The title compound [systematic name: 4,4',4'',4'''-methanetetrayltetracarboxylic acid-dimethyl sulfoxide-toluene (1/4/1)], C29H20O8, crystallizes from dimethyl sulfoxide (DMSO)/toluene to yield a structure in which the four carboxylic acid groups of each molecule donate a single hydrogen bond to the O atom of four molecules of DMSO. Packing of the resulting aggregates, which have crystallographic twofold symmetry, leaves space for the inclusion of partially disordered molecules of toluene, leading to the formation of crystals of overall composition tetrakis(4-carboxyphenyl)methane-dimethyl sulfoxide-toluene (1/4/1).
The title compound {alternatively called tetrakis[(4-bromobenzyloxy)methyl]methane}, C33H32Br4O4, was crystallized from ethyl acetate. The molecule has crystallographic twofold rotation symmetry. Key features of the structure include intramolecular edge-to-face aromatic interactions, intermolecular Br π interactions and intermolecular Br
π interactions and intermolecular Br O contacts.
O contacts.
 π interactions and intermolecular Br
π interactions and intermolecular Br O contacts.
O contacts.The title compound, C32H36O8Si, crystallizes from CH2Cl2 by slow evaporation to produce a close-packed structure. In this structure, the molecules have crystallographic 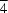 symmetry and no guest molecules are included. In contrast, crystallization of the closely related tetrakis(3,5-dihydroxyphenyl)silane from a range of solvents is directed by phenolic hydrogen bonding to yield an open diamondoid network, which is filled by a combination of interpenetration and inclusion of guests.
symmetry and no guest molecules are included. In contrast, crystallization of the closely related tetrakis(3,5-dihydroxyphenyl)silane from a range of solvents is directed by phenolic hydrogen bonding to yield an open diamondoid network, which is filled by a combination of interpenetration and inclusion of guests.
The title compound, C24H24N4O4Si, crystallizes from propionic acid-hexane to produce a close-packed structure with no included guests. In contrast, crystallization of the closely related tetrakis(1,2-dihydro-2-oxo-5-pyridyl)silane under similar conditions is directed by hydrogen bonding of the pyridinone groups to yield an open diamondoid network, which is filled by a combination of interpenetration and inclusion of guests.

 journal menu
journal menu













