Corrigendum to Acta Cryst. (2005), E61, o3523–o3525.
In the title compound, C18H20O5, one of the four methoxy groups is twisted out of the naphthalene ring plane as a result of steric hindrance. The orientation of the butenone substituent with respect to the attached ring is anticlinal.
The title compound, C17H14O6, has been isolated for the first time from Rhazya stricta. The dihedral angle between the mean planes through the benzopyran ring system and o-anisole group is 59.78 (5)°. The molecular packing is stabilized by O—H O and C—H
O and C—H O hydrogen bonds.
O hydrogen bonds.
 O and C—H
O and C—H O hydrogen bonds.
O hydrogen bonds.The title compound, C8H15NO7, the methyl carbamate of β-D-glucosamine has been synthesized, in moderate yield, from the reaction of β-D-glucosamine hydrochloride and methyl chloroformate. There are two crystallographically independent molecules in the asymmetric unit. In both molecules, the pyranose ring adopts a slightly distorted chair conformation. In the crystal structure, molecules are packed along the a axis, with intra- and intermolecular O—H O, N—H
O, N—H O and C—H
O and C—H O hydrogen bonds.
O hydrogen bonds.
 O, N—H
O, N—H O and C—H
O and C—H O hydrogen bonds.
O hydrogen bonds.The title compound, 2-hydroxy-2-(4-hydroxyphenyl)-N-methylethanaminium chloride, C9H14NO2+·Cl−, has been isolated for the first time from the genus Harrisonia. Its structure is stabilized by extensive intra- and intermolecular hydrogen bonds of type O—H O, N—H
O, N—H O, N—H
O, N—H Cl and C—H
Cl and C—H Cl, resulting in a three-dimensional network. Additional weak π–π stacking interactions between adjacent molecules further stabilize the crystal structure.
Cl, resulting in a three-dimensional network. Additional weak π–π stacking interactions between adjacent molecules further stabilize the crystal structure.
 O, N—H
O, N—H O, N—H
O, N—H Cl and C—H
Cl and C—H Cl, resulting in a three-dimensional network. Additional weak π–π stacking interactions between adjacent molecules further stabilize the crystal structure.
Cl, resulting in a three-dimensional network. Additional weak π–π stacking interactions between adjacent molecules further stabilize the crystal structure.The title compound, C16H16N2O2, was synthesized by the condensation of aniline and succinic anhydride. The molecule lies across a crystallographic inversion centre. The dihedral angle between the benzene and acetamide planes is 33.36 (7)°. N—H O hydrogen bonds link the molecules into chains along [010].
O hydrogen bonds link the molecules into chains along [010].
 O hydrogen bonds link the molecules into chains along [010].
O hydrogen bonds link the molecules into chains along [010].The title compound, C20H32O4, was isolated from Pulicaria unduleta. It has an ent-kaurane diterpeniod ring system. In the crystal structure, the molecules are linked via O—H O hydrogen bonds into a ribbon structure.
O hydrogen bonds into a ribbon structure.
 O hydrogen bonds into a ribbon structure.
O hydrogen bonds into a ribbon structure.The title compound, C12H13NO5, was synthesized by the condensation of methyl anthranilate and succinic anhydride. The dihedral angle between the phenylacetamide and carboxylic acid (–C—COOH) planes is 80.47 (5)°. In the crystal structure, inversion-related molecules form an O—H O hydrogen-bonded dimer. Adjacent dimers are interlinked by C—H
O hydrogen-bonded dimer. Adjacent dimers are interlinked by C—H O hydrogen bonds to form a chain along [110].
O hydrogen bonds to form a chain along [110].
 O hydrogen-bonded dimer. Adjacent dimers are interlinked by C—H
O hydrogen-bonded dimer. Adjacent dimers are interlinked by C—H O hydrogen bonds to form a chain along [110].
O hydrogen bonds to form a chain along [110].The title compound, C28H27O10Cl·CH3OH·H2O, was isolated from Physalis minima. The rigid molecule consists of eight fused rings involving three lactones. The spiro-fused γ-lactone rings are in half-chair and envelope conformations. The spiro-fused γ-lactone rings are fused to a cyclohexene ring, which is in a half-chair conformation. The outermost cyclohexene ring and δ-lactone rings adopt half-chair and envelope conformations, respectively. Intra- and intermolecular O—H O, C—H
O, C—H Cl and C—H
Cl and C—H O hydrogen bonds are observed.
O hydrogen bonds are observed.
 O, C—H
O, C—H Cl and C—H
Cl and C—H O hydrogen bonds are observed.
O hydrogen bonds are observed.In the title compound, C35H42O4, the chromene ring is in a distorted half-chair conformation and the cyclohexadienone ring adopts a screw-boat conformation. Intramolecular O—H O hydrogen bonds involving the carbonyl and hydroxyl groups are observed.
O hydrogen bonds involving the carbonyl and hydroxyl groups are observed.
 O hydrogen bonds involving the carbonyl and hydroxyl groups are observed.
O hydrogen bonds involving the carbonyl and hydroxyl groups are observed.The title compound, C13H10O5, is a benzophenone derivative containing phloroglucinol and p-hydroxybenzoyl groups. The p-hydroxybenzoyl group is twisted out of the plane of the phloroglucinol group as a result of steric hindrance [dihedral angle = 50.2 (1)°]. In the crystal packing, screw-related molecules are linked by O—H O hydrogen bonds to form a zigzag layer parallel to (10
O hydrogen bonds to form a zigzag layer parallel to (10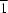 ). Adjacent layers are interlinked by O—H
). Adjacent layers are interlinked by O—H O hydrogen bonds involving the solvent water molecules.
O hydrogen bonds involving the solvent water molecules.
 O hydrogen bonds to form a zigzag layer parallel to (10
O hydrogen bonds to form a zigzag layer parallel to (10 O hydrogen bonds involving the solvent water molecules.
O hydrogen bonds involving the solvent water molecules.The title compound, C17H12O6, forms an infinite one-dimensional zigzag-like chain developing parallel to the b axis through O—H O hydrogen bonds. The chains are stacked along the c axis. The crystal structure is further stabilized by weak π–π and C—H
O hydrogen bonds. The chains are stacked along the c axis. The crystal structure is further stabilized by weak π–π and C—H π interactions.
π interactions.
 O hydrogen bonds. The chains are stacked along the c axis. The crystal structure is further stabilized by weak π–π and C—H
O hydrogen bonds. The chains are stacked along the c axis. The crystal structure is further stabilized by weak π–π and C—H π interactions.
π interactions.The title compound, C23H24O6, a xanthone derivative, was isolated from Garcinia polyantha Oliver. The orientation of the 3,7-dimethylocta-2,6-dienyl substituent with respect to the xanthone ring system is (+)synclinal. The crystal packing is stabilized by O—H O and C—H
O and C—H O intermolecular hydrogen bonds and π–π interactions.
O intermolecular hydrogen bonds and π–π interactions.
 O and C—H
O and C—H O intermolecular hydrogen bonds and π–π interactions.
O intermolecular hydrogen bonds and π–π interactions.
 journal menu
journal menu






















