The formation of the title compound, (S)-2-ammonio-1-butanol (2R,3R)-tartrate, C4H12NO+·C4H5O6-, the (S)-2-amino-1-butanol molecule is converted to a cationic form containing a positively charged amino group, and the tartaric acid molecule to a mono- or half-ionized tartrate anion. The structure is stabilized by a three-dimensional network of hydrogen bonds.
In the title structure, C14H10O5, the angle between the planes formed by the 2,4-dihydroxybenzoyl and o-benzoic acid moieties is 87.12 (4)°. In addition to an intramolecular O—H O hydrogen bond, intermolecular O—H
O hydrogen bond, intermolecular O—H O hydrogen bonds (H
O hydrogen bonds (H O = 1.76 and 1.89 Å) connect molecules to form a two-dimensional network parallel to (10
O = 1.76 and 1.89 Å) connect molecules to form a two-dimensional network parallel to (10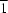 ).
).
 O hydrogen bond, intermolecular O—H
O hydrogen bond, intermolecular O—H O hydrogen bonds (H
O hydrogen bonds (H O = 1.76 and 1.89 Å) connect molecules to form a two-dimensional network parallel to (10
O = 1.76 and 1.89 Å) connect molecules to form a two-dimensional network parallel to (10The title compound, C18H19Br2N, was synthesized by N-alkylation of 1-bromohexane with 3,6-dibromo-9H-carbazole. The carbazole ring system is essentially planar and the n-hexyl chain is in the fully extended conformation.

 journal menu
journal menu












