C7H3O6S)2]·3H2O, [Cu3(ssal)2(dpa)3] units (dpa is 2,2′-dipyridylamine and ssal is 5-sulfonatosalicylate) are found, which consist of one crystallographically independent ssal anion, and two each of crystallographically independent dpa ligands and Cu atoms, the unit lying on a twofold rotation axis. One Cu atom is four-coordinated by one dpa ligand and one ssal anion within a square-planar geometry, whereas the second Cu atom is coordinated by one dpa ligand and two ssal anions which are related by symmetry. The first Cu atom has one longer contact to one O atom of a symmetry-related sulfonate group and is, therefore, five-coordinated within a distorted square-pyramidal coordination. The [Cu3(ssal)2(dpa)3] units are connected via the sulfonate groups into layers which are parallel to the ab plane. These layers are further connected by O—H O and N—H
O and N—H O hydrogen bonding via the water molecules.
O hydrogen bonding via the water molecules.
 O and N—H
O and N—H O hydrogen bonding via the water molecules.
O hydrogen bonding via the water molecules.The title compound, C12H9N2+·C7H5O6S-, consists of a 1,10-phenanthrolin-1-ium cation and a 5-sulfosalicylate anion. Hydrogen bonds link the anions into a one-dimensional chain. Furthermore, hydrogen bonds between the NH groups of 1,10-phenanthrolin-1-ium cations and the sulfonate O atoms attach the cations to the one-dimensional chain.
The Mn atom in the title compound, [Mn(C7H4O6S)(C10H8N2)2(H2O)]·2H2O, is coordinated by the two N-heterocycles, a water molecule and a 3-carboxylato-4-hydroxybenzenesulfonate dianion through the carboxylate group, thus imposing an octahedral environment on the metal atom. The coordinated and uncoordinated water molecules, the carboxylate and sulfonate groups interact via hydrogen bonds, producing a three-dimensional network architecture.
The chains of the title compound, [Co(C7H4O6S)(C10H8N2)(H2O)2]n, adopt a zigzag conformation as they propagate via glide planes. The octahedral coordination of the Co atom comprises the O atoms of two water molecules, the N atoms of the heterocycle, the carboxyl O atom of the dianion and the sulfonyl O atom of a symmetry-related dianion. The water molecules form hydrogen bonds within the chain only.
The title complex, [Cu(C7H4O6S)(C10H8N2)(H2O)2]n, consists of a polymeric neutral chain involving the 3-carboxylato-4-hydroxybenzenesulfonate ligand. The Cu atom shows a distorted octahedral coordination geometry, defined by two N atoms of the bipyridine, two O atoms of water molecules and the carboxyl O atom as well as one sulfonyl O atom of a symmetry-related bridging ligand. H atoms of water molecules are involved in O—H O hydrogen bonding, building a three dimensional network.
O hydrogen bonding, building a three dimensional network.
 O hydrogen bonding, building a three dimensional network.
O hydrogen bonding, building a three dimensional network. The cation interacts with the anion in the title compound, [Mn(C12H8N2)2(H2O)2](C7H4O6S)·4H2O, through hydrogen bonding with the coordinated water molecules in a hydrogen-bonded sheet structure. The Mn atom is in an octahedral environment in which the two water molecules occupy cis positions.
In the title polymeric compound, [Zn(C7H4O6S)(C10H8N2)(H2O)2], the octahedral coordination of the Zn atom comprises N-atom donors of 2,2′-bipyridine, O atoms of two water molecules, the carboxyl O atom of the 5-sulfosalicylate dianion and the sulfonyl O atom of a symmetry-related dianion. The water molecules with sulfonyl O atoms form hydrogen bonds between chains, giving rise to a two-dimensional network.
The title complex, [Mn(C7H4O6S)(C12H8N2)(H2O)2]n, comprises a one-dimensional chain in which each Mn atom displays a distorted octahedral geometry. Hydrogen-bonding interactions between chains generate a two-dimensional architecture.
In the title one-dimensional chain complex, [Co(C7H4O6S)(C12H8N2)(H2O)2]n, each CoII atom displays an octahedral geometry. Hydrogen-bonding interactions between chains generate a two-dimensional architecture.
The title complex, {(C10H10N2)[Mn(SO4)2(C10H8N2)(H2O)2]·2H2O}n, consists of an anionic one-dimensional chain, diprotonated 4,4′-bipyridine cations and uncoordinated water molecules. In the anionic chain, the Mn atom lies on a center of inversion in an octahedral geometry. Hydrogen bonds link the chains into a three-dimensional architecture.
In the title complex, [Ni(C12H8N2)2(H2O)2](C7H4O6S)·4H2O, the cation interacts with the anion and uncoordinated water molecules through hydrogen bonding with the coordinated water molecules, giving rise to a hydrogen-bonded sheet architecture. In the cation, the Ni atom exists in an octahedral geometry with two cis-arranged water ligands.
The crystal structure of the title compound, {[Cu3(C7H3O6S)2(C7H6N2)6]·8H2O}n, is built of trinuclear complexes [Cu3(bz)6(ssal)2] (bz = benzimidazole and ssal = 5-sulfonatosalicylate trianion) and uncoordinated water molecules. The molecule of the complex occupies a special position on a crystallographic inversion centre. The Cu atom located on the inversion centre has a square planar coordination made up of the two N atoms of monodentate benzimidazole ligands and the O atoms of two bridging salicylates. The other crystallographically independent Cu atom has a square pyramidal environment formed by the N atoms of two more monodentate benzimidazole ligands, two chelate O atoms of the bridging salicylate and the sulfonate O atom of the sulfonatosalicylate ligand belonging to the neighbouring trimetallic complex. The latter coordination links the molecules of the complex into infinite chains running along the a axis. These chains are further connected by O—H O and N—H
O and N—H O hydrogen bonding into a three-dimensional network.
O hydrogen bonding into a three-dimensional network.
 O and N—H
O and N—H O hydrogen bonding into a three-dimensional network.
O hydrogen bonding into a three-dimensional network.The title complex, {[Cu(C7H3O6S)(H2O)](C5H7N2)}n, consists of a polymeric anionic chain and protonated 2-aminopyridine. In the chain, each CuII atom has a distorted octahedral environment. The fully deprotonated 5-sulfonatosalicylate ligand acts as a μ4-bridging link, resulting in a dinuclear Cu2O6 motif. N—H O hydrogen bonds between cations and anions lead to a two-dimensional architecture.
O hydrogen bonds between cations and anions lead to a two-dimensional architecture.
 O hydrogen bonds between cations and anions lead to a two-dimensional architecture.
O hydrogen bonds between cations and anions lead to a two-dimensional architecture.The structure of the title complex is built up from a [Cu(C8H4F3O2S)(C12H8N2)(H2O)]+ cation and a [Cu(C8H4F3O2S)(C7H4O6S)(C12H8N2)]- anion. Both the cation and anion contain CuII atoms having a square-pyramidal geometry. Hydrogen bonds between cations and anions generate a one-dimensional chain and stabilize the crystal packing.
The structure of the title compound, {[Cu3(C7H3O6S)2(C12H8N2)2(H2O)2]·4H2O}n, consists of centrosymmetric trinuclear [Cu3(ssal)2(phen)2(H2O)2] (phen is 1,10-phenanthroline and ssal is the 5-sulfonatosalicylate trianion) building units and non-coordinated water molecules. The central (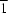 site symmetry) Cu atom is octahedrally coordinated and the other unique Cu atom adopts a square-pyramidal geometry. The μ3-ssal ligands link trinuclear units into a chain running along the a axis via bridging sulfonate groups. The chains are connected by O—H
site symmetry) Cu atom is octahedrally coordinated and the other unique Cu atom adopts a square-pyramidal geometry. The μ3-ssal ligands link trinuclear units into a chain running along the a axis via bridging sulfonate groups. The chains are connected by O—H O bonds into a three-dimensional network.
O bonds into a three-dimensional network.
 O bonds into a three-dimensional network.
O bonds into a three-dimensional network.
 journal menu
journal menu
























