Open  access
access
 access
accessIn the title complex, [Mn2(C15H13N2O3)3]ClO4·CH3OH, the two MnII ions are bridged by two phenolate O atoms from two ligands, forming an Mn2O2 quadrangle. Each MnII ion has a distorted octahedral coordination geometry. One MnII ion is coordinated by two N atoms and four O atoms from two ligands, and the other is coordinated by one N atom and five O atoms from three ligands. A dimer is formed by intermolecular N—H O hydrogen bonds. The dimers, perchlorate anions and methanol solvent molecules are further connected into a chain along [
O hydrogen bonds. The dimers, perchlorate anions and methanol solvent molecules are further connected into a chain along [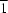 01] through N—H
01] through N—H O and O—H
O and O—H O hydrogen bonds.
O hydrogen bonds.
 O hydrogen bonds. The dimers, perchlorate anions and methanol solvent molecules are further connected into a chain along [
O hydrogen bonds. The dimers, perchlorate anions and methanol solvent molecules are further connected into a chain along [ O and O—H
O and O—H O hydrogen bonds.
O hydrogen bonds.
 journal menu
journal menu










