In the structure of the title salt [systematic name: 3-(10,11-dihydro-5H-dibenzo[a,d][7]annulen-5-ylidene)-N,N-dimethylpropan-1-aminium 2,4,6-trinitrophenolate] of a tricyclic antidepressant, C20H24N+·C6H2N3O7−, the dimethylaminopropyl subunit possesses a classical static conformational disorder. The central cycloheptadiene ring adopts a bent conformation that is intermediate between boat and chair forms, leading to a butterfly shape for the hetero-tricyclic moiety. In a complementary fashion, donors from amitriptyline and acceptors from picrate form intermolecular C—H O hydrogen bonds and N—H
O hydrogen bonds and N—H O salt bridges. These hydrogen bonds cluster amitriptyline and picrate ions into a closed R44(36) hetero-tetramer, whereas intermolecular C—H
O salt bridges. These hydrogen bonds cluster amitriptyline and picrate ions into a closed R44(36) hetero-tetramer, whereas intermolecular C—H π interactions between amitriptyline ions cluster them into homo-dimers. Significant π–π stacking interactions are also observed between aromatic rings of amitriptyline and picrate, and these, combined with the C—H
π interactions between amitriptyline ions cluster them into homo-dimers. Significant π–π stacking interactions are also observed between aromatic rings of amitriptyline and picrate, and these, combined with the C—H π interactions, associate molecules into linear arrays along the [1
π interactions, associate molecules into linear arrays along the [1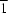 1] direction.
1] direction.
 O hydrogen bonds and N—H
O hydrogen bonds and N—H O salt bridges. These hydrogen bonds cluster amitriptyline and picrate ions into a closed R44(36) hetero-tetramer, whereas intermolecular C—H
O salt bridges. These hydrogen bonds cluster amitriptyline and picrate ions into a closed R44(36) hetero-tetramer, whereas intermolecular C—H π interactions between amitriptyline ions cluster them into homo-dimers. Significant π–π stacking interactions are also observed between aromatic rings of amitriptyline and picrate, and these, combined with the C—H
π interactions between amitriptyline ions cluster them into homo-dimers. Significant π–π stacking interactions are also observed between aromatic rings of amitriptyline and picrate, and these, combined with the C—H π interactions, associate molecules into linear arrays along the [1
π interactions, associate molecules into linear arrays along the [1
 journal menu
journal menu









