The structure of sodium calcium magnesium iron phosphate, (Na0,38,Ca0,31)MgFe2P3O12, consists of an [MgFe2(PO4)3]- framework, built up by chains of edge-sharing (FeO6-FeO6-MgO6) octahedra running along the [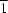 01] direction. The chains are linked together via the common corners of phosphate tetrahedra to form sheets parallel to the ab plane. Adjacent sheets are also connected through PO4 tetrahedra, thus giving rise to an open framework with two different kinds of channels where the Na+ and Ca2+ cations are located.
01] direction. The chains are linked together via the common corners of phosphate tetrahedra to form sheets parallel to the ab plane. Adjacent sheets are also connected through PO4 tetrahedra, thus giving rise to an open framework with two different kinds of channels where the Na+ and Ca2+ cations are located.
The title compound, 2C6H18N22+·P4O124−·2H2O, has been synthesized at room temperature and studied by single-crystal X-ray diffraction. The asymmetric unit consists of two hexanediaminium cations, four PO4 tetrahedra and two water molecules. The organic cations are linked to the cyclic P4O124− anions via N—H O hydrogen bonds to form two-dimensional layers, and are further linked by water molecules to form a three-dimensional network.
O hydrogen bonds to form two-dimensional layers, and are further linked by water molecules to form a three-dimensional network.
 O hydrogen bonds to form two-dimensional layers, and are further linked by water molecules to form a three-dimensional network.
O hydrogen bonds to form two-dimensional layers, and are further linked by water molecules to form a three-dimensional network.The title compound, C13H16N22+·0.5P4O124−, has been prepared at room temperature and studied by single-crystal X-ray diffraction. The asymmetric unit consists of one 4,4′-diphenylmethylendiammonium organic cation and two PO4 tetrahedra that form one half of a cyclic P4O124− anion that lies about an inversion centre. The P4O12 rings are linked through N—H O hydrogen bonds to build layers parallel to the ab plane. The two NH3+ groups at the 4- and 4′-positions of the cations link these layers into a three-dimensional network.
O hydrogen bonds to build layers parallel to the ab plane. The two NH3+ groups at the 4- and 4′-positions of the cations link these layers into a three-dimensional network.
 O hydrogen bonds to build layers parallel to the ab plane. The two NH3+ groups at the 4- and 4′-positions of the cations link these layers into a three-dimensional network.
O hydrogen bonds to build layers parallel to the ab plane. The two NH3+ groups at the 4- and 4′-positions of the cations link these layers into a three-dimensional network.
 journal menu
journal menu












