Open  access
access
 access
accessThe title synthetic analog of purine nucleosides, C16H16Cl2N4O7, has its acetylated β-furanose ring in a 3′β-envelope conformation, with the corresponding C atom deviating by 0.602 (5) Å from the rest of the ring. The planar part of the furanose ring forms a dihedral angle of 65.0 (1)° with the mean plane of the purine bicycle. In the crystal, molecules form a three-dimensional network through multiple C—H O and C—H
O and C—H N hydrogen bonds and C—H
N hydrogen bonds and C—H π interactions.
π interactions.
 O and C—H
O and C—H N hydrogen bonds and C—H
N hydrogen bonds and C—H π interactions.
π interactions.Open  access
access
 access
accessThe asymmetric unit of the title compound contains two independent molecules with similar conformations, the cyclohexene rings adopting the same envelope conformation. In the crystal, adjacent molecules are connected via N—H O hydrogen bonds and weak C—H
O hydrogen bonds and weak C—H O interactions, forming supramolecular layers parallel to (
O interactions, forming supramolecular layers parallel to (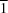 01).
01).
 O hydrogen bonds and weak C—H
O hydrogen bonds and weak C—H O interactions, forming supramolecular layers parallel to (
O interactions, forming supramolecular layers parallel to (Open  access
access
 access
accessThe title compound, C44H62O6S2 {systematic name: (1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bR)-5a,5b,8,8,11a-pentamethyl-1-(prop-1-en-2-yl)-3a-[(tosyloxy)methyl]icosahydro-1H-cyclopenta[a]chrysen-9-yl 4-methylbenzenesulfonate}, was obtained by tosylation of naturally occurring betulin. All the cyclohexane rings adopt chair conformations and the cyclopentane ring adopts a twisted envelope conformation, with the C atom bearing the tosylmethyl substituent forming the flap. In the crystal, molecules form a three-dimensional network through multiple weak C—H O hydrogen bonds.
O hydrogen bonds.
 O hydrogen bonds.
O hydrogen bonds.
 journal menu
journal menu












