In the title compound, [Cu3(C7H3N2O7)2(C7H2N2O7)2(H2O)4]0.54[Cu(C7H3N2O7)2(H2O)2]0.92·4H2O, the CuII ion located on an inversion centre has a fractional site occupancy of 0.54. When this CuII ion is present, a trinuclear complex also involving two adjacent CuII ions arises; when it is not, then the result is two discrete mononuclear complexes. A very similar structure was reported previously by Valigura, Melnik, Koman, Martiska, Korabik, Mrozinski & Glowiak [Inorg. Chem. Commun. (2004), 7, 548-552]. However, in their material, arising from a different synthesis, the CuII ion with 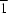 site symmetry is fully occupied, and thus only a trinuclear complex is formed. Here, the mononuclear CuII complex assumes a square-pyramidal coordination geometry, formed by two water molecules and two 3,5-dinitrosalicylate anions, one of which is monodentate and the other bidentate.
site symmetry is fully occupied, and thus only a trinuclear complex is formed. Here, the mononuclear CuII complex assumes a square-pyramidal coordination geometry, formed by two water molecules and two 3,5-dinitrosalicylate anions, one of which is monodentate and the other bidentate.
The title complex, [Ni(C4H4O4S)(C3H4N2)3]·H2O, displays a distorted octahedral NiN3O2S coordination geometry, formed by three imidazole (imid) ligands and one thiodiacetate dianion. The NiII atom is displaced from one of the imid planes, and the angle between the corresponding Ni—N bond and the imid plane is 21.94 (19)°. This spatial orientation is stabilized by an intermolecular C—H π interaction between one imid ligand and the C—H bond of an adjacent imid ligand.
π interaction between one imid ligand and the C—H bond of an adjacent imid ligand.
 π interaction between one imid ligand and the C—H bond of an adjacent imid ligand.
π interaction between one imid ligand and the C—H bond of an adjacent imid ligand.
 journal menu
journal menu











