In the title complex, [Hg(NO3)2(C10H15N3OS)2], the HgII ion (site symmetry 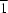 ) adopts a grossly distorted octahedral geometry by chelation of two neutral Him-thz ligands [Him-thz is 2-(2′-thiazole)-4,4,5,5-tetramethyldihydro-1H-imidazolyl-1-hydroxy] through their thiazole N atoms [Hg—N = 2.846 (2) Å] and reduced imino nitroxide N atoms [Hg—N = 2.055 (2) Å], leading to a five-membered ring, together with two O atoms from two nitrate anions [Hg—O = 2.859 (3) Å]. The molecules form one-dimensional chains by way of intermolecular O—H
) adopts a grossly distorted octahedral geometry by chelation of two neutral Him-thz ligands [Him-thz is 2-(2′-thiazole)-4,4,5,5-tetramethyldihydro-1H-imidazolyl-1-hydroxy] through their thiazole N atoms [Hg—N = 2.846 (2) Å] and reduced imino nitroxide N atoms [Hg—N = 2.055 (2) Å], leading to a five-membered ring, together with two O atoms from two nitrate anions [Hg—O = 2.859 (3) Å]. The molecules form one-dimensional chains by way of intermolecular O—H O hydrogen bonds.
O hydrogen bonds.
 O hydrogen bonds.
O hydrogen bonds.The title crystal structure, [Co(NO3)(C10H15N3OS)2]NO3, consists of CoII complex cations and NO3− anions, located on different twofold axes and linked to each other via O—H O hydrogen bonding. The CoII complex assumes a distorted octahedral coordination geometry, formed by a chelating nitrate and two ImTh molecules (ImTh is 1-hydroxy-4,4,5,5-tetramethyl-2-(1,3-thiazol-2-yl)-4,5-dihydro-1H-imidazole). Within the ImTh molecule, the N—O bond distance of 1.386 (2) Å is much longer than the N—O bonds found in related complexes with the nitroxide radical.
O hydrogen bonding. The CoII complex assumes a distorted octahedral coordination geometry, formed by a chelating nitrate and two ImTh molecules (ImTh is 1-hydroxy-4,4,5,5-tetramethyl-2-(1,3-thiazol-2-yl)-4,5-dihydro-1H-imidazole). Within the ImTh molecule, the N—O bond distance of 1.386 (2) Å is much longer than the N—O bonds found in related complexes with the nitroxide radical.
 O hydrogen bonding. The CoII complex assumes a distorted octahedral coordination geometry, formed by a chelating nitrate and two ImTh molecules (ImTh is 1-hydroxy-4,4,5,5-tetramethyl-2-(1,3-thiazol-2-yl)-4,5-dihydro-1H-imidazole). Within the ImTh molecule, the N—O bond distance of 1.386 (2) Å is much longer than the N—O bonds found in related complexes with the nitroxide radical.
O hydrogen bonding. The CoII complex assumes a distorted octahedral coordination geometry, formed by a chelating nitrate and two ImTh molecules (ImTh is 1-hydroxy-4,4,5,5-tetramethyl-2-(1,3-thiazol-2-yl)-4,5-dihydro-1H-imidazole). Within the ImTh molecule, the N—O bond distance of 1.386 (2) Å is much longer than the N—O bonds found in related complexes with the nitroxide radical.In the crystal structure of the title compound, C10H17N3O2S, the packing of the molecules is mainly a result of two types of O—H N hydrogen-bonding interactions, forming a two-dimensional network.
N hydrogen-bonding interactions, forming a two-dimensional network.
 N hydrogen-bonding interactions, forming a two-dimensional network.
N hydrogen-bonding interactions, forming a two-dimensional network.
 journal menu
journal menu












