Open  access
access
 access
accessThe asymmetric unit of the title compound, C26H25ClN2O3, contains two independent molecules (A and B). The conformation of the two molecules differs essentially in the dihedral angle involving the two benzene rings. They are inclined to one another by 52.47 (10) in A and by 31.75 (11)° in B. In both molecules, the six-membered piperidin-3-one rings have chair conformations. In molecule A, all four five-membered rings have twist conformations. In molecule B, only three of the four five-membered rings have twist conformations. The fourth, of the inden-1-one moiety, has an envelope conformation with the spiro C atom, bonded to the N atom of the pyrrolidine ring, as the flap. A weak intramolecular O—H N hydrogen bond occurs in each independent molecule while a C—H
N hydrogen bond occurs in each independent molecule while a C—H O interaction is also observed in molecule A. In the crystal, pairs of O—H
O interaction is also observed in molecule A. In the crystal, pairs of O—H O hydrogen bonds link the molecules, forming inversion dimers with graph-set motif R22(12). These dimers are further interconnected by C—H
O hydrogen bonds link the molecules, forming inversion dimers with graph-set motif R22(12). These dimers are further interconnected by C—H O and C—H
O and C—H π interactions, forming a three-dimensional network.
π interactions, forming a three-dimensional network.
 N hydrogen bond occurs in each independent molecule while a C—H
N hydrogen bond occurs in each independent molecule while a C—H O interaction is also observed in molecule A. In the crystal, pairs of O—H
O interaction is also observed in molecule A. In the crystal, pairs of O—H O hydrogen bonds link the molecules, forming inversion dimers with graph-set motif R22(12). These dimers are further interconnected by C—H
O hydrogen bonds link the molecules, forming inversion dimers with graph-set motif R22(12). These dimers are further interconnected by C—H O and C—H
O and C—H π interactions, forming a three-dimensional network.
π interactions, forming a three-dimensional network.Open  access
access
 access
accessThe title compound comprises a 2-iminopyridine ring fused with a cyclooctane ring, which adopts a twist boat–chair conformation. Intermolecular C—H N interactions form
N interactions form 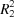 (14) ring motifs and molecules are further connected by weak C—H
(14) ring motifs and molecules are further connected by weak C—H π interactions.
π interactions.
 N interactions form
N interactions form  π interactions.
π interactions.Open  access
access
 access
accessThe asymmetric unit of the title compound, C19H16N2O5S, contains four independent molecules (A, B, C and D), with two molecules (B and D) displaying disorder in their methylsulfanyl groups [occupancy ratios of 0.797 (11):0.203 (11) and 0.85 (2):0.15 (2)]. The nitro groups are twisted slightly out of the planes of the 2-pyrroline rings to which they are bonded with dihedral angles of 10.17 (1), 8.01 (1), 9.44 (1) and 8.87 (1)° in molecules A, B, C and D, respectively. The 2-pyrroline rings are almost orthogonal to the attached tolyl rings, forming dihedral angles of 73.44 (1), 81.21 (1), 88.18 (8) and 73.94 (1)° for molecules A, B, C and D, respectively. A weak intramolecular O—H O interaction is observed in molecules B and C. The two hydroxy groups in each molecule are involved in intermolecular O—H
O interaction is observed in molecules B and C. The two hydroxy groups in each molecule are involved in intermolecular O—H O hydrogen bonding. In the crystal, molecules are connected via O—H
O hydrogen bonding. In the crystal, molecules are connected via O—H O and C—H
O and C—H O hydrogen bonds, forming a complex three-dimensional network.
O hydrogen bonds, forming a complex three-dimensional network.
 O interaction is observed in molecules B and C. The two hydroxy groups in each molecule are involved in intermolecular O—H
O interaction is observed in molecules B and C. The two hydroxy groups in each molecule are involved in intermolecular O—H O hydrogen bonding. In the crystal, molecules are connected via O—H
O hydrogen bonding. In the crystal, molecules are connected via O—H O and C—H
O and C—H O hydrogen bonds, forming a complex three-dimensional network.
O hydrogen bonds, forming a complex three-dimensional network.Open  access
access
 access
accessTwo isotypic title compounds comprise a 2-iminopyridine ring fused with a cyclooctane ring. In one compound, the cyclooctane ring adopts a twisted chair–chair conformation, while in the second, this ring adopts a twisted boat–chair conformation.
Open  access
access
 access
accessIn the title compound C21H17N5O4·C2H6OS, the central six-membered ring derived from 1,4-dihydropyridine adopts a distorted boat conformation with a small puckering amplitude of 0.127 (3) Å. The sums of bond angles around the pyridine N atom [358.7 (2)°] and the other imidazolidine N atom [60 (2)°] indicate that these atoms are in sp2 hybridization, leading to an essentially planar imidazolidine ring. The last heterocycle, an oxindole moiety, is also nearly planar with an r.m.s. deviation of 0.0185 (1) Å. The amine NH2 group forms an intramolecular hydrogen bond with the benzoyl group, giving a S(6) motif. In the crystal, N—H O hydrogen bonds lead to the formation of chains along the c-axis direction. Within the chains there are further N—H
O hydrogen bonds lead to the formation of chains along the c-axis direction. Within the chains there are further N—H O and C—H
O and C—H O hydrogen bonds enclosing R22(14) ring motifs. The chains are linked via N—H
O hydrogen bonds enclosing R22(14) ring motifs. The chains are linked via N—H O and C—H
O and C—H O hydrogen bonds involving the dimethyl sulfoxide solvent molecule which acts as both an acceptor and a donor..
O hydrogen bonds involving the dimethyl sulfoxide solvent molecule which acts as both an acceptor and a donor..
 O hydrogen bonds lead to the formation of chains along the c-axis direction. Within the chains there are further N—H
O hydrogen bonds lead to the formation of chains along the c-axis direction. Within the chains there are further N—H O and C—H
O and C—H O hydrogen bonds enclosing R22(14) ring motifs. The chains are linked via N—H
O hydrogen bonds enclosing R22(14) ring motifs. The chains are linked via N—H O and C—H
O and C—H O hydrogen bonds involving the dimethyl sulfoxide solvent molecule which acts as both an acceptor and a donor..
O hydrogen bonds involving the dimethyl sulfoxide solvent molecule which acts as both an acceptor and a donor..Open  access
access
 access
accessIn the title compound, C20H22N2O2, the central pyridine ring forms a dihedral angle of 76.32 (8)° with the pseudo-axial benzene ring. The cyclooctane ring adopts a twisted boat chair conformation. In the crystal, weak intermolecular C—H π interactions between inversion-related molecules result in the formation of linear double chains along the b-axis direction.
π interactions between inversion-related molecules result in the formation of linear double chains along the b-axis direction.
 π interactions between inversion-related molecules result in the formation of linear double chains along the b-axis direction.
π interactions between inversion-related molecules result in the formation of linear double chains along the b-axis direction.Open  access
access
 access
accessIn the title compound, C22H18BrN5O4, the central six-membered ring, derived from 1,4-dihydropyridine, adopts a distorted boat conformation with a puckering amplitude of 0.197 (3) Å, the imidazole ring adopts a twisted conformation with a puckering amplitude of 0.113 (3) Å, and the oxindole moiety is planar with an r.m.s. deviation of 0.0125 Å. Two intramolecular N—H O hydrogen bonds are formed, each closing an S(6) loop. In the crystal, strong N—H
O hydrogen bonds are formed, each closing an S(6) loop. In the crystal, strong N—H O hydrogen bonds lead to the formation of zigzag chains along the c axis. These are consolidated in the three-dimensional crystal packing by weak N—H
O hydrogen bonds lead to the formation of zigzag chains along the c axis. These are consolidated in the three-dimensional crystal packing by weak N—H O hydrogen bonding, as well as by C—H
O hydrogen bonding, as well as by C—H O, C—H
O, C—H Br and C—H
Br and C—H π interactions. A small region of electron density well removed from the main molecule was removed with the SQUEEZE procedure in PLATON [Spek (2009). Acta Cryst. D65, 148–155] following unsuccessful attempts to model it as a plausible solvent molecule. The unit-cell characteristics do not take into account this feature of the structure.
π interactions. A small region of electron density well removed from the main molecule was removed with the SQUEEZE procedure in PLATON [Spek (2009). Acta Cryst. D65, 148–155] following unsuccessful attempts to model it as a plausible solvent molecule. The unit-cell characteristics do not take into account this feature of the structure.
 O hydrogen bonds are formed, each closing an S(6) loop. In the crystal, strong N—H
O hydrogen bonds are formed, each closing an S(6) loop. In the crystal, strong N—H O hydrogen bonds lead to the formation of zigzag chains along the c axis. These are consolidated in the three-dimensional crystal packing by weak N—H
O hydrogen bonds lead to the formation of zigzag chains along the c axis. These are consolidated in the three-dimensional crystal packing by weak N—H O hydrogen bonding, as well as by C—H
O hydrogen bonding, as well as by C—H O, C—H
O, C—H Br and C—H
Br and C—H π interactions. A small region of electron density well removed from the main molecule was removed with the SQUEEZE procedure in PLATON [Spek (2009). Acta Cryst. D65, 148–155] following unsuccessful attempts to model it as a plausible solvent molecule. The unit-cell characteristics do not take into account this feature of the structure.
π interactions. A small region of electron density well removed from the main molecule was removed with the SQUEEZE procedure in PLATON [Spek (2009). Acta Cryst. D65, 148–155] following unsuccessful attempts to model it as a plausible solvent molecule. The unit-cell characteristics do not take into account this feature of the structure.Keywords: crystal structure.
Open  access
access
 access
accessIn the title compound, C19H19FN2O, the cyclooctene ring adopts a twisted boat-chair conformation. The dihedral angle between the plane of the fluorophenyl substituent and that of the pyridine ring is 76.39 (8)°. The F and ortho-H atoms of the fluorobenzene ring are disordered, with occupancy factors of 0.226 (5) and 0.774 (5). In the crystal, no significant interactions are observed between the molecules beyond van der Waals contacts.
Open  access
access
 access
accessIn the title compound, C24H14BrN3S, the dihedral angles between the planes of the pyridine ring and the pendant thiophene ring, the indole ring system (r.m.s. deviation = 0.022 Å) and the bromobenzene ring are 9.37 (17), 21.90 (12) and 69.01 (15)°, respectively. The approximate coplanarity of the central ring and the indole ring system is supported by two intramolecular C—H N interactions. In the crystal, inversion dimers linked by pairs of N—H
N interactions. In the crystal, inversion dimers linked by pairs of N—H N hydrogen bonds generate R22(16) loops and the dimers are linked by C—H
N hydrogen bonds generate R22(16) loops and the dimers are linked by C—H π and aromatic π–π stacking [shortest centroid–centroid separation = 3.729 (3) Å] into a three-dimensional network.
π and aromatic π–π stacking [shortest centroid–centroid separation = 3.729 (3) Å] into a three-dimensional network.
 N interactions. In the crystal, inversion dimers linked by pairs of N—H
N interactions. In the crystal, inversion dimers linked by pairs of N—H N hydrogen bonds generate R22(16) loops and the dimers are linked by C—H
N hydrogen bonds generate R22(16) loops and the dimers are linked by C—H π and aromatic π–π stacking [shortest centroid–centroid separation = 3.729 (3) Å] into a three-dimensional network.
π and aromatic π–π stacking [shortest centroid–centroid separation = 3.729 (3) Å] into a three-dimensional network.Open  access
access
 access
accessThe asymmetric unit of the title compound, C21H15Cl2N5O4, contains two independent molecules (A and B) having similar conformations. The amine (NH2) group forms an intramolecular hydrogen bond with the benzoyl group, giving an S(6) ring motif in both molecules. The central six-membered rings adopt sofa conformations and the imidazole rings are planar (r.m.s deviations = 0.0150 and 0.0166 Å). The pyridine and imidazole rings are inclined to one another by 3.54 (1) and 3.03 (1)° in molecules A and B, respectively. In the crystal, molecules are linked by N—H O hydrogen bonds, forming chains along the a axis which enclose R22(16) ring motifs. The rings are linked by weak N—H
O hydrogen bonds, forming chains along the a axis which enclose R22(16) ring motifs. The rings are linked by weak N—H O and C—H
O and C—H O hydrogen bonds and C—H
O hydrogen bonds and C—H π interactions forming sheets lying parallel to (001). A region of disordered electron density, most probably disordered solvent molecules, occupying voids of ca 753 Å3 for an electron count of 260, was treated using the SQUEEZE routine in PLATON [Spek (2009). Acta Cryst. D65, 148–155]. Their formula mass and unit-cell characteristics were not taken into account during refinement.
π interactions forming sheets lying parallel to (001). A region of disordered electron density, most probably disordered solvent molecules, occupying voids of ca 753 Å3 for an electron count of 260, was treated using the SQUEEZE routine in PLATON [Spek (2009). Acta Cryst. D65, 148–155]. Their formula mass and unit-cell characteristics were not taken into account during refinement.
 O hydrogen bonds, forming chains along the a axis which enclose R22(16) ring motifs. The rings are linked by weak N—H
O hydrogen bonds, forming chains along the a axis which enclose R22(16) ring motifs. The rings are linked by weak N—H O and C—H
O and C—H O hydrogen bonds and C—H
O hydrogen bonds and C—H π interactions forming sheets lying parallel to (001). A region of disordered electron density, most probably disordered solvent molecules, occupying voids of ca 753 Å3 for an electron count of 260, was treated using the SQUEEZE routine in PLATON [Spek (2009). Acta Cryst. D65, 148–155]. Their formula mass and unit-cell characteristics were not taken into account during refinement.
π interactions forming sheets lying parallel to (001). A region of disordered electron density, most probably disordered solvent molecules, occupying voids of ca 753 Å3 for an electron count of 260, was treated using the SQUEEZE routine in PLATON [Spek (2009). Acta Cryst. D65, 148–155]. Their formula mass and unit-cell characteristics were not taken into account during refinement.Open  access
access
 access
accessIn the title compound, C26H16ClN3, the dihedral angles between the central pyridine ring and the pendant phenyl, chlorobenzene and indole rings are 18.52 (12), 48.97 (11) and 21.20 (10)°, respectively. An intramolecular C—H Nc (c = cyanide) hydrogen bond occurs. In the crystal, inversion dimers linked by pairs of N—H
Nc (c = cyanide) hydrogen bond occurs. In the crystal, inversion dimers linked by pairs of N—H Nc hydrogen bonds generate R22(16) loops.
Nc hydrogen bonds generate R22(16) loops.
 Nc (c = cyanide) hydrogen bond occurs. In the crystal, inversion dimers linked by pairs of N—H
Nc (c = cyanide) hydrogen bond occurs. In the crystal, inversion dimers linked by pairs of N—H Nc hydrogen bonds generate R22(16) loops.
Nc hydrogen bonds generate R22(16) loops.Open  access
access
 access
accessIn the title compound, C27H19N3O, the dihedral angles between the plane of the pyridine ring and those of the indole (r.m.s. deviation = 0.018 Å), phenyl and methoxybenzene substituents are 33.60 (6), 25.28 (7) and 49.31 (7)°, respectively. The N atom of the carbonitrile group is significantly displaced [0.288 (2) Å] from the plane of the pyridine ring, perhaps due to steric crowding. In the crystal, inversion dimers linked by pairs of N—H Nn (n = nitrile) hydrogen bonds generate R22(16) loops. Aromatic π–π stacking [centroid–centroid separation = 3.6906 (7) Å] and very weak C—H
Nn (n = nitrile) hydrogen bonds generate R22(16) loops. Aromatic π–π stacking [centroid–centroid separation = 3.6906 (7) Å] and very weak C—H π interactions are also observed".
π interactions are also observed".
 Nn (n = nitrile) hydrogen bonds generate R22(16) loops. Aromatic π–π stacking [centroid–centroid separation = 3.6906 (7) Å] and very weak C—H
Nn (n = nitrile) hydrogen bonds generate R22(16) loops. Aromatic π–π stacking [centroid–centroid separation = 3.6906 (7) Å] and very weak C—H π interactions are also observed".
π interactions are also observed".
 journal menu
journal menu





















