The asymmetric unit of the title compound, {[La(C4H5O2)3(H2O)2]·C5H5N5·H2O}n, consists of an LaIII cation, three crotonate (but-2-enoate) anions and two coordinated water molecules forming the neutral complex, completed by an external adenine molecule and one hydration water molecule. The LaO10 coordination polyhedra, connected through the sharing of a single edge, form isolated chains running along the [100] direction. These one-dimensional structures are characterized by two different centrosymmetric La2O2 loops, with La La distances of 4.5394 (6) and 4.5036 (6) Å. The unbound adenine and water solvent molecules form a highly planar hydrogen-bonded array parallel to (110) (r.m.s. deviation from the mean plane < 0.10 Å) which intersects the isolated La–crotonate chains in a slanted fashion to form an extremely connected hydrogen-bonded three-dimensional structure.
La distances of 4.5394 (6) and 4.5036 (6) Å. The unbound adenine and water solvent molecules form a highly planar hydrogen-bonded array parallel to (110) (r.m.s. deviation from the mean plane < 0.10 Å) which intersects the isolated La–crotonate chains in a slanted fashion to form an extremely connected hydrogen-bonded three-dimensional structure.
 La distances of 4.5394 (6) and 4.5036 (6) Å. The unbound adenine and water solvent molecules form a highly planar hydrogen-bonded array parallel to (110) (r.m.s. deviation from the mean plane < 0.10 Å) which intersects the isolated La–crotonate chains in a slanted fashion to form an extremely connected hydrogen-bonded three-dimensional structure.
La distances of 4.5394 (6) and 4.5036 (6) Å. The unbound adenine and water solvent molecules form a highly planar hydrogen-bonded array parallel to (110) (r.m.s. deviation from the mean plane < 0.10 Å) which intersects the isolated La–crotonate chains in a slanted fashion to form an extremely connected hydrogen-bonded three-dimensional structure.The asymmetric unit of the title dimeric compounds, [Ln2(C4H5O2)6(H2O)4]·C5H5N5·7H2O, with Ln = Dy, (I), and Sm, (II), consists of an LnIII cation, three crotonate (but-2-enoate) anions and two coordinated water molecules forming the neutral complex, cocrystallized with half of an external adenine molecule and 3.5 water molecules. The metal complex has crystallographic inversion symmetry. The LnO9 coordination polyhedra are connected through the sharing of a single edge to form isolated dimeric units, with Ln Ln separations of 4.1766 (12) Å for (I) and 4.2340 (12) Å for (II). The unbound adenine molecule and one of the solvent water molecules are disordered around an inversion centre into two overlapping, equally populated, units. The structure is sustained by a complex hydrogen-bonding scheme involving all possible O-H and N-H groups as donors, and crotonate and water O and adenine N atoms as acceptors. The system is compared with recently published related compounds.
Ln separations of 4.1766 (12) Å for (I) and 4.2340 (12) Å for (II). The unbound adenine molecule and one of the solvent water molecules are disordered around an inversion centre into two overlapping, equally populated, units. The structure is sustained by a complex hydrogen-bonding scheme involving all possible O-H and N-H groups as donors, and crotonate and water O and adenine N atoms as acceptors. The system is compared with recently published related compounds.
 Ln separations of 4.1766 (12) Å for (I) and 4.2340 (12) Å for (II). The unbound adenine molecule and one of the solvent water molecules are disordered around an inversion centre into two overlapping, equally populated, units. The structure is sustained by a complex hydrogen-bonding scheme involving all possible O-H and N-H groups as donors, and crotonate and water O and adenine N atoms as acceptors. The system is compared with recently published related compounds.
Ln separations of 4.1766 (12) Å for (I) and 4.2340 (12) Å for (II). The unbound adenine molecule and one of the solvent water molecules are disordered around an inversion centre into two overlapping, equally populated, units. The structure is sustained by a complex hydrogen-bonding scheme involving all possible O-H and N-H groups as donors, and crotonate and water O and adenine N atoms as acceptors. The system is compared with recently published related compounds.The title ionic compound, (C7H8N3)2[Ho2(C4H5O2)8], is constructed from two almost identical independent centrosymmetric anionic dimers balanced by two independent 2-amino-1H-benzimidazol-3-ium (Habim+) cations. The asymmetric part of each dimer is made up of one HoIII cation and four crotonate (crot or but-2-enoate) anions, two of them acting in a simple η2-chelating mode and the remaining two acting in two different μ2:η2 fashions, viz. purely bridging and bridging–chelating. Symmetry-related HoIII cations are linked by two Ho—O—Ho and two Ho—O—C—O—Ho bridges which lead to rather short intracationic Ho Ho distances [3.8418 (3) and 3.8246 (3) Å]. In addition to the obvious Coulombic interactions linking the cations and anions, the isolated [Ho2(crot)8]2− and Habim+ ions are linked by a number of N—H
Ho distances [3.8418 (3) and 3.8246 (3) Å]. In addition to the obvious Coulombic interactions linking the cations and anions, the isolated [Ho2(crot)8]2− and Habim+ ions are linked by a number of N—H O hydrogen bonds, in which all N—H groups of the cation are involved as donors and all (simple chelating) crot O atoms are involved as acceptors. These interactions result in compact two-dimensional structures parallel to (110), which are linked to each other by weaker π–π contacts between Habim+ benzene groups.
O hydrogen bonds, in which all N—H groups of the cation are involved as donors and all (simple chelating) crot O atoms are involved as acceptors. These interactions result in compact two-dimensional structures parallel to (110), which are linked to each other by weaker π–π contacts between Habim+ benzene groups.
 Ho distances [3.8418 (3) and 3.8246 (3) Å]. In addition to the obvious Coulombic interactions linking the cations and anions, the isolated [Ho2(crot)8]2− and Habim+ ions are linked by a number of N—H
Ho distances [3.8418 (3) and 3.8246 (3) Å]. In addition to the obvious Coulombic interactions linking the cations and anions, the isolated [Ho2(crot)8]2− and Habim+ ions are linked by a number of N—H O hydrogen bonds, in which all N—H groups of the cation are involved as donors and all (simple chelating) crot O atoms are involved as acceptors. These interactions result in compact two-dimensional structures parallel to (110), which are linked to each other by weaker π–π contacts between Habim+ benzene groups.
O hydrogen bonds, in which all N—H groups of the cation are involved as donors and all (simple chelating) crot O atoms are involved as acceptors. These interactions result in compact two-dimensional structures parallel to (110), which are linked to each other by weaker π–π contacts between Habim+ benzene groups.The title compound, C9H8N2, presents two almost identical independent molecules in the asymmetric unit, both of them exhibiting an extremely planar isoquinoline core (maximum r.m.s. deviation = 0.014 Å). The most significant deviation is found in the -NH2 groups, which present a noticeable pyramidalization around the N atom, a feature also present in related structures containing the molecule as a ligand. The supramolecular structure is based on pairs of parallel hydrogen-bonded chains formed by just one molecular type each, defined by the strongest hydrogen bonds in the structure, which are of the N-H N type. These parallel chains are linked into pairs (or strips) via weaker C-H
N type. These parallel chains are linked into pairs (or strips) via weaker C-H N hydrogen bonds. Related strips generated by the c-glide plane define two families running along [
N hydrogen bonds. Related strips generated by the c-glide plane define two families running along [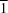 10] and [110], giving rise to an interesting system of interwoven chains stabilized by a number of weaker contacts of the C-H
10] and [110], giving rise to an interesting system of interwoven chains stabilized by a number of weaker contacts of the C-H
![[pi]](/logos/entities/pi_rmgif.gif) type.
type.
 N type. These parallel chains are linked into pairs (or strips) via weaker C-H
N type. These parallel chains are linked into pairs (or strips) via weaker C-H N hydrogen bonds. Related strips generated by the c-glide plane define two families running along [
N hydrogen bonds. Related strips generated by the c-glide plane define two families running along [
![[pi]](/logos/entities/pi_rmgif.gif) type.
type.
 journal menu
journal menu







![[mu]](/logos/entities/mu_rmgif.gif) -but-2-enoato-bis[diaquabis(but-2-enoato)dysprosium(III)] adenine monosolvate heptahydrate and the samarium(II) analogue
-but-2-enoato-bis[diaquabis(but-2-enoato)dysprosium(III)] adenine monosolvate heptahydrate and the samarium(II) analogue




