The title complex, {[Cd(NCS)2(H2O)2]·2C6H9N3}n, crystalizes in the triclinic space group P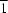 . The asymmetric unit contains one 2-amino-4,6-dimethylpyrimidine molecule, a thiocyanate ion, a water molecule and a cadmium ion (site symmetry
. The asymmetric unit contains one 2-amino-4,6-dimethylpyrimidine molecule, a thiocyanate ion, a water molecule and a cadmium ion (site symmetry 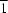 ). The cadmium ions are symmetrically doubly bridged by thiocyanate ions, leading to a polymeric chain motif. The octahedral coordination geometry of cadmium is completed by a pair of inversion-related water molecules. The 2-aminopyrimidine molecules are not coordinated to cadmium but are hydrogen-bonded to the coordinated water molecules and thiocyanate ions. There is also a pyrimidine–pyrimidine interaction via an N—H
). The cadmium ions are symmetrically doubly bridged by thiocyanate ions, leading to a polymeric chain motif. The octahedral coordination geometry of cadmium is completed by a pair of inversion-related water molecules. The 2-aminopyrimidine molecules are not coordinated to cadmium but are hydrogen-bonded to the coordinated water molecules and thiocyanate ions. There is also a pyrimidine–pyrimidine interaction via an N—H N hydrogen bond.
N hydrogen bond.
 N hydrogen bond.
N hydrogen bond.In the title complex, [Ni(NCS)2(H2O)4]·4C6H9N3·2H2O, the nickel(II) ion occupies an inversion centre and is octahedrally coordinated by two N-bonded thiocyanate ions and four water molecules. The two distinct 2-amino-4,6-dimethylpyrimidine molecules adopt distinct base-pairing dimerization patterns via different pairs of N—H N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H
N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H O, O—H
O, O—H S(thiocyanate) and N—H
S(thiocyanate) and N—H N(thiocyanate) hydrogen bonds.
N(thiocyanate) hydrogen bonds.
 N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H
N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H O, O—H
O, O—H S(thiocyanate) and N—H
S(thiocyanate) and N—H N(thiocyanate) hydrogen bonds.
N(thiocyanate) hydrogen bonds. The title compound, [Cu2(bnpp)2(bpa)2](ClO4)2·0.5CH3CN [bpa is bis(2-pyridylmethyl)amine (C12H13N3) and bnpp is bis(4-nitrophenyl)phosphate (C12H8N2O8P)], contains two CuII ions, each coordinated in a distorted square-pyramidal geometry formed by the tridentate chelating ligand bpa and two O-atom donors from two bnpp ligands. Two bnpp anions bridge two CuII ions to form the dinuclear complex cation, one O-atom donor of each bnpp coordinating in a basal site and another coordinating in the apical position with a Jahn–Teller elongated bond of 2.189 (4) or 2.244 (3) Å.
The title compound, 2-{(E)-3-[(E)-4-chlorophenylimino]-4-hydroxyphenyldiazenyl}benzoic acid hemihydrate, C20H14ClN3O3·0.5H2O, forms sheets, parallel to the (100) plane, that are linked by weak aromatic C—H O interactions. These sheets are in turn linked by strong hydrogen bonds to interstitial water molecules.
O interactions. These sheets are in turn linked by strong hydrogen bonds to interstitial water molecules.
 O interactions. These sheets are in turn linked by strong hydrogen bonds to interstitial water molecules.
O interactions. These sheets are in turn linked by strong hydrogen bonds to interstitial water molecules. The title compound, [Mg3(C9H16NO)6], represents a trimeric complex with all Mg atoms having octahedral coordination. The central Mg atom occupies a special position on a crystallographic inversion centre and is coordinated by six O atoms [Mg—O = 2.069 (2), 2.083 (2) and 2.072 (2) Å], all of which have bridging functions. Each of the peripheral Mg atoms is coordinated by three bridging O atoms [Mg—O = 2.090 (2), 2.086 (2) and 2.095 (2) Å] and three terminal N atoms of the butylimino groups [Mg—N = 2.206 (2), 2.202 (2) and 2.196 (2) Å].
The title compound, [Mn(C16H12N4O6)(CH4O)(H2O)]ClO4, arose as an unexpected oxidation product, and contains an MnIII cation coordinated by an equatorial salen-type ligand and Jahn–Teller-distorted axially coordinated water and methanol molecules. A network of O—H O and O—H
O and O—H N hydrogen bonds helps to establish the crystal packing.
N hydrogen bonds helps to establish the crystal packing.
 O and O—H
O and O—H N hydrogen bonds helps to establish the crystal packing.
N hydrogen bonds helps to establish the crystal packing.The structure of the title compound [systematic name: 2-hydroxy-6-methyl-3-(1-methylethyl)benzaldehyde oxime] C11H15NO2, exhibits intra- as well as intermolecular hydrogen bonding, involving participation of the phenolic OH group in intramolecular hydrogen bonding and the hydroxyl group of the oxime in intermolecular hydrogen bonding. The H atom of the phenolic hydroxyl group forms a strong O—H N intramolecular hydrogen bond with an O
N intramolecular hydrogen bond with an O N distance of 2.5788 (14) Å, which is in the middle of the expected range for such hydrogen bonds. The H atom of the hydroxyl group (in the oxime functionality) forms a weaker hydrogen bond with the phenolic hydroxyl group of a neighboring molecule [O
N distance of 2.5788 (14) Å, which is in the middle of the expected range for such hydrogen bonds. The H atom of the hydroxyl group (in the oxime functionality) forms a weaker hydrogen bond with the phenolic hydroxyl group of a neighboring molecule [O O = 2.8317 (14) Å], forming an extended chain, as expected for phenolic aldoximes which have bulky substituents on the aryl ring.
O = 2.8317 (14) Å], forming an extended chain, as expected for phenolic aldoximes which have bulky substituents on the aryl ring.
 N intramolecular hydrogen bond with an O
N intramolecular hydrogen bond with an O N distance of 2.5788 (14) Å, which is in the middle of the expected range for such hydrogen bonds. The H atom of the hydroxyl group (in the oxime functionality) forms a weaker hydrogen bond with the phenolic hydroxyl group of a neighboring molecule [O
N distance of 2.5788 (14) Å, which is in the middle of the expected range for such hydrogen bonds. The H atom of the hydroxyl group (in the oxime functionality) forms a weaker hydrogen bond with the phenolic hydroxyl group of a neighboring molecule [O O = 2.8317 (14) Å], forming an extended chain, as expected for phenolic aldoximes which have bulky substituents on the aryl ring.
O = 2.8317 (14) Å], forming an extended chain, as expected for phenolic aldoximes which have bulky substituents on the aryl ring.The title compound [systematic name: (6-methoxy-1,4a-dimethyl-1,2,3,4,4a,9,10,10a-octahydrophenanthren-1-yl)morpholin-4-yl-methanone], C22H31NO3, was synthesized from natural podocarpic acid via amidation of the acid chloride derivative. The crystal structure shows that the morpholino ring adopts a full chair conformation, while the amide N-C bond has partial double-bond character.

 journal menu
journal menu

















