1-[6-(1H-Pyrrolo[2,3-b]pyridin-1-yl)pyridin-2-yl]-1H-pyrrolo[2,3-b]pyridin-7-ium tetrachloridoferrate(III), (C19H14N5)[FeCl4], (II), and [2,6-bis(1H-pyrrolo[2,3-b]pyridin-1-yl-κN7)pyridine-κN]bis(nitrato-κO)copper(II), [Cu(NO3)2(C19H13N5)], (III), were prepared by self-assembly from FeCl3·6H2O or Cu(NO3)2·3H2O and 2,6-bis(1H-pyrrolo[2,3-b]pyridin-1-yl)pyridine [commonly called 2,6-bis(azaindole)pyridine, bap], C19H13N5, (I). Compound (I) crystallizes with Z′ = 2 in the P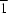 space group, with both independent molecules adopting a trans–trans conformation. Compound (II) is a salt complex with weak C—H
space group, with both independent molecules adopting a trans–trans conformation. Compound (II) is a salt complex with weak C—H Cl interactions giving rise to a zigzag network with π-stacking down the a axis. Complex (III) lies across a twofold rotation axis in the C2/c space group. The CuII center in (III) has an N3O2 trigonal–bipyramidal environment. The nitrate ligand coordinates in a monodentate fashion, while the bap ligand adopts a twisted tridentate binding mode. C—H
Cl interactions giving rise to a zigzag network with π-stacking down the a axis. Complex (III) lies across a twofold rotation axis in the C2/c space group. The CuII center in (III) has an N3O2 trigonal–bipyramidal environment. The nitrate ligand coordinates in a monodentate fashion, while the bap ligand adopts a twisted tridentate binding mode. C—H O interactions give rise to a ribbon motif.
O interactions give rise to a ribbon motif.
 Cl interactions giving rise to a zigzag network with π-stacking down the a axis. Complex (III) lies across a twofold rotation axis in the C2/c space group. The CuII center in (III) has an N3O2 trigonal–bipyramidal environment. The nitrate ligand coordinates in a monodentate fashion, while the bap ligand adopts a twisted tridentate binding mode. C—H
Cl interactions giving rise to a zigzag network with π-stacking down the a axis. Complex (III) lies across a twofold rotation axis in the C2/c space group. The CuII center in (III) has an N3O2 trigonal–bipyramidal environment. The nitrate ligand coordinates in a monodentate fashion, while the bap ligand adopts a twisted tridentate binding mode. C—H O interactions give rise to a ribbon motif.
O interactions give rise to a ribbon motif.catena-Poly[[[dichloridoiron(II)]-μ-N,N′-bis(2-pyridylmethylene)benzene-1,4-diamine] methanol disolvate], [FeCl2(C18H14N4)]·2CH3OH, forms a one-dimensional coordination polymer. The polymeric chains run parallel to the c axis. O—H Cl—Fe and C—H
Cl—Fe and C—H O hydrogen-bonding interactions with methanol solvent molecules stabilize the open supramolecular framework. Each FeII atom adopts an octahedral geometry coordinated by four N atoms from two N,N′-bis(2-pyridylmethylene)benzene-1,4-diamine ligands and completed by two cis Cl atoms. The compound has C2 (and Ci) molecular symmetry, which is coincident with the crystallographic twofold symmetry at (0, y,
O hydrogen-bonding interactions with methanol solvent molecules stabilize the open supramolecular framework. Each FeII atom adopts an octahedral geometry coordinated by four N atoms from two N,N′-bis(2-pyridylmethylene)benzene-1,4-diamine ligands and completed by two cis Cl atoms. The compound has C2 (and Ci) molecular symmetry, which is coincident with the crystallographic twofold symmetry at (0, y, 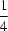 ). The one-dimensional structure is propagated via the crystallographic inversion center located at the benzene ring centroid (0,
). The one-dimensional structure is propagated via the crystallographic inversion center located at the benzene ring centroid (0, 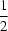 , 0).
, 0).
 Cl—Fe and C—H
Cl—Fe and C—H O hydrogen-bonding interactions with methanol solvent molecules stabilize the open supramolecular framework. Each FeII atom adopts an octahedral geometry coordinated by four N atoms from two N,N′-bis(2-pyridylmethylene)benzene-1,4-diamine ligands and completed by two cis Cl atoms. The compound has C2 (and Ci) molecular symmetry, which is coincident with the crystallographic twofold symmetry at (0, y,
O hydrogen-bonding interactions with methanol solvent molecules stabilize the open supramolecular framework. Each FeII atom adopts an octahedral geometry coordinated by four N atoms from two N,N′-bis(2-pyridylmethylene)benzene-1,4-diamine ligands and completed by two cis Cl atoms. The compound has C2 (and Ci) molecular symmetry, which is coincident with the crystallographic twofold symmetry at (0, y, 
 journal menu
journal menu










