The title complex, {[Cd(NCS)2(H2O)2]·2C6H9N3}n, crystalizes in the triclinic space group P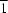 . The asymmetric unit contains one 2-amino-4,6-dimethylpyrimidine molecule, a thiocyanate ion, a water molecule and a cadmium ion (site symmetry
. The asymmetric unit contains one 2-amino-4,6-dimethylpyrimidine molecule, a thiocyanate ion, a water molecule and a cadmium ion (site symmetry 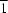 ). The cadmium ions are symmetrically doubly bridged by thiocyanate ions, leading to a polymeric chain motif. The octahedral coordination geometry of cadmium is completed by a pair of inversion-related water molecules. The 2-aminopyrimidine molecules are not coordinated to cadmium but are hydrogen-bonded to the coordinated water molecules and thiocyanate ions. There is also a pyrimidine–pyrimidine interaction via an N—H
). The cadmium ions are symmetrically doubly bridged by thiocyanate ions, leading to a polymeric chain motif. The octahedral coordination geometry of cadmium is completed by a pair of inversion-related water molecules. The 2-aminopyrimidine molecules are not coordinated to cadmium but are hydrogen-bonded to the coordinated water molecules and thiocyanate ions. There is also a pyrimidine–pyrimidine interaction via an N—H N hydrogen bond.
N hydrogen bond.
 N hydrogen bond.
N hydrogen bond.In the title complex, [Ni(NCS)2(H2O)4]·4C6H9N3·2H2O, the nickel(II) ion occupies an inversion centre and is octahedrally coordinated by two N-bonded thiocyanate ions and four water molecules. The two distinct 2-amino-4,6-dimethylpyrimidine molecules adopt distinct base-pairing dimerization patterns via different pairs of N—H N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H
N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H O, O—H
O, O—H S(thiocyanate) and N—H
S(thiocyanate) and N—H N(thiocyanate) hydrogen bonds.
N(thiocyanate) hydrogen bonds.
 N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H
N bonds. They are also hydrogen bonded to the coordinated and uncoordinated water molecules and thiocyanate ions through O—H O, O—H
O, O—H S(thiocyanate) and N—H
S(thiocyanate) and N—H N(thiocyanate) hydrogen bonds.
N(thiocyanate) hydrogen bonds. In the crystal structure of the title compound, 2,4-diamino-5-(4-chlorophenyl)-6-ethylpyrimidin-1-ium 3-carboxy-4-hydroxybenzenesulfonate monohydrate, C12H14ClN4+·C7H5O6S−·H2O, the asymmetric unit contains two crystallographically independent protonated pyrimethamine (PMN) molecules, a pair of sulfosalicylate anions and two water molecules. Both the PMN molecules are protonated at one of the pyrimidine N atoms. The component species interact through numerous intermolecular N—H O, N—H
O, N—H N and O—H
N and O—H O hydrogen bonds and C—H
O hydrogen bonds and C—H O interactions.
O interactions.
 O, N—H
O, N—H N and O—H
N and O—H O hydrogen bonds and C—H
O hydrogen bonds and C—H O interactions.
O interactions.In the title compound, 2C14H19N4O3+·C7H3NO42−·5H2O, the asymmetric unit contains two trimethoprim cations (A and B), a dipicolinate anion and five water molecules. One of the picolinate carboxylate groups interacts with cation A via a pair of N—H O hydrogen bonds, forming a ring motif
O hydrogen bonds, forming a ring motif 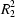 (8). A loop of O—H
(8). A loop of O—H O hydrogen bonds is formed between four water molecules and the two carboxylate groups to form a supramolecular chain along the a axis. Molecules A and B constitute a base pair via N—H
O hydrogen bonds is formed between four water molecules and the two carboxylate groups to form a supramolecular chain along the a axis. Molecules A and B constitute a base pair via N—H N hydrogen bonds, and these base pairs are cross-linked by N—H
N hydrogen bonds, and these base pairs are cross-linked by N—H O and C—H
O and C—H O hydrogen bonds.
O hydrogen bonds.
 O hydrogen bonds, forming a ring motif
O hydrogen bonds, forming a ring motif  O hydrogen bonds is formed between four water molecules and the two carboxylate groups to form a supramolecular chain along the a axis. Molecules A and B constitute a base pair via N—H
O hydrogen bonds is formed between four water molecules and the two carboxylate groups to form a supramolecular chain along the a axis. Molecules A and B constitute a base pair via N—H N hydrogen bonds, and these base pairs are cross-linked by N—H
N hydrogen bonds, and these base pairs are cross-linked by N—H O and C—H
O and C—H O hydrogen bonds.
O hydrogen bonds.Open  access
access
 access
accessIn the title compound [systematic name: 2,4-diamino-5-(3,4,5-trimethoxybenzyl)pyrimidinium tetrafluoroborate], C14H19N4O3+·BF4−, the trimethoprim (TMP) molecule is protonated at one of the pyrimidine N atoms. The protonated N atom and 2-amine group of the TMP cation interact with the tetrafluoroborate anion through a pair of N—H F hydrogen bonds [graph set R22(8)]. The inversion-related TMP cations are linked through a pair of N—H
F hydrogen bonds [graph set R22(8)]. The inversion-related TMP cations are linked through a pair of N—H N hydrogen bonds. The 2-amine group of one TMP cation and the 4-amine group of another cation are bridged by a methoxy O atom, via N—H
N hydrogen bonds. The 2-amine group of one TMP cation and the 4-amine group of another cation are bridged by a methoxy O atom, via N—H O hydrogen bonds.
O hydrogen bonds.
 F hydrogen bonds [graph set R22(8)]. The inversion-related TMP cations are linked through a pair of N—H
F hydrogen bonds [graph set R22(8)]. The inversion-related TMP cations are linked through a pair of N—H N hydrogen bonds. The 2-amine group of one TMP cation and the 4-amine group of another cation are bridged by a methoxy O atom, via N—H
N hydrogen bonds. The 2-amine group of one TMP cation and the 4-amine group of another cation are bridged by a methoxy O atom, via N—H O hydrogen bonds.
O hydrogen bonds.
 journal menu
journal menu














