The anionic component of the langbeinite-type title compound, ammonium oxonium dititanium(III,IV) tris(phosphate), is an infinite three-dimensional [Ti2(PO4)3]2− network based on PO4 tetrahedra corner-linked to TiO6 octahedra; the charge-balancing ammonium and oxonium cations occupy the cavities in the network and interact by way of N—H O and O—H
O and O—H O hydrogen bonds. The two independent Ti atoms, the ammonium N atom and the oxonium O atom lie on special positions of site symmetry 3.
O hydrogen bonds. The two independent Ti atoms, the ammonium N atom and the oxonium O atom lie on special positions of site symmetry 3.
 O and O—H
O and O—H O hydrogen bonds. The two independent Ti atoms, the ammonium N atom and the oxonium O atom lie on special positions of site symmetry 3.
O hydrogen bonds. The two independent Ti atoms, the ammonium N atom and the oxonium O atom lie on special positions of site symmetry 3.The reaction of ferric sulfate with ethylenediamine in the presence of acid under hydrothermal conditions results in the title compound, (C2H10N2)2[FeIII(OH)(SO4)2](SO4)·H2O. The six-coordinate ferrate dianion participates in a μ2-hydroxo-di-μ2-sulfate-bridged chain. The component species interact with each other by an extensive network of O—H O and N—H
O and N—H O hydrogen bonds. The two independent FeIII atoms lie on sites of
O hydrogen bonds. The two independent FeIII atoms lie on sites of 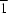 symmetry.
symmetry.
 O and N—H
O and N—H O hydrogen bonds. The two independent FeIII atoms lie on sites of
O hydrogen bonds. The two independent FeIII atoms lie on sites of The zirconium(IV) ion in the title compound, (C5H6N)4[Zr(SO4)4]·H2O, exists in a distorted dodecahedral geometry with four bidentate sulfate groups. The tetraanion, two of the pyridinium cations and water molecule all lie on special positions with m site symmetry. A network of N—H O and O—H
O and O—H O hydrogen bonds helps to stabilize the structure.
O hydrogen bonds helps to stabilize the structure.
 O and O—H
O and O—H O hydrogen bonds helps to stabilize the structure.
O hydrogen bonds helps to stabilize the structure.In the crystal structure of the title compound, (C10H10N2)[Fe(H2O)6](SO4)2·2H2O, hydrogen bonds link the 4,4'-bipyridinium cation, which lies on a special position of site symmetry 2, the hexaaquairon(II) cation, which lies on a center of symmetry, the sulfate anions and the uncoordinated water molecules into a three-dimensional network structure.
In the crystal structure of the polymeric title compound, {(C4H12N2)[Fe(OH)(SO4)2]·2H2O}n, the anion exists as a linear chain whose two independent FeIII atoms both occupy special positions of 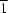 site symmetry. The two independent cations, which also lie on inversion sites, interact with the polyanionic chain and with the uncoordinated water molecules to form a tight-held three-dimensional network.
site symmetry. The two independent cations, which also lie on inversion sites, interact with the polyanionic chain and with the uncoordinated water molecules to form a tight-held three-dimensional network.

 journal menu
journal menu














