Both 6-(1H-indol-3-yl)-3-methyl-4-(4-methylphenyl)-1-phenyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile and 6-(1H-indol-3-yl)-3-methyl-4-(4-methoxyphenyl)-1-phenyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile crystallize from dimethylformamide solutions as stoichiometric 1:1 solvates, viz. C29H21N5·C3H7NO, (I), and C29H21N5O·C3H7NO, (II), respectively; however, 6-(1H-indol-3-yl)-3-methyl-1-phenyl-4-(3,4,5-trimethoxyphenyl)-1H-pyrazolo[3,4-b]pyridine-5-carbonitrile, C31H25N5O3, (III), crystallizes in the unsolvated form. The heterocyclic components of (I) are linked by C—H π(arene) hydrogen bonds to form cyclic centrosymmetric dimers, from which the solvent molecules are pendent, linked by N—H
π(arene) hydrogen bonds to form cyclic centrosymmetric dimers, from which the solvent molecules are pendent, linked by N—H O hydrogen bonds. In (II), the heterocyclic components are linked by a combination of C—H
O hydrogen bonds. In (II), the heterocyclic components are linked by a combination of C—H N and C—H
N and C—H π(arene) hydrogen bonds into chains containing two types of centrosymmetric ring, and the pendent solvent molecules are linked to these chains by N—H
π(arene) hydrogen bonds into chains containing two types of centrosymmetric ring, and the pendent solvent molecules are linked to these chains by N—H O hydrogen bonds. Molecules of (III) are linked into simple C(12) chains by an N—H
O hydrogen bonds. Molecules of (III) are linked into simple C(12) chains by an N—H O hydrogen bond, and these chains are weakly linked into pairs by an aromatic π–π stacking interaction.
O hydrogen bond, and these chains are weakly linked into pairs by an aromatic π–π stacking interaction.
 π(arene) hydrogen bonds to form cyclic centrosymmetric dimers, from which the solvent molecules are pendent, linked by N—H
π(arene) hydrogen bonds to form cyclic centrosymmetric dimers, from which the solvent molecules are pendent, linked by N—H O hydrogen bonds. In (II), the heterocyclic components are linked by a combination of C—H
O hydrogen bonds. In (II), the heterocyclic components are linked by a combination of C—H N and C—H
N and C—H π(arene) hydrogen bonds into chains containing two types of centrosymmetric ring, and the pendent solvent molecules are linked to these chains by N—H
π(arene) hydrogen bonds into chains containing two types of centrosymmetric ring, and the pendent solvent molecules are linked to these chains by N—H O hydrogen bonds. Molecules of (III) are linked into simple C(12) chains by an N—H
O hydrogen bonds. Molecules of (III) are linked into simple C(12) chains by an N—H O hydrogen bond, and these chains are weakly linked into pairs by an aromatic π–π stacking interaction.
O hydrogen bond, and these chains are weakly linked into pairs by an aromatic π–π stacking interaction.The molecules of 2-amino-4,6-bis[N-methyl-N-(4-methylphenyl)amino]pyrimidine-5-carbaldehyde, C21H23N5O, (I), and 2-amino-4-(indolin-1-yl)-6-methoxypyrimidine-5-carbaldehyde, C14H14N4O2, (II), which crystallizes with Z' = 2 in the space group P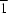 , exhibit polarized electronic structures. Molecules of (I) are linked by a combination of N-H
, exhibit polarized electronic structures. Molecules of (I) are linked by a combination of N-H O, C-H
O, C-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds into a three-dimensional framework structure, while those of (II) are linked into sheets by a combination of two N-H
(arene) hydrogen bonds into a three-dimensional framework structure, while those of (II) are linked into sheets by a combination of two N-H O hydrogen bonds and one N-H
O hydrogen bonds and one N-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond.
(arene) hydrogen bond.
 O, C-H
O, C-H O and C-H
O and C-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bonds into a three-dimensional framework structure, while those of (II) are linked into sheets by a combination of two N-H
(arene) hydrogen bonds into a three-dimensional framework structure, while those of (II) are linked into sheets by a combination of two N-H O hydrogen bonds and one N-H
O hydrogen bonds and one N-H
![[pi]](/logos/entities/pi_rmgif.gif) (arene) hydrogen bond.
(arene) hydrogen bond.In the title compound, C23H24N4O3, the pyrimidine ring adopts an almost perfect boat conformation, and the bond distances provide evidence for some polarization of the molecular-electronic structure. Two independent N-H O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
 O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
O hydrogen bonds link the molecules into chains of edge-fused R24(8) and R22(20) rings.
 journal menu
journal menu











